Mastering Sequential ChIP (ChIP-reChIP): A Step-by-Step Guide to Mapping Bivalent Chromatin Domains
This comprehensive guide details the Chromatin Immunoprecipitation followed by re-Chromatin Immunoprecipitation (ChIP-reChIP) protocol, specifically optimized for the precise co-localization analysis of bivalent histone marks such as H3K4me3 and H3K27me3.
Mastering Sequential ChIP (ChIP-reChIP): A Step-by-Step Guide to Mapping Bivalent Chromatin Domains
Abstract
This comprehensive guide details the Chromatin Immunoprecipitation followed by re-Chromatin Immunoprecipitation (ChIP-reChIP) protocol, specifically optimized for the precise co-localization analysis of bivalent histone marks such as H3K4me3 and H3K27me3. Targeting researchers and drug development professionals, it covers the foundational biology of bivalent domains, provides a robust methodological workflow, addresses critical troubleshooting and optimization strategies, and validates the protocol against alternative techniques. The article aims to empower scientists to accurately interrogate the complex epigenetic landscape governing cellular plasticity and disease, offering practical insights for both basic and translational research.
Decoding Bivalent Chromatin: The Biological Rationale for Sequential ChIP
Bivalent chromatin domains, first characterized in embryonic stem cells (ESCs), are defined by the co-presence of the active mark histone H3 lysine 4 trimethylation (H3K4me3) and the repressive mark histone H3 lysine 27 trimethylation (H3K27me3) on the same nucleosome. This unique combinatorial signature "poises" key developmental regulator genes for rapid activation or stable silencing upon differentiation cues. Understanding this epigenetic state is critical for research in development, cellular reprogramming, and disease, particularly cancer, where bivalency is often dysregulated. Within the broader thesis on advanced chromatin analysis, this document details the application of Chromatin Immunoprecipitation followed by Re-ChIP (ChIP-reChIP) as the definitive protocol for directly demonstrating the simultaneous presence of these opposing marks on individual nucleosomes, moving beyond correlative co-localization studies.
Core Concepts and Quantitative Data
Table 1: Key Characteristics of Bivalent Histone Marks and Their Effectors
| Feature | H3K4me3 | H3K27me3 | Bivalent Domain (H3K4me3 & H3K27me3) |
|---|---|---|---|
| Associated State | Active Transcription | Facultative Heterochromatin / Repression | Poised/Pluripotent State |
| Primary Writers | COMPASS family (e.g., MLL1-4, SET1A/B) | Polycomb Repressive Complex 2 (PRC2) | Co-occupancy of COMPASS and PRC2 |
| Primary Erasers | KDM5 family, LSD1 | KDM6 family (UTX, JMJD3) | - |
| Primary Readers | TAF3, ING family, CHD1 | CBX family (in PRC1), Polycomb-like proteins | - |
| Genomic Location | Promoters of active genes | Promoters of silenced genes | Promoters of key developmental regulators (e.g., HOX, PAX, SOX families) |
| Functional Outcome | Promotes transcription initiation | Silences gene expression | Maintains low-level transcription or full repression; permits rapid fate decisions |
Table 2: Prevalence of Bivalent Domains Across Cell Types (Representative Data)
| Cell Type | Approx. Number of Bivalent Domains | Key Genomic Targets | Notes |
|---|---|---|---|
| Mouse Embryonic Stem Cells (mESCs) | ~2,200 - 3,500 | Homeobox (Hox), Transcription factors | Canonical system for bivalency; ~8% of H3K4me3 peaks are bivalent. |
| Human Embryonic Stem Cells (hESCs) | ~2,000 - 5,000 | Developmental transcription factors | Conserved feature of pluripotency. |
| Differentiated Somatic Cells | Drastically reduced (< 500) | Tissue-specific regulators | Resolved to monovalent active or repressed states upon lineage commitment. |
| Cancer Cells (e.g., Leukemia, Glioma) | Often aberrantly high or retained | Oncogenes, Tumor suppressors | Contributes to tumor plasticity, therapy resistance, and blocked differentiation. |
Detailed Protocol: Sequential ChIP-reChIP for Bivalent Marks
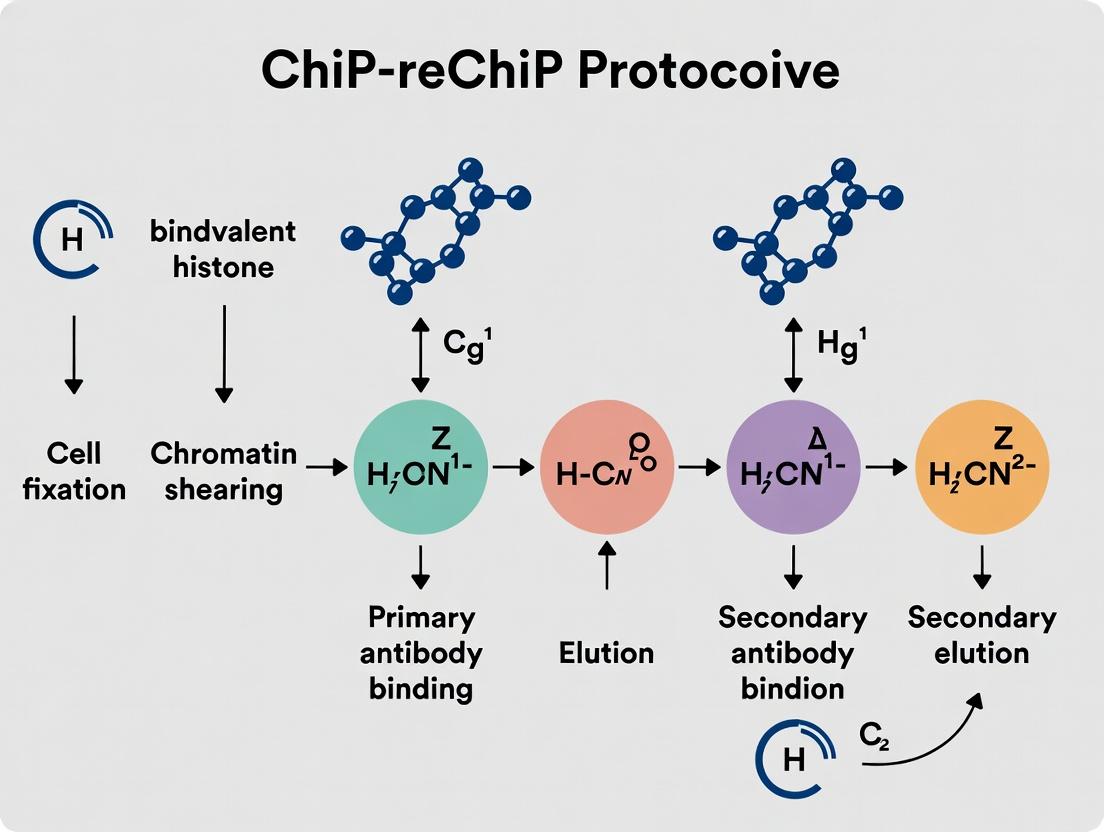
Title: ChIP-reChIP Workflow for Bivalent Nucleosome Validation.
Protocol: Sequential ChIP-reChIP for H3K4me3/H3K27me3
A. Cell Fixation and Lysis
- Crosslinking: Treat ~5x10^6 cells with 1% formaldehyde for 10 min at RT. Quench with 125mM glycine.
- Harvest & Wash: Pellet cells, wash twice with cold PBS containing protease inhibitors (PI).
- Lysis: Resuspend cell pellet in 1 mL Lysis Buffer 1 (50mM HEPES-KOH pH7.5, 140mM NaCl, 1mM EDTA, 10% Glycerol, 0.5% NP-40, 0.25% Triton X-100, +PI) for 10 min on ice. Pellet.
- Nuclear Wash: Resuspend in 1 mL Lysis Buffer 2 (10mM Tris-HCl pH8.0, 200mM NaCl, 1mM EDTA, 0.5mM EGTA, +PI) for 10 min on ice. Pellet.
- Nuclear Lysis: Resuspend pellet in 300μL Lysis Buffer 3 (10mM Tris-HCl pH8.0, 100mM NaCl, 1mM EDTA, 0.5mM EGTA, 0.1% Na-Deoxycholate, 0.5% N-lauroylsarcosine, +PI). Aliquot and freeze at -80°C or proceed.
B. Chromatin Shearing
- Thaw lysate on ice. Sonicate using a focused ultrasonicator (e.g., Covaris) or tip sonicator to achieve fragments of 200-500 bp. Optimize for your system.
- Clarification: Centrifuge at 20,000 x g for 10 min at 4°C. Transfer supernatant to a new tube. Take a 50μL aliquot as "Input." Measure DNA concentration.
C. First Chromatin Immunoprecipitation (Anti-H3K4me3)
- Pre-clear: Dilute sheared chromatin to 1mL with ChIP Dilution Buffer (0.01% SDS, 1.1% Triton X-100, 1.2mM EDTA, 16.7mM Tris-HCl pH8.1, 167mM NaCl, +PI). Add 20μL of pre-washed Protein A/G magnetic beads. Rotate for 1h at 4°C. Discard beads.
- Immunoprecipitation: Add 5-10μg of high-specificity anti-H3K4me3 antibody (see Toolkit) to pre-cleared chromatin. Rotate overnight at 4°C.
- Bead Capture: Add 50μL pre-washed Protein A/G magnetic beads. Rotate for 2h at 4°C.
- Bead Washes: Sequentially wash beads for 5 min each on a rotator at 4°C with:
- Low Salt Wash Buffer (0.1% SDS, 1% Triton X-100, 2mM EDTA, 20mM Tris-HCl pH8.1, 150mM NaCl)
- High Salt Wash Buffer (0.1% SDS, 1% Triton X-100, 2mM EDTA, 20mM Tris-HCl pH8.1, 500mM NaCl)
- LiCl Wash Buffer (0.25M LiCl, 1% NP-40, 1% Na-Deoxycholate, 1mM EDTA, 10mM Tris-HCl pH8.1)
- Two washes with TE Buffer (10mM Tris-HCl pH8.0, 1mM EDTA).
D. Elution and Preparation for Re-ChIP
- Elution: Elute bound complexes from beads twice with 125μL of Re-ChIP Elution Buffer (1% SDS, 10mM DTT in TE buffer) by incubating at 37°C for 30 min with gentle agitation. Combine eluates (~250μL total).
- Dilution: Dilute the combined eluate 1:40 with Re-ChIP Dilution Buffer (1% Triton X-100, 2mM EDTA, 150mM NaCl, 20mM Tris-HCl pH8.1, +PI). This reduces SDS concentration for the second IP.
E. Second Chromatin Immunoprecipitation (Anti-H3K27me3)
- Immunoprecipitation: Add 5-10μg of anti-H3K27me3 antibody to the diluted eluate. Rotate overnight at 4°C.
- Bead Capture & Washes: Repeat step C.3 and C.4 using the same wash series.
F. Final Elution and DNA Recovery
- Final Elution: Elute from beads twice with 125μL of standard ChIP Elution Buffer (1% SDS, 0.1M NaHCO3). Combine eluates.
- Reverse Crosslinks: Add NaCl to a final concentration of 200mM to all eluates and saved Input samples. Incubate at 65°C overnight.
- DNA Purification: Add RNase A, then Proteinase K. Purify DNA using phenol-chloroform extraction or spin columns. Elute in 20-50μL TE buffer or water.
- Analysis: Analyze by quantitative PCR (qPCR) at known bivalent (e.g., MYF5, PAX6 promoters) and control loci. For genome-wide profiling, prepare libraries for next-generation sequencing (ChIP-reChIP-seq).
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Bivalent Mark ChIP-reChIP
| Reagent / Material | Function / Role | Critical Notes for Bivalency Studies |
|---|---|---|
| High-Specificity Anti-H3K4me3 Antibody | First IP: Captures nucleosomes containing the active mark. | Must be validated for ChIP-seq; avoid cross-reactivity. Rabbit monoclonal antibodies (e.g., C42D8, Diagenode C15410003) are preferred. |
| High-Specificity Anti-H3K27me3 Antibody | Second IP (Re-ChIP): Captures from the H3K4me3-enriched pool to isolate bivalent nucleosomes. | Must be validated for sequential IP. Mouse monoclonal (e.g., Millipore 07-449) or rabbit monoclonal (e.g., Cell Signaling 9733S) are common. |
| Protein A/G Magnetic Beads | Solid-phase support for antibody-antigen complex capture. | Ensure efficient capture and low non-specific binding. Magnetic beads simplify wash steps. |
| Focused Ultrasonicator (e.g., Covaris) | Shears crosslinked chromatin to ideal fragment size. | Reproducible shearing is critical for IP efficiency and resolution. |
| DTT-containing Re-ChIP Elution Buffer | Elutes first IP complexes under mild, non-denaturing conditions. | DTT breaks the antibody-antigen bond without disrupting nucleosome integrity, preserving H3K27me3 for the second IP. |
| Validated Positive Control Primer Sets | qPCR validation at known bivalent and monovalent loci. | Essential for protocol optimization. Examples: MYF5 promoter (bivalent in ESCs), active gene (H3K4me3-only), Hox genes. |
| Next-Generation Sequencing Library Prep Kit | For genome-wide ChIP-reChIP-seq analysis. | Requires low-input/ChIP-seq compatible kits due to low yield from sequential IP. |
| Cell Line with Known Bivalency | Positive control biological system (e.g., H1 or RWA hESCs, mouse ESCs). | Necessary for establishing and troubleshooting the protocol. |
Pathway and State Transition Diagram
Title: Regulatory Logic of Bivalent Promoter State Transitions.
The Biological Significance of Bivalent Domains in Development and Disease
Bivalent chromatin domains, first characterized in embryonic stem cells (ESCs), are genomic regions marked by the simultaneous presence of both activating (H3K4me3) and repressive (H3K27me3) histone modifications. These domains are hypothesized to poise key developmental regulator genes for rapid activation or stable silencing upon lineage commitment. This article details their biological roles and provides application notes and protocols within the context of a broader thesis on ChIP-reChIP methodologies for bivalent mark research.
Table 1: Prevalence and Characteristics of Bivalent Domains Across Cell Types
| Cell Type/Tissue | Approximate Number of Bivalent Domains | Key Target Gene Classes | Functional Outcome |
|---|---|---|---|
| Mouse Embryonic Stem Cells (mESCs) | 2,200-3,500 | Homeobox (Hox), Transcription factors (Sox, Pax), Signaling regulators | Lineage priming, pluripotency maintenance |
| Human Embryonic Stem Cells (hESCs) | 2,000-3,000 | Developmental transcription factors, Cell fate determinants | Multilineage differentiation potential |
| Differentiated Somatic Cells | < 500 (highly reduced) | Tissue-specific regulators | Stable silencing or activation |
| Cancer Cells (e.g., AML, Glioblastoma) | 300-1,500 (aberrant) | Tumor suppressors, Developmental oncogenes | Epigenetic block to differentiation, pathogenesis |
Table 2: Key Experimental Findings on Bivalent Domain Dynamics
| Study Focus | Key Quantitative Finding | Method Used | Biological Implication |
|---|---|---|---|
| Resolution upon Differentiation | ~80% of bivalent domains resolve to monovalent (H3K4me3-only or H3K27me3-only) states. | ChIP-seq | Commitment to specific lineage. |
| Disease Association | In AML, >200 gained bivalent domains are associated with Polycomb Repressive Complex 2 (PRC2) binding. | ChIP-reChIP, CUT&Tag | Oncogenic gene silencing. |
| Enzymatic Regulation | EZH2 (PRC2) inhibition reduces H3K27me3 at bivalent loci by 60-70%, leading to aberrant gene activation. | Pharmacological inhibition + ChIP-qPCR | Validates PRC2 as therapeutic target. |
Application Notes & Protocols: ChIP-reChIP for Bivalent Histone Marks
Protocol 1: Sequential Chromatin Immunoprecipitation (ChIP-reChIP)
Objective: To sequentially immunoprecipitate chromatin to isolate nucleosomes carrying both H3K4me3 and H3K27me3 marks.
Key Research Reagent Solutions:
| Reagent/Material | Function/Justification |
|---|---|
| Crosslinking Solution: 1% Formaldehyde in PBS. | Fixes protein-DNA and protein-protein interactions. |
| Cell Lysis Buffer: 50 mM HEPES-KOH pH 7.5, 140 mM NaCl, 1 mM EDTA, 10% Glycerol, 0.5% NP-40, 0.25% Triton X-100. | Lyse cell membrane, extract nuclei. |
| MNase (Micrococcal Nuclease) | Digests chromatin to mononucleosomes for high-resolution IP. |
| ChIP-Grade Antibodies: Anti-H3K4me3 (rabbit monoclonal), Anti-H3K27me3 (mouse monoclonal). | High-specificity antibodies for sequential IP. |
| Magnetic Protein A/G Beads | Solid-phase support for antibody-chromatin complex capture. |
| Elution Buffer (reChIP-specific): 10 mM DTT in 1x TE buffer. | Gently breaks antibody-antigen bond to elute first IP complex without denaturing histones. |
| DNA Clean-up Columns (Silica-based) | Purify final eluted DNA for downstream qPCR or sequencing. |
Detailed Workflow:
Cell Fixation & Chromatin Preparation:
- Crosslink 1x10^7 cells with 1% formaldehyde for 10 min at RT. Quench with 125 mM glycine.
- Pellet cells, wash with cold PBS. Resuspend in Cell Lysis Buffer for 10 min on ice. Centrifuge.
- Resuspend nuclei in MNase Digestion Buffer. Add MNase enzyme (titrated for ~80% mononucleosomes). Incubate 37°C, 20 min. Stop with EDTA.
- Clarify by centrifugation. Aliquot supernatant containing soluble chromatin.
First Immunoprecipitation (IP):
- Pre-clear chromatin with Protein A/G beads for 1 hour at 4°C.
- Incubate pre-cleared chromatin with 2-5 µg of first antibody (e.g., Anti-H3K4me3) overnight at 4°C with rotation.
- Add magnetic Protein A/G beads for 2 hours to capture immune complexes.
- Wash beads sequentially with: Low Salt Wash Buffer, High Salt Wash Buffer, LiCl Wash Buffer, and TE Buffer.
Elution for Re-Chromatin Immunoprecipitation:
- Do not use standard SDS elution buffer. Instead, elute the bound complexes from the first IP by incubating beads in 250 µL of reChIP Elution Buffer (10 mM DTT in TE) for 30 min at 37°C with gentle agitation.
- Collect supernatant (eluted H3K4me3-bound chromatin).
Second Immunoprecipitation (Re-IP):
- Dilute the eluate 1:20 with Dilution Buffer (1% Triton X-100, 2 mM EDTA, 150 mM NaCl, 20 mM Tris-HCl, pH 8.1).
- Add second antibody (e.g., Anti-H3K27me3) and incubate overnight at 4°C.
- Capture with fresh Protein A/G beads, wash as in step 2.
Final Elution & Analysis:
- Elute the final complexes in standard SDS-containing ChIP Elution Buffer.
- Reverse crosslinks at 65°C overnight.
- Treat with Proteinase K and RNase A. Purify DNA using silica columns.
- Analyze by qPCR at known bivalent loci (e.g., promoters of HOX genes) or prepare libraries for next-generation sequencing (ChIP-reChIP-seq).
Critical Notes:
- Antibody Compatibility: The first and second antibodies must be from different host species to prevent re-capture by the same beads.
- Stringency: The DTT elution is mild; optimize concentration to efficiently elute the first complex while preserving histone epitopes.
- Controls: Essential controls include: Input DNA, single H3K4me3 ChIP, single H3K27me3 ChIP, and IgG control for each IP step.
Protocol 2: Validation by Sequential CUT&Tag (sCUT&Tag)
Objective: A tagmentation-based, lower-input alternative to profile bivalent domains.
Brief Methodology:
- Permeabilize nuclei from ~50,000 cells and bind with Concanavalin A-coated magnetic beads.
- Incubate with primary antibody against first mark (e.g., H3K4me3), then a Guinea Pig anti-Rabbit IgG secondary.
- Add pA-Tn5 adapter complex pre-loaded with sequencing adapters. Activate tagmentation to cut DNA near the first mark.
- Gently wash, then repeat steps 2-3 with antibody for the second mark (e.g., H3K27me3) and a pA-Tn5 complex loaded with a different pair of adapters.
- Extract DNA, amplify with dual-indexed primers for both adapter sets, and sequence. Reads containing both adapter sets mark bivalent nucleosomes.
Visualizations
Bivalent Domain Fate in Differentiation
ChIP-reChIP Protocol Workflow
Enzymatic Regulation of a Bivalent Domain
Within the study of bivalent chromatin domains—genomic regions concurrently harboring active (H3K4me3) and repressive (H3K27me3) histone marks—standard chromatin immunoprecipitation (ChIP) reveals critical limitations. This application note, framed within a thesis on ChIP-reChIP for bivalent mark research, details why single-step ChIP is insufficient for resolving co-localized epigenetic marks and provides a detailed protocol for sequential immunoprecipitation (ChIP-reChIP).
Limitations of Standard ChIP in Bivalent Domain Analysis
Standard ChIP isolates DNA bound by a single protein or histone modification. For bivalent domains, this yields only a partial, non-definitive picture. Co-occupancy at the same allele cannot be distinguished from marks existing on different alleles in a cell population.
Table 1: Quantitative Shortcomings of Standard ChIP vs. ChIP-reChIP
| Aspect | Standard ChIP (H3K4me3 or H3K27me3) | Sequential ChIP (ChIP-reChIP) |
|---|---|---|
| Co-localization Resolution | Indirect inference from overlapping peaks; cannot confirm same nucleosome or allele. | Direct confirmation of both marks on the same chromatin fragment. |
| False Positive Rate for Bivalency | High. Overlap in population data may stem from distinct cellular subpopulations. | Very Low. Stringent sequential purification confirms true molecular co-existence. |
| Sensitivity for Rare Bivalent Loci | Low. Signal from dominant mark may mask co-occupancy. | High. Specifically enriches for doubly-modified fragments. |
| Quantitative Data Output | % Input for single mark. Correlation requires separate experiments. | % Input from sequential IP; true bivalent enrichment factor. |
Title: Standard ChIP Workflow for Bivalent Marks
ChIP-reChIP Protocol for Bivalent Histone Marks
This protocol confirms the presence of H3K4me3 and H3K27me3 on the same chromatin fiber.
Part I: Initial Chromatin Preparation and First IP
Day 1: Crosslinking & Sonication
- Crosslink 10^7 cells with 1% formaldehyde for 10 min at room temperature. Quench with 125 mM glycine.
- Lyse cells sequentially with: LB1 (50 mM HEPES-KOH pH 7.5, 140 mM NaCl, 1 mM EDTA, 10% Glycerol, 0.5% NP-40, 0.25% Triton X-100) for 10 min; LB2 (10 mM Tris-HCl pH 8.0, 200 mM NaCl, 1 mM EDTA, 0.5 mM EGTA) for 10 min; LB3 (10 mM Tris-HCl pH 8.0, 100 mM NaCl, 1 mM EDTA, 0.5 mM EGTA, 0.1% Na-Deoxycholate, 0.5% N-lauroylsarcosine).
- Resuspend pellet in 1 mL LB3 and sonicate (e.g., Bioruptor) to shear chromatin to 200-500 bp fragments. Clear debris by centrifugation.
Day 2: First Immunoprecipitation (H3K4me3)
- Pre-clear chromatin with 50 µL Protein A/G beads for 1 hour at 4°C.
- Incubate supernatant with 5-10 µg of anti-H3K4me3 antibody overnight at 4°C with rotation.
- Add 50 µL pre-blocked Protein A/G beads for 2 hours.
- Wash beads sequentially for 5 min each: Low Salt Wash Buffer (0.1% SDS, 1% Triton X-100, 2 mM EDTA, 20 mM Tris-HCl pH 8.0, 150 mM NaCl), High Salt Wash Buffer (same with 500 mM NaCl), LiCl Wash Buffer (0.25 M LiCl, 1% NP-40, 1% Na-Deoxycholate, 1 mM EDTA, 10 mM Tris-HCl pH 8.0), and twice with TE Buffer.
Part II: Elution and Second Immunoprecipitation (ReChIP)
- Elute First IP Complexes: Add 100 µL of 10 mM DTT in ChIP Elution Buffer (1% SDS, 10 mM EDTA, 50 mM Tris-HCl pH 8.0). Incubate 30 min at 37°C with shaking (1200 rpm). Centrifuge and collect supernatant.
- Dilution & Second IP: Dilute eluate 1:40 with ReChIP Dilution Buffer (0.01% SDS, 1.1% Triton X-100, 1.2 mM EDTA, 16.7 mM Tris-HCl pH 8.0, 167 mM NaCl). Split into two fractions: one for the second IP, one as "First IP Eluate" control.
- Immunoprecipitate for H3K27me3: Add anti-H3K27me3 antibody (5 µg) to the main fraction. Incubate overnight at 4°C.
- Capture & Wash: Add 25 µL pre-blocked beads for 2 hours. Perform washes as in Step 7.
Part III: Reverse Crosslinks & DNA Purification
- Elute all samples (ReChIP sample, First IP Eluate control, and a 1% Input sample): Add 150 µL ChIP Elution Buffer and incubate at 65°C overnight with shaking to reverse crosslinks.
- Treat with RNase A (30 min at 37°C) and Proteinase K (2 hours at 55°C).
- Purify DNA using a PCR purification kit. Elute in 30 µL EB Buffer.
- Analyze by qPCR for known bivalent promoters (e.g., POUSF1, PAX6) and control regions.
Title: ChIP-reChIP Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for ChIP-reChIP on Bivalent Marks
| Item | Function & Rationale |
|---|---|
| High-Quality, ChIP-Grade Antibodies | Specificity is critical. Anti-H3K4me3 (e.g., Millipore 07-473) and anti-H3K27me3 (e.g., Cell Signaling 9733) are well-validated. Low cross-reactivity ensures clean reChIP. |
| Protein A/G Magnetic Beads | Offer lower background and easier handling/elution than agarose beads, improving sequential IP efficiency. |
| DTT (Dithiothreitol) | Used in the primary elution step to break the antibody-antigen bond without fully denaturing chromatin, preserving epitopes for the second IP. |
| Dual-Chromatin Sonication Device | Consistent, high-quality shearing to 200-500 bp is essential to ensure a single nucleosome remains intact for dual-mark analysis. |
| ChIP-Quality Protease Inhibitors | Preserve histone modifications and protein complexes during lengthy lysis and IP steps. |
| Validated qPCR Primers | For known bivalent and monovalent control loci. Necessary for validating protocol success before costly sequencing. |
| PCR Purification Kit (Silica Membrane) | For efficient recovery of low-concentration DNA after reverse crosslinking. |
Data Interpretation and Application in Drug Discovery
ChIP-reChIP data provides a definitive map of bivalent domains, which are pivotal in maintaining pluripotency and are frequently altered in disease.
Table 3: Example qPCR Analysis of a Bivalent Locus (POUSF1 Promoter)
| Sample | % Input (Mean ± SD) | Enrichment Relative to IgG |
|---|---|---|
| Input (1%) | 1.00 | N/A |
| Standard ChIP: H3K4me3 | 0.85 ± 0.10 | 12.1x |
| Standard ChIP: H3K27me3 | 0.72 ± 0.08 | 9.8x |
| ChIP-reChIP: Sequential IP | 0.15 ± 0.03 | 21.4x |
The sequential IP yields a lower % input but a higher specific enrichment, confirming true co-occupancy. For drug developers, this method can precisely monitor the resolution of bivalency (e.g., loss of H3K27me3 upon differentiation therapy) in response to epigenetic inhibitors, providing a robust pharmacodynamic biomarker.
Chromatin Immunoprecipitation (ChIP) is a cornerstone technique for studying protein-DNA interactions. Sequential Chromatin Immunoprecipitation, or ChIP-reChIP, extends this power by enabling the isolation of chromatin fragments bound by two specific proteins or carrying two specific post-translational modifications simultaneously. This is particularly crucial for studying complex epigenetic states, such as bivalent domains, where activating (e.g., H3K4me3) and repressive (e.g., H3K27me3) histone marks co-exist on the same nucleosome, poising key developmental genes for expression. This application note details the core principles, a robust protocol, and essential tools for successful ChIP-reChIP experiments within the context of bivalent mark research.
Key Principles & Quantitative Considerations
The success of ChIP-reChIP hinges on several critical parameters. The sequential immunoprecipitation steps cumulatively reduce yield, making high-quality starting material and efficient antibodies essential.
Table 1: Critical Quantitative Parameters for ChIP-reChIP Success
| Parameter | Recommended Specification | Impact on Experiment |
|---|---|---|
| Starting Crosslinked Material | 5-10 x 10^6 cells per IP | Ensures sufficient final DNA yield for analysis. |
| Chromatin Shearing Size | 200-500 bp (optimized) | Fragments must contain intact nucleosomes (<~1kb) but be small enough for specificity. |
| First ChIP Elution Efficiency | >70% (ideal) | Minimizes loss of target material before second IP. |
| Antibody Specificity & Affinity | Validated for sequential IP (ChIP-grade) | Non-specific binding or low affinity drastically reduces final specificity. |
| Final DNA Yield | 1-50 ng (highly target-dependent) | Requires sensitive detection (qPCR, library prep for seq). |
Table 2: Expected Outcome Metrics for Bivalent Domain Analysis
| Analysis Method | Input Requirement (Final DNA) | Typical Enrichment Fold (over IgG) | Key Interpretation Metric |
|---|---|---|---|
| qPCR at Known Loci | 0.5-2 ng | H3K4me3: 10-50x; H3K27me3: 5-30x; Re-ChIP: 5-20x* | % Input for Re-ChIP vs. single ChIPs. |
| Sequencing (ChIP-reChIP-seq) | 1-10 ng (post-amplification) | Library complexity & unique reads at bivalent promoters. | Peak overlap and statistical co-localization. |
*Re-ChIP enrichment is typically lower than single ChIP due to sequential efficiency losses.
Detailed ChIP-reChIP Protocol for Bivalent Histone Marks
Reagents & Solutions (The Scientist's Toolkit)
Table 3: Essential Research Reagent Solutions
| Item | Function / Specification |
|---|---|
| Crosslinking Reagent | 1% Formaldehyde in PBS. Fixes protein-DNA and protein-protein interactions. |
| Glycine (2.5 M) | Quenches formaldehyde to stop crosslinking. |
| Cell Lysis Buffer | (10 mM Tris-HCl pH 8.0, 10 mM NaCl, 0.2% NP-40) + protease inhibitors. Lyses cell membrane. |
| Nuclear Lysis/Sonication Buffer | (50 mM Tris-HCl pH 8.0, 10 mM EDTA, 1% SDS) + protease inhibitors. Lyses nucleus for chromatin shearing. |
| Dilution Buffer | (16.7 mM Tris-HCl pH 8.0, 167 mM NaCl, 1.2 mM EDTA, 1.1% Triton X-100, 0.01% SDS) + protease inhibitors. Reduces SDS concentration for IP. |
| Magnetic Protein A/G Beads | For antibody immobilization and capture of immune complexes. |
| Low Salt Wash Buffer | (20 mM Tris-HCl pH 8.0, 150 mM NaCl, 2 mM EDTA, 1% Triton X-100, 0.1% SDS) |
| High Salt Wash Buffer | (20 mM Tris-HCl pH 8.0, 500 mM NaCl, 2 mM EDTA, 1% Triton X-100, 0.1% SDS) |
| LiCl Wash Buffer | (10 mM Tris-HCl pH 8.0, 250 mM LiCl, 1 mM EDTA, 1% NP-40, 1% Na-Deoxycholate) |
| TE Buffer | (10 mM Tris-HCl pH 8.0, 1 mM EDTA) For final washes and elution. |
| Elution Buffer (First ChIP) | (10 mM Tris-HCl pH 8.0, 1 mM EDTA, 1% SDS, 10 mM DTT). DTT reduces antibodies for gentle elution. |
| Primary Antibodies | Key: Validated ChIP-grade anti-H3K4me3 (rabbit) and anti-H3K27me3 (mouse) for sequential IP. Species must differ. |
Step-by-Step Workflow
Day 1: Crosslinking, Lysis, and Sonication
- Crosslink 5-10 million cells with 1% formaldehyde for 10 min at RT. Quench with 125 mM glycine.
- Pellet cells, wash with cold PBS. Resuspend in 1 mL Cell Lysis Buffer, incubate 15 min on ice. Centrifuge to pellet nuclei.
- Resuspend nuclear pellet in 1 mL Sonication Buffer. Sonicate to shear chromatin to 200-500 bp fragments (optimize for your sonicator). Centrifuge to clear debris.
- Dilute supernatant 10-fold with Dilution Buffer. Aliquot as "Input" (1%) and save at -80°C. The remainder is the "Chromatin Extract."
Day 2: First Chromatin Immunoprecipitation
- Pre-clear Chromatin Extract with 20 μL magnetic Protein A/G beads for 1 hour at 4°C.
- Incubate pre-cleared chromatin with 2-5 μg of the first antibody (e.g., anti-H3K4me3) overnight at 4°C with rotation.
- Add 50 μL magnetic beads and incubate for 2 hours.
- Wash beads sequentially with: Low Salt Buffer (2x), High Salt Buffer (1x), LiCl Buffer (1x), TE Buffer (2x).
Day 2: Elution and Second Chromatin Immunoprecipitation
- First Elution: Elute bound complexes from beads in 100 μL Elution Buffer (First ChIP) by incubating at 37°C for 30 min with shaking. Collect supernatant.
- Neutralization & Dilution: Dilute the eluate 20-fold with Dilution Buffer to reduce SDS concentration. This is the "Eluate for Second IP."
- Second Immunoprecipitation: Add the second antibody (e.g., anti-H3K27me3) to the diluted eluate. Repeat the overnight incubation and bead capture steps (6-8).
- Final Washes: Wash the beads from the second IP as in step 8.
Day 3: Reverse Crosslinks & DNA Purification
- Final Elution: Elute all samples (Re-ChIP, single ChIPs, Saved Input) in 120 μL of standard ChIP Elution Buffer (TE + 1% SDS) at 65°C for 15 min.
- Reverse Crosslinks: Add 5 μL of 5M NaCl to all samples (including Input). Incubate at 65°C overnight.
- DNA Recovery: Treat with RNase A, then Proteinase K. Purify DNA using phenol-chloroform extraction or spin columns. Elute in 30-50 μL TE or water.
- Analysis: Analyze DNA by qPCR at known bivalent (e.g., HOX genes) and control loci, or prepare libraries for sequencing.
Visualizations
ChIP-reChIP Experimental Workflow
Core Principle: Sequential Isolation of Bivalent Nucleosomes
Application Notes on ChIP-reChIP for Bivalent Histone Marks
Bivalent chromatin domains, defined by the simultaneous presence of opposing histone modifications such as H3K4me3 (activating) and H3K27me3 (repressive), are fundamental to pluripotency and cell fate decisions. Their dysregulation is a hallmark of cancer, contributing to aberrant differentiation and oncogenic gene expression programs. The sequential Chromatin Immunoprecipitation (ChIP-reChIP) protocol is the definitive technique for directly demonstrating the co-existence of these marks on the same nucleosome within a single cell population, providing critical insights for therapeutic discovery.
Quantitative Data Summary: Bivalent Domains in Development and Disease
Table 1: Prevalence and Dynamics of Bivalent Domains
| Biological Context | Key H3K4me3/H3K27me3 Target Genes | Approximate Number of Bivalent Domains | Functional Consequence |
|---|---|---|---|
| Human Embryonic Stem Cells (hESCs) | Developmental regulators (e.g., HOX, PAX, TBX families) | ~2,200-3,500 promoters | Poised for rapid activation or silencing upon differentiation. |
| Differentiated Somatic Cells | Subset of hESC bivalent domains | ~100-500 promoters | Resolution to monovalent states; stable silencing or activation. |
| Hematological Malignancies (e.g., AML) | Tumor suppressor genes, lineage regulators | Increased at specific loci (e.g., CDKN2A, GATA family) | Aberrant silencing maintenance blocks differentiation. |
| Solid Tumors (e.g., Glioblastoma) | Developmental signaling pathways (WNT, SHH) | Context-dependent gains/losses | Promotes stem-like properties and therapy resistance. |
| Drug Treatment (EZH2 Inhibitors) | Previously bivalent, PRC2-silenced genes | N/A (Functional Reversal) | H3K27me3 loss leads to H3K4me3-driven gene reactivation. |
Detailed ChIP-reChIP Protocol for Bivalent Mark Analysis
Protocol: Sequential Immunoprecipitation of H3K4me3 and H3K27me3
Day 1: Crosslinking and Chromatin Preparation
- Crosslink cells (1x10^7) in 1% formaldehyde for 10 min at room temperature. Quench with 125mM glycine.
- Lyse cells in SDS Lysis Buffer. Pellet nuclei and resuspend in ChIP IP Buffer.
- Shear chromatin to an average size of 200-500 bp using a focused ultrasonicator (e.g., 8 cycles of 30 sec ON, 30 sec OFF, 4°C). Verify fragment size by agarose gel electrophoresis.
- Centrifuge sheared lysate at 14,000 rpm for 10 min at 4°C. Collect supernatant (chromatin input). Aliquot and store at -80°C.
Day 2: First Immunoprecipitation (H3K27me3)
- Pre-clear chromatin (50-100 µg) with Protein A/G Magnetic Beads for 1 hour at 4°C.
- Incubate pre-cleared chromatin with 5-10 µg of high-specificity anti-H3K27me3 antibody (e.g., Clone C36B11) overnight at 4°C with rotation.
- Add magnetic beads and incubate for 2 hours. Wash beads sequentially with: Low Salt Wash Buffer (once), High Salt Wash Buffer (once), LiCl Wash Buffer (once), and TE Buffer (twice).
Day 3: Elution and Second Immunoprecipitation (H3K4me3)
- Elution from first ChIP: To the washed bead complex, add 100 µL of ChIP Elution Buffer (1% SDS, 0.1M NaHCO3) and 1 µL of Proteinase K (20 mg/mL). Incubate at 62°C for 2 hours, then 95°C for 10 min to reverse crosslinks. Cool and centrifuge. Transfer supernatant (eluted H3K27me3-bound chromatin).
- Dilution: Dilute the eluate 1:50 with ChIP IP Buffer (to reduce SDS concentration).
- Second IP: Use the diluted eluate as input for a standard ChIP protocol with an anti-H3K4me3 antibody (e.g., Clone D1A8). Repeat steps 6-7.
- Final Elution: Elute the ChIP-reChIP material as in step 8. Purify DNA using a PCR purification kit. Analyze by qPCR with primers for known bivalent promoters (e.g., HOXA9, MEIS1) and negative control regions.
Visualization of Pathways and Workflows
Title: ChIP-reChIP Experimental Workflow
Title: Bivalent Domain Fate in Stem Cells and Cancer
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Bivalent Chromatin Research
| Reagent/Material | Function & Rationale |
|---|---|
| High-Specificity ChIP-Grade Antibodies (e.g., anti-H3K4me3, anti-H3K27me3) | Critical for low-background, high-efficiency IP. Validated for sequential IP. Non-specific antibodies cause high noise. |
| Magnetic Beads (Protein A/G) | Enable efficient wash steps and gentle elution required for the two-step ChIP-reChIP protocol. |
| Focused Ultrasonicator | Provides consistent, high-powered shearing to achieve optimal chromatin fragment size (200-500 bp). |
| ChIP-Validated qPCR Primers | For known bivalent and control loci. Essential for validating the success of the ChIP-reChIP experiment before sequencing. |
| EZH2/PRC2 Inhibitors (e.g., GSK126, Tazemetostat) | Tool compounds to dissect bivalent domain function. Used to probe dependence on H3K27me3 for gene silencing in cancer models. |
| Next-Generation Sequencing Library Prep Kit | For genome-wide analysis of ChIP-reChIP output (e.g., ChIP-seq, ChIPmentation). Must be compatible with low DNA input. |
A Detailed Protocol: From Cell Fixation to Final qPCR/NGS Analysis
Within the context of a thesis on Chromatin Immunoprecipitation-Re-Chromatin Immunoprecipitation (ChIP-reChIP) for investigating bivalent histone marks (e.g., co-existing H3K4me3 and H3K27me3), rigorous pre-protocol planning is paramount. Success hinges on validated reagents and robust controls to ensure data specificity and interpretability. This document outlines essential application notes and protocols for this critical planning phase.
Application Notes: The Validation Imperative
For ChIP-reChIP, antibody validation is exponentially more critical than for standard ChIP. The protocol sequentially targets two epitopes, compounding any non-specificity. A recent survey (2023) indicated that over 30% of commercial histone modification antibodies fail specificity tests in common validation paradigms. Key validation strategies must be employed:
- Target Specificity: Use knockout/knockdown cell lines (e.g., EZH2 KO for H3K27me3) or peptide competition assays.
- Application Specificity: Validate specifically for ChIP, not just western blot. A 2024 review highlighted that only ~15% of published ChIP-seq studies used genetically validated antibodies.
- Lot-to-Lot Consistency: Document antibody lot numbers, as performance can vary.
Table 1: Quantitative Summary of Antibody Validation Challenges in Epigenetics
| Validation Metric | Typical Pass Rate in Commercial Antibodies (2023-2024) | Critical Test for ChIP-reChIP |
|---|---|---|
| Specificity in Western Blot | ~70% | Necessary but not sufficient |
| Specificity in Peptide Array/Competition | ~65% | High-value primary screen |
| Specificity in Genetic Knockout/Knockdown | <50% | Gold standard for chromatin antibodies |
| ChIP-seq Grade Certification | ~40% | Required starting point |
Protocols for Pre-Protocol Validation
Protocol 1: Peptide Competition Assay for Antibody Validation
Purpose: To confirm antibody binding is specific to the intended histone modification. Materials:
- Candidate antibody (anti-H3K4me3, anti-H3K27me3).
- Biotinylated peptide with the target modification.
- Non-modified control peptide.
- Streptavidin-coated plates.
- Standard ELISA detection reagents. Methodology:
- Immobilize the biotinylated target peptide on a streptavidin plate.
- Pre-incubate the antibody with a molar excess (10-100x) of either soluble competing peptide (modified or non-modified) for 1 hour.
- Transfer antibody/peptide mix to the peptide-coated plate and proceed with standard ELISA.
- Interpretation: Signal should be abolished only by the modified competing peptide, not the non-modified control.
Protocol 2: Genetic Validation Using CRISPR-Cas9 Cell Lines
Purpose: To provide definitive evidence of antibody specificity in a cellular context. Materials:
- Wild-type and knockout cell lines (e.g., SETD1A/B KO for H3K4me3 loss; EZH1/2 DKO for H3K27me3 loss).
- Fixation and chromatin shearing reagents.
- Validated control antibody (e.g., H3 total). Methodology:
- Generate or acquire knockout cell lines for the histone methyltransferases responsible for depositing the mark of interest.
- Perform parallel ChIP-qPCR on wild-type and knockout cells using the candidate antibody.
- Use primers for genomic regions known to carry the mark (positive control) and known barren regions (negative control).
- Interpretation: Enrichment should be abolished specifically at positive control regions in the knockout line, while input and total histone controls remain stable.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for ChIP-reChIP Bivalent Mark Studies
| Item | Function in ChIP-reChIP | Critical Consideration |
|---|---|---|
| Crosslinking Agent (e.g., Formaldehyde) | Fixes protein-DNA and protein-protein interactions. | Optimization of concentration/time is key for sequential IP efficiency. |
| Chromatin Shearing System (Ultrasonicator) | Fragments chromatin to 200-500 bp. | Over-shearing can destroy epitopes; under-shearing reduces resolution. |
| Protein A/G Magnetic Beads | Solid-phase matrix for antibody capture. | High binding capacity reduces antibody requirement; low non-specific DNA binding is crucial. |
| Validated Primary Antibodies (Pair) | Sequential immunoprecipitation of two histone marks. | Must be validated for sequential IP. Species/host should differ if elution is not harsh. |
| Mild Elution Buffer (e.g., 10mM DTT) | Elutes first IP complex without denaturing epitopes for the second IP. | Critical for sequential ChIP; harsh elution (SDS) will destroy the 2nd epitope. |
| DNA Clean-up/Purification Kit | Isolates final eluted DNA for qPCR or sequencing. | High sensitivity recovery for low-yield reChIP material is essential. |
| Control qPCR Primers | Assess enrichment at target and control loci. | Must include positive control loci for each mark individually and for bivalent domains, plus negative control loci. |
Experimental Controls Framework
A tiered control system is mandatory:
- Technical Controls: Input DNA, No Antibody (Beads-only), Mock reChIP (IgG in 1st IP).
- Biological Controls: Cell lines lacking the marks (genetic knockouts).
- Target-Specific Controls: qPCR primers for known bivalent domains (e.g., promoters of developmental regulators like HOX genes), active mark-only regions, silent mark-only regions, and barren genomic regions.
Visualization of Workflows and Relationships
Title: Pre-Protocol Planning Workflow for ChIP-reChIP
Title: ChIP-reChIP for Bivalent Histone Marks
This application note details the critical first step for sequential chromatin immunoprecipitation (ChIP-reChIP), a powerful technique for investigating the co-localization of histone modifications, such as bivalent marks (e.g., H3K4me3 and H3K27me3), on the same nucleosome. The robustness of the final reChIP data is entirely dependent on the initial crosslinking efficiency and the quality of the prepared chromatin. This protocol is optimized to preserve fragile protein-DNA-histone interactions while generating chromatin fragments of ideal size and concentration for two sequential immunoprecipitations.
Key Parameters for Optimization
The success of chromatin preparation hinges on several quantitative parameters. The following table summarizes the optimized conditions derived from current methodologies.
Table 1: Optimized Crosslinking and Chromatin Preparation Parameters
| Parameter | Optimized Condition | Rationale & Impact on reChIP |
|---|---|---|
| Crosslinker | 1% Formaldehyde | Standard for DNA-protein fixation; higher concentrations can mask epitopes. |
| Crosslinking Time | 8-10 minutes at room temperature | Balances complex fixation with epitope accessibility for IP. |
| Quenching Agent | 125 mM Glycine | Efficiently stops crosslinking to prevent over-fixation. |
| Cell Lysis Buffer | Containing Protease Inhibitors & 0.1% SDS | Gently lyses cells and nuclei while preserving chromatin integrity. |
| Sonication Goal | 200-500 bp DNA fragments (avg. ~300 bp) | Fragments smaller than a nucleosome array (~1 kb) but large enough to contain multiple nucleosomes for dual-mark analysis. |
| Chromatin Yield | 50-100 µg per 10⁷ cells | Provides sufficient material for two IPs and necessary controls. |
| Fragment Analysis | Agarose Gel Electrophoresis or Bioanalyzer | Critical QC step; ensures proper sonication efficiency. |
Detailed Protocol: Optimized Crosslinking & Chromatin Preparation
A. Cell Fixation and Lysis
- Culture & Harvest: Grow approximately 10⁷ mammalian cells to 70-80% confluence. Harvest by scraping (adherent) or centrifugation (suspension). Wash cells once with 10 mL ice-cold 1X PBS.
- Crosslinking: Resuspend cell pellet in 10 mL of 1X PBS. Add 270 µL of 37% formaldehyde (to achieve 1% final concentration). Incubate for 8-10 minutes at room temperature with gentle agitation.
- Quenching: Add 1 mL of 1.25 M glycine (125 mM final concentration) to quench the crosslinking. Incubate for 5 minutes at room temperature with gentle agitation.
- Washing: Pellet cells at 800 x g for 5 minutes at 4°C. Wash the pellet twice with 10 mL of ice-cold 1X PBS. The pellet can be flash-frozen in liquid nitrogen and stored at -80°C or processed immediately.
- Cell Lysis: Resuspend cell pellet in 1 mL of Cell Lysis Buffer (10 mM Tris-HCl pH 8.0, 10 mM NaCl, 0.2% NP-40/Igepal, plus fresh protease inhibitor cocktail). Incubate on ice for 15 minutes. Pellet nuclei at 2000 x g for 5 minutes at 4°C. Discard supernatant.
- Nuclear Lysis: Resuspend nuclei pellet in 1 mL of Nuclear Lysis Buffer (50 mM Tris-HCl pH 8.0, 10 mM EDTA, 1% SDS, plus fresh protease inhibitors). Incubate on ice for 10 minutes.
B. Chromatin Shearing by Sonication
- Transfer the lysate to a 1.5 mL sonication microtube. Perform sonication using a focused ultrasonicator (e.g., Covaris, Bioruptor). The settings must be empirically determined for each cell type and instrument.
- Example Conditions (Covaris S2): Duty Cycle: 5%, Intensity: 4, Cycles/Burst: 200, Time: 10 minutes (with cooling).
- Goal: Shear chromatin to an average size of 200-500 bp.
- Clarification: Pellet insoluble debris by centrifuging the sonicated lysate at 20,000 x g for 15 minutes at 4°C. Transfer the supernatant (soluble chromatin) to a new tube.
- QC Analysis: Take a 20 µL aliquot of chromatin. Reverse crosslinks by adding 80 µL of TE buffer + 2 µL of 5M NaCl and incubating at 65°C overnight. Purify DNA using a PCR purification kit. Analyze 10 µL on a 1.5% agarose gel or a Bioanalyzer/TapeStation to verify fragment size distribution.
- Quantification: Measure the chromatin concentration (DNA concentration) using a spectrophotometer. Adjust calculations for the presence of SDS. A typical yield is 50-100 µg of chromatin from 10⁷ cells.
- Storage: Aliquot chromatin and store at -80°C. Avoid repeated freeze-thaw cycles.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Crosslinking & Chromatin Prep
| Item | Function & Key Feature |
|---|---|
| UltraPure Formaldehyde (16%, methanol-free) | Provides clean, consistent crosslinking without methanol interference. |
| Protease Inhibitor Cocktail (EDTA-free) | Preserves protein integrity and histone modifications during lysis. EDTA-free is compatible with subsequent Mg²⁺-dependent steps. |
| Focused Ultrasonicator (e.g., Covaris) | Provides reproducible, controlled acoustic shearing for consistent chromatin fragment sizes with low heat generation. |
| Dynabeads Protein A/G | Magnetic beads for efficient IP; crucial for the sequential elution required in reChIP. |
| HDAC Inhibitors (e.g., Sodium Butyrate) | Optional additive to lysis buffers to prevent loss of acetylated marks during processing. |
| High-Sensitivity DNA Assay Kit (Qubit/Bioanalyzer) | Accurately quantifies low-concentration, sheared DNA for chromatin normalization. |
Visualization of Workflow and Critical Relationship
Title: Chromatin Prep Workflow for reChIP
Title: Sonication QC Impact on reChIP Success
Within the context of a ChIP-reChIP protocol for investigating bivalent histone marks (e.g., co-existing H3K4me3 and H3K27me3), the first ChIP step is critical. This step selectively enriches for chromatin fragments harboring one of the two marks, setting the stage for the subsequent reChIP. The setup and elution strategy must balance yield, specificity, and the preservation of chromatin integrity for the second immunoprecipitation. This application note details a robust protocol optimized for bivalent mark studies.
The following table summarizes optimized conditions for the 1st ChIP step, derived from current methodologies.
Table 1: Optimized Parameters for 1st ChIP in reChIP Studies
| Parameter | Recommended Condition | Rationale & Impact |
|---|---|---|
| Cross-linking | 1% Formaldehyde, 10 min, room temp | Standard fixation for histone-DNA interactions. Over-fixation reduces shearing efficiency. |
| Chromatin Shearing | Sonication to 200-500 bp fragments (avg. ~300 bp) | Ideal fragment size for IP resolution and subsequent reChIP efficiency. |
| Antibody Amount | 1-5 µg per 100 µg chromatin (see Table 2) | High-specificity antibody is paramount to minimize background carryover. |
| Immunoprecipitation | Overnight incubation at 4°C with rotation | Ensures adequate antigen-antibody complex formation. |
| Beads Type | Protein A/G Magnetic Beads | Allow for efficient washing and gentle elution. |
| Wash Stringency | 2x Low Salt, 1x High Salt, 1x LiCl, 2x TE Buffer | Removes non-specifically bound chromatin while preserving complexes. |
| Elution Method | Non-Denaturing Elution (Recommended) | Preserves antibody epitope for 2nd ChIP. Critical for sequential IP. |
| Elution Buffer | 30 µL 0.5% SDS, 1x TE, 10 mM DTT, 37°C, 30 min | Gently dissociates chromatin from beads/antibody without denaturing histones. |
Table 2: Example Antibody Performance Data for 1st ChIP
| Target Mark | Antibody Clone / Catalog # | Recommended Use (per 100 µg chromatin) | Typical Enrichment (qPCR vs Input) |
|---|---|---|---|
| H3K4me3 | Rabbit Monoclonal, C42D8 | 2 µg | 50-200 fold at active gene promoters |
| H3K27me3 | Rabbit Monoclonal, C36B11 | 3-5 µg | 20-100 fold at repressed loci |
| Control IgG | Species-matched | Equivalent to test Ab | < 2 fold (background level) |
Detailed 1st ChIP Protocol for reChIP
A. Chromatin Preparation (Pre-1st ChIP)
- Cross-link cells with 1% formaldehyde for 10 minutes. Quench with 125 mM glycine.
- Lyse cells sequentially with Cell Lysis Buffer (10 mM Tris-HCl pH 8.0, 10 mM NaCl, 0.2% NP-40) and Nuclear Lysis Buffer (50 mM Tris-HCl pH 8.0, 10 mM EDTA, 1% SDS).
- Shear chromatin via sonication to an average fragment size of 200-500 bp. Centrifuge to remove debris.
- Dilute sheared chromatin 10-fold in ChIP Dilution Buffer (0.01% SDS, 1.1% Triton X-100, 1.2 mM EDTA, 16.7 mM Tris-HCl pH 8.0, 167 mM NaCl). Retain 1% as "Input" control.
B. First Immunoprecipitation
- Pre-clear chromatin with 20 µL protein A/G magnetic beads for 1 hour at 4°C.
- Incubate supernatant with target-specific antibody (Table 2) overnight at 4°C with rotation.
- Capture immune complexes by adding 30 µL pre-blocked protein A/G beads for 2 hours at 4°C.
- Wash beads sequentially on a magnetic rack:
- 2x with 1 mL Low Salt Wash Buffer (0.1% SDS, 1% Triton X-100, 2 mM EDTA, 20 mM Tris-HCl pH 8.0, 150 mM NaCl)
- 1x with 1 mL High Salt Wash Buffer (0.1% SDS, 1% Triton X-100, 2 mM EDTA, 20 mM Tris-HCl pH 8.0, 500 mM NaCl)
- 1x with 1 mL LiCl Wash Buffer (0.25 M LiCl, 1% NP-40, 1% sodium deoxycholate, 1 mM EDTA, 10 mM Tris-HCl pH 8.0)
- 2x with 1 mL TE Buffer (10 mM Tris-HCl pH 8.0, 1 mM EDTA)
C. Critical Elution Strategy for reChIP
- After the final TE wash, resuspend beads in 100 µL of 1x TE Buffer.
- Transfer the bead suspension to a fresh tube.
- Add 10 µL of 10% SDS and 2 µL of 0.5M DTT (Final: 0.5% SDS, 10 mM DTT in ~112 µL).
- Incubate at 37°C for 30 minutes with gentle agitation. Do NOT use high-temperature or harsh denaturing elution.
- Place tube on magnet and carefully transfer the eluate (containing immunoprecipitated chromatin complexes) to a fresh tube. This is the material for the 2nd ChIP.
- Dilute the eluate with 10 volumes of Dilution Buffer (as in Step A4) to reduce SDS concentration for the next IP step.
Visualization of Workflow
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for 1st ChIP in reChIP Protocols
| Item | Function & Importance |
|---|---|
| High-Specificity Monoclonal Antibodies (e.g., Cell Signaling Tech C42D8) | Minimizes non-specific binding, crucial for reducing background in the final reChIP result. |
| Protein A/G Magnetic Beads | Enable efficient, low-background washing and gentle, non-mechanical elution of complexes. |
| Protease/Phosphatase Inhibitor Cocktails | Preserve histone modifications and protein integrity during chromatin preparation. |
| Diagenode Bioruptor Pico or similar sonicator | Provides consistent, high-quality shearing to ideal fragment sizes for ChIP-reChIP. |
| Non-Denaturing Elution Buffer (SDS/DTT-based) | The cornerstone of reChIP; gently releases chromatin while preserving histone marks and antibody epitopes for the 2nd IP. |
| Glycine (Quenching Solution) | Prevents over-crosslinking, which is detrimental to shearing and antigen accessibility. |
| RNAse A & Proteinase K | Essential for reversing cross-links and digesting proteins after elution, prior to DNA purification. |
Re-ChIP (Sequential Chromatin Immunoprecipitation) is a critical technique for investigating the simultaneous presence of two distinct epigenetic marks or transcription factors on the same chromatin fragment. Within the context of bivalent histone mark research—specifically the co-localization of active (H3K4me3) and repressive (H3K27me3) marks on promoter regions of developmental genes—Re-ChIP provides definitive proof of their bivalency on individual nucleosomes. This protocol details the sequential immunoprecipitation step following the initial ChIP, enabling the identification of true chromatin complexes harboring multiple, specific modifications.
Key Experimental Protocol: Re-ChIP Procedure
Important Prerequisite: This protocol begins with chromatin that has already been immunoprecipitated and eluted in the first ChIP step (e.g., using an anti-H3K4me3 antibody). The eluate contains the chromatin complexes bound to the primary antibody-protein A/G beads.
Materials & Buffers:
- Re-ChIP Elution Buffer: 10 mM DTT in 1X TE buffer (pH 8.0).
- Re-ChIP Dilution Buffer: 1% Triton X-100, 2 mM EDTA, 150 mM NaCl, 20 mM Tris-HCl (pH 8.1).
- High Salt Wash Buffer: 0.1% SDS, 1% Triton X-100, 2 mM EDTA, 500 mM NaCl, 20 mM Tris-HCl (pH 8.1).
- LiCl Wash Buffer: 0.25 M LiCl, 1% NP-40, 1% deoxycholate, 1 mM EDTA, 10 mM Tris-HCl (pH 8.1).
- Final Elution Buffer: 1% SDS, 0.1 M NaHCO₃.
- Proteinase K
- Antibody for second IP (e.g., anti-H3K27me3).
Detailed Protocol:
- Elution from 1st ChIP Beads: To the beads from the first ChIP, add 25-50 µL of Re-ChIP Elution Buffer. Incubate at 37°C for 30 minutes with gentle agitation.
- Centrifugation: Pellet the beads at 2,000 x g for 1 minute. Carefully transfer the supernatant (containing the eluted chromatin complexes) to a new microcentrifuge tube.
- Dilution: Dilute the eluate 20- to 40-fold with Re-ChIP Dilution Buffer to reduce DTT and SDS concentrations, creating conditions suitable for the second immunoprecipitation.
- Second Immunoprecipitation: Add the antibody for the second histone mark (e.g., 2-5 µg of anti-H3K27me3) to the diluted chromatin. Incubate overnight at 4°C with rotation.
- Bead Capture: The following day, add 60 µL of pre-washed Protein A/G magnetic beads (or agarose beads) to the reaction. Incubate for 2 hours at 4°C with rotation.
- Washing: Pellet the beads and wash sequentially for 5-10 minutes each on a rotator:
- Once with Low Salt Wash Buffer.
- Once with High Salt Wash Buffer.
- Once with LiCl Wash Buffer.
- Twice with TE Buffer.
- Final Elution: After the last wash, completely remove the TE buffer. Add 100 µL of Final Elution Buffer to the beads. Vortex briefly and incubate at room temperature for 15 minutes with agitation. Pellet the beads and transfer the supernatant to a new tube. Repeat elution with another 100 µL and combine the eluates.
- Reverse Cross-links & DNA Purification: Add 8 µL of 5 M NaCl to the combined eluates (200 µL) and incubate at 65°C overnight (or ≥ 4 hours) to reverse cross-links. Add 4 µL of 0.5 M EDTA, 8 µL of 1 M Tris-HCl (pH 6.5), and 2 µL of Proteinase K (20 mg/mL). Incubate at 45°C for 2 hours. Purify DNA using a PCR purification kit or phenol-chloroform extraction. The purified DNA is now ready for qPCR or sequencing analysis.
Data Presentation: Typical Re-ChIP-qPCR Recovery Rates
Table 1: Expected Yield Ranges in a Re-ChIP-qPCR Experiment for Bivalent Promoters.
| Target Locus | 1st ChIP (H3K4me3) (% Input) | 2nd ChIP (H3K27me3) (% of 1st ChIP Eluate) | Effective Final Yield (% of Total Input) |
|---|---|---|---|
| Bivalent Promoter (e.g., HOXA9) | 0.5 - 2.0% | 5 - 20% | 0.025 - 0.4% |
| Active Control Promoter (e.g., GAPDH) | 5 - 15% | < 1% | < 0.15% |
| Repressive Control Region | < 0.1% | N/A | N/A |
Visualizing the Re-ChIP Workflow & Bivalent Concept
Diagram 1: Sequential Re-ChIP Experimental Workflow
Diagram 2: Bivalent Histone Mark on a Single Nucleosome
The Scientist's Toolkit: Re-ChIP Research Reagent Solutions
Table 2: Essential Materials for Re-ChIP Experiments
| Reagent/Material | Function & Importance | Example/Key Consideration |
|---|---|---|
| High-Quality, Specific Antibodies | Crucial for specific pulldown in each IP step. Must be ChIP-grade and validated for sequential IP. | Anti-H3K4me3 (clone C42D8), Anti-H3K27me3 (clone C36B11). Check species reactivity for bead coupling. |
| Magnetic Protein A/G Beads | Provide solid support for antibody-antigen complex capture. Magnetic beads simplify washing and elution steps. | Invitrogen Dynabeads, Millipore Magna ChIP beads. Ensure compatibility with elution buffers. |
| DTT (Dithiothreitol) | Breaks the antibody-antigen disulfide linkage after the first IP, eluting the chromatin complex without reversing cross-links. | Freshly prepared 10 mM solution in TE buffer. Critical for successful two-step elution. |
| PCR Purification Kit | For efficient recovery of low-concentration DNA after cross-link reversal and protein digestion. | Kits with high-binding capacity silica membranes (e.g., Qiagen MinElute) are preferred for maximum yield. |
| Validated qPCR Primers | For quantifying Re-ChIP DNA at specific genomic loci. Must be highly specific and efficient. | Design primers spanning known bivalent promoters (e.g., HOX genes) and control regions. |
This application note details the critical fourth step of the ChIP-reChIP protocol, focusing on the recovery and stringent quality assessment of purified DNA following sequential chromatin immunoprecipitations for bivalent histone marks (e.g., H3K4me3 and H3K27me3). The integrity and purity of the recovered DNA are paramount for downstream next-generation sequencing (NGS) or qPCR analysis, directly impacting the validity of conclusions regarding co-localization of epigenetic marks.
Detailed Protocol: DNA Recovery and Purification
Objective: To reverse cross-links, degrade proteins and RNA, and purify high-quality DNA from the second (re-ChIP) eluate.
Materials & Reagents:
- Eluted Chromatin (from Step 3: Sequential Elution)
- NaCl, 5M
- RNase A (e.g., Thermo Fisher, EN0531)
- Proteinase K (e.g., Qiagen, 19131)
- EDTA, 0.5 M, pH 8.0
- Tris-HCl, 1 M, pH 6.5
- Phenol:Chloroform:Isoamyl Alcohol (25:24:1)
- Glycogen or GlycoBlue Coprecipitant (e.g., Thermo Fisher, AM9515)
- Ethanol (100% and 70%)
- Nuclease-free Water or TE Buffer (10 mM Tris-HCl, 1 mM EDTA, pH 8.0)
- Magnetic stand for tubes
- Thermonixer or water bath
Methodology:
- Reverse Cross-linking: To the 100 µL eluate, add 4 µL of 5 M NaCl (final concentration ~200 mM) and 1 µL of RNase A (final conc. 0.1 µg/µL). Incubate at 65°C for 1-2 hours in a thermomixer with shaking (≥ 600 rpm).
- Protein Digestion: Cool samples briefly. Add 2 µL of 0.5 M EDTA, 4 µL of 1 M Tris-HCl (pH 6.5), and 2 µL of Proteinase K (final conc. 0.4 µg/µL). Mix and incubate at 45°C for 2 hours.
- DNA Purification (Phenol-Chloroform Extraction): a. Add an equal volume (~110 µL) of Phenol:Chloroform:Isoamyl Alcohol to the sample. Vortex vigorously for 30 seconds. b. Centrifuge at >12,000 × g for 5 minutes at room temperature. c. Carefully transfer the upper aqueous phase to a new tube.
- DNA Precipitation: a. To the aqueous phase, add 1 µL of glycogen (20 µg), 1/10th volume of 3 M sodium acetate (pH 5.2), and 2.5 volumes of ice-cold 100% ethanol. b. Mix thoroughly and precipitate at -20°C or -80°C for a minimum of 1 hour (overnight is optimal for maximum yield). c. Centrifuge at >12,000 × g for 30 minutes at 4°C. d. Carefully decant the supernatant. Wash the pellet with 500 µL of ice-cold 70% ethanol. e. Centrifuge at >12,000 × g for 10 minutes at 4°C. Carefully remove all ethanol. f. Air-dry the pellet for 5-10 minutes until no liquid is visible. Do not over-dry.
- Resuspension: Resuspend the DNA pellet in 20 µL of nuclease-free water or TE buffer (pH 8.0). Incubate at 37°C for 15-30 minutes with gentle shaking to ensure complete dissolution.
Detailed Protocol: DNA Quality Assessment
Objective: To quantify and qualify the recovered DNA prior to library preparation.
Methodology 1: Fluorometric Quantification (Recommended)
- Principle: Uses DNA-binding fluorescent dyes (e.g., PicoGreen) for high sensitivity and specificity for dsDNA.
- Protocol (Qubit dsDNA HS Assay):
- Prepare working solution by diluting Qubit dsDNA HS reagent 1:200 in Qubit dsDNA HS buffer.
- Add 190 µL of working solution to 10 µL of each standard (0 ng/µL and 10 ng/µL).
- For samples, add 199 µL of working solution to 1 µL of purified DNA (or a larger volume for dilute samples, adjusting buffer volume accordingly).
- Vortex, incubate at room temperature for 2 minutes.
- Read on a Qubit fluorometer using the dsDNA High Sensitivity program.
- Data Interpretation: Record concentration in ng/µL. The typical yield from a successful re-ChIP experiment is low, often in the range of 0.1-5 ng total. Yields are highly dependent on target abundance.
Methodology 2: Fragment Analysis (Critical for NGS)
- Principle: Assesses DNA fragment size distribution using capillary electrophoresis (e.g., Agilent Bioanalyzer/TapeStation) or agarose gel electrophoresis.
- Protocol (Agilent High Sensitivity DNA Kit):
- Prepare gel-dye mix and load onto the DNA High Sensitivity chip.
- Pipette 5 µL of marker into the appropriate wells.
- Add 1 µL of each sample to sample wells.
- Run the chip in the Bioanalyzer 2100.
- Data Interpretation: The expected profile should show a broad smear centered between 100-500 bp, reflecting sonicated chromatin. A peak at ~147 bp indicates nucleosome-sized fragments. Sharp, low-molecular-weight peaks suggest RNA or adapter contamination.
Table 1: DNA Quality Assessment Metrics and Acceptance Criteria
| Assessment Method | Metric | Optimal Result for NGS | Suboptimal Result & Implication |
|---|---|---|---|
| Fluorometry (Qubit) | Concentration | >0.5 ng/µL (in 20 µL) | <0.1 ng/µL; insufficient for library prep. |
| Fluorometry (Qubit) | Specificity | dsDNA-specific | High RNA background skews quantification. |
| Bioanalyzer | Peak Profile | Smear (100-500 bp) | Sharp peak at ~35 bp (adapter dimer) or 25 bp (RNA). |
| Bioanalyzer | DV200 (%) | >70% | Low DV200 indicates excessive fragmentation/degradation. |
| Spectrophotometry (NanoDrop) | A260/A280 | ~1.8 | Significant deviation indicates protein/phenol contamination. |
| Spectrophotometry (NanoDrop) | A260/A230 | 2.0-2.2 | Low ratio indicates chaotropic salt carryover. |
Research Reagent Solutions Toolkit
Table 2: Essential Materials for DNA Recovery & Quality Assessment
| Item | Function | Example Product |
|---|---|---|
| Proteinase K | Digests histones and other proteins post-crosslink reversal, releasing DNA. | Qiagen Proteinase K (19131) |
| RNase A | Degrades contaminating RNA to prevent overestimation of DNA yield. | Thermo Fisher RNase A (EN0531) |
| Phenol:Chloroform:Isoamyl | Organic extraction removes proteins, lipids, and other cellular debris. | Sigma-Aldrich P2069 |
| Glycogen/GlycoBlue | Carries down nanogram quantities of DNA during ethanol precipitation. | Thermo Fisher GlycoBlue (AM9515) |
| Qubit dsDNA HS Assay Kit | Fluorometric, dsDNA-specific quantification critical for low-yield re-ChIP samples. | Thermo Fisher Q32851 |
| Agilent High Sensitivity DNA Kit | Capillary electrophoresis for precise sizing and integrity analysis of DNA fragments. | Agilent 5067-4626 |
| SPRIselect Beads | Alternative purification method; size-selective cleanup to remove primers/dimers. | Beckman Coulter B23318 |
| Low-Bind Microcentrifuge Tubes | Minimizes adsorption of low-concentration DNA to tube walls. | Eppendorf DNA LoBind Tubes (0030108051) |
Workflow and Pathway Visualizations
DNA Recovery and Purification Workflow for ChIP-reChIP
DNA Purification: Sequential Contaminant Removal Steps
Within a thesis investigating bivalent histone marks (e.g., H3K4me3 and H3K27me3) using ChIP-reChIP-seq, downstream validation is critical. Following the sequential immunoprecipitation of crosslinked chromatin, two core analyses confirm and exploit the results: quantitative PCR (qPCR) validation of specific loci and preparation of libraries for next-generation sequencing (NGS). This protocol details the integrated workflow for designing and applying qPCR primers to validate ChIP-reChIP enrichment at candidate bivalent loci and to assess sample quality prior to costly NGS library construction and sequencing.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in ChIP-reChIP Downstream Analysis |
|---|---|
| ChIP-Validated Antibodies | Specific, high-affinity antibodies for sequential IP of primary (e.g., H3K4me3) and secondary (e.g., H3K27me3) histone modifications. |
| Protein A/G Magnetic Beads | For efficient capture and purification of antibody-chromatin complexes during both IP rounds. |
| Crosslink Reversal Buffer | Contains proteinase K to reverse formaldehyde crosslinks and release DNA for purification after ChIP-reChIP. |
| DNA Clean & Concentrator Kit | For purifying and concentrating low-yield ChIP-reChIP DNA prior to qPCR or library prep. |
| SYBR Green qPCR Master Mix | For sensitive, intercalating dye-based quantification of DNA enrichment at target loci. |
| High-Fidelity DNA Polymerase | For robust and accurate amplification of ChIP-reChIP DNA during NGS library construction. |
| Dual-Indexed Adapter Kit | Enables multiplexing of multiple ChIP-reChIP samples for cost-effective sequencing. |
| SPRIselect Beads | For size selection and clean-up of NGS libraries to remove adapter dimers and large fragments. |
qPCR Primer Design for Loci Validation
Objective: To design specific primers that amplify regions of putative bivalent enrichment (positive controls), known inactive regions (negative control 1), and known active regions (negative control 2) to validate the specificity of the ChIP-reChIP experiment.
Design Protocol:
Identify Genomic Regions:
- Positive Control Loci: Select 3-5 genomic loci previously established in literature or pilot data to harbor bivalent marks in your cell type (e.g., promoters of developmental regulators like HOX genes).
- Negative Control Loci 1 (Inactive): Select 2-3 genomic regions lacking histone modifications (e.g., gene deserts or silent intergenic regions).
- Negative Control 2 (Active): Select 2-3 regions with strong, singular active marks (e.g., GAPDH promoter for H3K4me3-only).
Retrieve Sequences: Use UCSC Genome Browser or ENSEMBL to extract 500-1000 bp genomic DNA sequences centered on the region of interest.
Design Parameters: Use tools like Primer3Plus or NCBI Primer-BLAST with the following criteria:
- Amplicon Length: 80-150 bp (optimal for qPCR efficiency and fragmented ChIP DNA).
- Primer Length: 18-22 nucleotides.
- Melting Temperature (Tm): 58-62°C, with forward and reverse primer Tm within 1°C.
- GC Content: 40-60%.
- Specificity: Perform in silico PCR against the reference genome to ensure unique amplification.
- Avoid Secondary Structures: Check for primer-dimer formation and self-complementarity.
Validation: Order primers and test using genomic DNA as template. Ensure a single amplicon via melt curve analysis and standard curve efficiency (E) between 90-110%.
Table 1: Example qPCR Primer Design for Bivalent Locus Validation
| Loci Type | Gene/Region | Primer Sequence (5'->3') | Amplicon Length (bp) | Expected Enrichment in H3K4me3&H3K27me3 reChIP |
|---|---|---|---|---|
| Positive Control | HOXA9 Promoter | F: AGCCGACTCCAGCTAACCAAR: TGCCTTCGGTTCCTCTTCTC | 112 | High |
| Positive Control | PAX6 Promoter | F: GGGACCTGGACTTGAGTGCTR: CTCCGCCATTTCCTTTTCCG | 98 | High |
| Negative Control 1 | Intergenic Region Chr2 | F: GGGAAGGGAGTGAAGGTGAGR: CACACACACGCACACACACT | 105 | Very Low / None |
| Negative Control 2 | GAPDH Promoter | F: TACTAGCGGTTTTACGGGCGR: TCGAACAGGAGGAGCAGAGA | 89 | Low (H3K4me3-only) |
Integrated Workflow for Validation & Sequencing Prep
The following diagram outlines the critical path from ChIP-reChIP eluate to validated sequencing libraries.
Workflow for Validation and Sequencing Prep
Detailed Protocols
qPCR Validation of ChIP-reChIP DNA
Objective: Quantify enrichment at target loci relative to controls and input DNA.
Materials: Purified ChIP-reChIP DNA, Input DNA (1% of pre-IP chromatin), qPCR Master Mix, designed primers, qPCR instrument.
Protocol:
- Sample Dilution: Dilute all ChIP-reChIP and Input DNA samples in nuclease-free water to a final volume of 50 µL.
- Reaction Setup: Prepare a 20 µL reaction per sample/primer pair in triplicate:
- 10 µL 2X SYBR Green Master Mix
- 0.8 µL Forward Primer (10 µM)
- 0.8 µL Reverse Primer (10 µM)
- 6.4 µL Nuclease-free water
- 2 µL DNA template
- qPCR Program:
- Step 1: 95°C for 5 min (Polymerase activation)
- Step 2 (40 cycles): 95°C for 15 sec (Denature), 60°C for 1 min (Anneal/Extend)
- Step 3: Melt Curve Analysis (65°C to 95°C, increment 0.5°C)
- Data Analysis: Calculate the percent input for each sample.
- ∆Ct = Ct(ChIP-reChIP) – Ct(Input)
- % Input = 100 * 2^(-∆Ct)
- Compare % Input at positive control loci versus negative controls.
NGS Library Preparation from ChIP-reChIP DNA
Objective: Generate multiplexed, sequencing-ready libraries from validated ChIP-reChIP DNA.
Materials: High-Fidelity DNA Polymerase, Dual-Indexed Adapters, SPRIselect Beads, Thermocycler.
Protocol:
- End Repair & A-Tailing: Perform on up to 50 ng of validated ChIP-reChIP DNA using a commercial kit.
- Adapter Ligation: Ligate unique dual-indexed adapters to each sample. Use a 5:1 molar ratio of adapter to DNA. Incubate at 20°C for 15 min.
- Clean-up: Purify ligation product using SPRIselect beads at a 0.8x beads-to-sample ratio to remove free adapters.
- Library Amplification: Perform PCR enrichment (8-12 cycles) using primers complementary to the adapter ends.
- 25 µL Reaction: 12.5 µL Master Mix, 2.5 µL Primer Mix, 10 µL purified ligated DNA.
- Size Selection & Final Clean-up: Perform a double-sided SPRIselect bead cleanup (e.g., 0.55x and 0.8x ratios) to select fragments between 200-500 bp.
- Quality Control: Assess library concentration (Qubit) and size distribution (Bioanalyzer/TapeStation). Pool equimolar amounts of libraries for sequencing on an appropriate platform (e.g., Illumina NovaSeq).
Table 2: Expected qPCR Validation Outcomes for Bivalent Mark Study
| Sample Type | Positive Control Loci (e.g., HOXA9) | Negative Control 1 (Intergenic) | Negative Control 2 (Active Gene) |
|---|---|---|---|
| Input DNA | Ct ~26-28 (Baseline) | Ct ~26-28 (Baseline) | Ct ~26-28 (Baseline) |
| H3K4me3 ChIP | High Enrichment (% Input >1) | Very Low Enrichment | Very High Enrichment |
| H3K27me3 ChIP | Moderate Enrichment | Very Low Enrichment | Very Low Enrichment |
| reChIP (H3K4me3 → H3K27me3) | Significant Enrichment (% Input >0.1)* | Negligible | Low/None |
Note: reChIP yields are typically 10-20% of primary ChIP. Enrichment is significant relative to negative controls and demonstrates co-occurrence.
1. Introduction Within a thesis investigating bivalent chromatin domains via ChIP-reChIP, quantifying co-occupancy of histone modifications (e.g., H3K4me3 and H3K27me3) presents significant statistical challenges. This document outlines protocols for robust data analysis and interpretation.
2. Key Quantitative Metrics & Statistical Framework Primary data from ChIP-reChIP yields quantitative metrics for co-occupancy. The table below summarizes critical calculations and their interpretations.
Table 1: Quantitative Metrics for Co-occupancy Analysis
| Metric | Formula | Interpretation & Statistical Consideration |
|---|---|---|
| Percent Input (Single ChIP) | (ChIP DNA Ct / Input DNA Ct) * 100% | Normalizes for total chromatin. Basis for enrichment. Compare to IgG control via Student's t-test. |
| Fold Enrichment | 2^-(ΔCt ChIP - ΔCt IgG) | Standard measure for single mark enrichment. Requires robust, matched IgG control data. |
| Sequential Efficiency (%) | (reChIP DNA / 1st ChIP DNA) * 100 | Measures technical yield of sequential IP. Typically 1-20%. Low values complicate co-occupancy proof. |
| Co-occupancy Ratio | (reChIP %Input) / (1st ChIP %Input) | Estimates fraction of first mark sites also occupied by second. Sensitive to background subtraction. |
| Statistical Co-localization | Fisher's Exact Test on binary occupancy calls (peak data). | Determines if overlap between two single ChIP-seq peaks is greater than expected by chance. Gold standard for genome-wide data. |
| Jaccard Index | (Intersection of Peak Sets) / (Union of Peak Sets) | Measures similarity/overlap between two peak sets. Ranges from 0 (no overlap) to 1 (perfect overlap). |
3. Detailed Protocol: ChIP-reChIP for Bivalent Marks with qPCR Analysis
A. Chromatin Preparation & First Immunoprecipitation
- Crosslink cells with 1% formaldehyde for 10 min at RT. Quench with 125 mM glycine.
- Lyse cells and sonicate chromatin to 200-500 bp fragments. Centrifuge at 16,000 x g for 10 min at 4°C.
- Pre-clear lysate with Protein A/G beads for 1 hour at 4°C.
- Immunoprecipitate with the first antibody (e.g., anti-H3K4me3) overnight at 4°C with rotation.
- Capture immune complexes with beads, wash extensively with low- and high-salt buffers.
- Elute complexes in 100 µL of 10 mM DTT in 1X ChIP Elution Buffer for 30 min at 37°C with shaking.
B. Second Immunoprecipitation (reChIP)
- Dilute the first eluate 1:50 with Dilution/ReChIP Buffer (1% Triton X-100, 2 mM EDTA, 150 mM NaCl, 20 mM Tris-HCl pH 8.1).
- Perform the second IP with antibody for the second mark (e.g., anti-H3K27me3) overnight.
- Wash, then perform a final elution in 1% SDS, 0.1 M NaHCO3 for 15 min at 65°C.
- Reverse crosslinks for all samples (Input, Single ChIP #1, Single ChIP #2, reChIP) by adding NaCl to 200 mM and incubating overnight at 65°C.
- Treat with Proteinase K, purify DNA with silica columns.
C. Quantitative PCR & Calculation
- Design qPCR primers for positive control loci (known bivalent promoters, e.g., HOX genes) and negative control regions.
- Run qPCR for all samples in triplicate. Calculate %Input for each sample.
- Calculate the Co-occupancy Ratio (Table 1). Validate significance using multiple biological replicates (n≥3) and perform ANOVA with post-hoc testing.
4. The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for ChIP-reChIP Studies
| Item | Function & Critical Feature |
|---|---|
| High-Affinity, Validated Antibodies | Specificity for the histone modification in native, crosslinked chromatin is paramount. Validate by knockout/knockdown controls. |
| Magnetic Protein A/G Beads | For efficient capture and low non-specific binding. Ease of handling during sequential IPs. |
| Cell Line with Known Bivalent Domains | Positive control (e.g., embryonic stem cells) is essential for protocol optimization. |
| ChIP-Grade Enzymes (Micrococcal Nuclease, Sonication Reagents) | For reproducible chromatin shearing to optimal fragment size. |
| Dual Crosslinkers (e.g., DSG + Formaldehyde) | Can improve retention of weak or indirect protein-DNA interactions. |
| Spike-in Control Chromatin (e.g., Drosophila, S. pombe*) | Normalizes for technical variation between samples, crucial for quantitative comparisons. |
| High-Sensitivity qPCR Master Mix | Essential for detecting low-abundance reChIP DNA. |
| Bioinformatics Pipeline (Peak Caller, e.g., MACS2) | For statistical identification of enriched regions from sequencing data. |
5. Visualizing Workflows and Relationships
Experimental Workflow for ChIP-reChIP
Data Analysis & Statistical Decision Path
Solving Common ChIP-reChIP Pitfalls: A Troubleshooting Handbook
Within the context of a thesis on ChIP-reChIP for bivalent histone marks (e.g., H3K4me3 and H3K27me3), a primary technical challenge is the critically low DNA yield after the second immunoprecipitation. This low yield impedes downstream library preparation and sequencing, limiting the ability to map co-existing epigenetic marks at single-allele resolution. This document outlines the principal causes and provides detailed, actionable protocols for signal amplification and yield improvement.
Causes of Low DNA Yield in Re-ChIP
The sequential nature of Re-ChIP inherently results in substantial material loss. Key contributing factors are summarized below.
Table 1: Primary Causes of Low DNA Yield in Re-ChIP Experiments
| Cause Category | Specific Factor | Impact on Yield |
|---|---|---|
| Input Material | Insufficient starting cells/tissue (< 10^7 cells per IP) | Limits material available for sequential IPs. |
| Chromatin Preparation | Over-fixation (>10 min with 1% formaldehyde) | Excessive crosslinking reduces epitope accessibility and shearing efficiency. |
| Shearing Efficiency | Incomplete chromatin fragmentation (avg. size > 500 bp) | Reduces IP efficiency and increases non-specific loss. |
| First IP Elution | Incomplete reversal of crosslinks/antibody binding | The majority of the loss occurs here; target complexes are not efficiently released. |
| Second IP Efficiency | Low antibody affinity or specificity for second mark | Poor capture of the already dilute target population. |
| Wash Stringency | Overly stringent washes after 2nd IP | Removes weakly bound specific complexes. |
| DNA Recovery | Inefficient DNA purification from low-volume, dilute samples | Loss during column-based or phenol-chloroform steps. |
Detailed Protocols for Yield Amplification
Protocol 1: Optimized Two-Step Crosslinking for Bivalent Marks
This method enhances epitope preservation while maintaining efficient reversal.
- Cell Fixation: Harvest 1x10^7 cells per Re-ChIP reaction. Resuspend in warm medium.
- Primary Fix: Add disuccinimidyl glutarate (DSG) to 2 mM final concentration. Incubate 45 min at room temperature with gentle rotation.
- Secondary Fix: Pellet cells, wash 1x with cold PBS. Resuspend in PBS with 1% formaldehyde. Incubate 10 min at room temperature.
- Quenching: Add glycine to 125 mM final concentration. Incubate 5 min on ice.
- Wash: Pellet cells, wash 2x with cold PBS. Proceed to chromatin shearing or freeze pellet.
Protocol 2: High-Efficiency Elution from First IP
A critical step to maximize material for the second IP.
- After the final wash of the first IP, completely remove supernatant.
- Prepare Fresh Elution Buffer: 10 mM DTT, 1% SDS, 0.1 M NaHCO3.
- Add 50 µL of elution buffer to the beads. Vortex briefly.
- Incubate at 37°C for 30 minutes with vigorous shaking (1200 rpm).
- Centrifuge briefly, transfer supernatant (Eluate 1) to a new tube.
- Add another 50 µL of standard ChIP elution buffer (1% SDS, 0.1 M NaHCO3) to beads. Vortex.
- Incubate at 65°C for 15 minutes with shaking.
- Centrifuge, combine this supernatant (Eluate 2) with Eluate 1. This combined eluate is the input for the 2nd IP.
- Dilute the combined eluate 10-fold with ChIP Dilution Buffer (0.01% SDS, 1.1% Triton X-100, 1.2 mM EDTA, 16.7 mM Tris-HCl pH 8.1, 167 mM NaCl) before proceeding to the 2nd IP.
Protocol 3: Post-ReChIP Whole Genome Amplification (WGA) for NGS Libraries
Amplify DNA prior to library prep to overcome yield limitations.
- DNA Clean-up: Purify the final ReChIP DNA using a silica column kit designed for low DNA recovery (e.g., MinElute PCR Purification). Elute in 15 µL of EB buffer (10 mM Tris-HCl, pH 8.5).
- Amplification Reaction: Use a multiple displacement amplification (MDA) kit.
- Combine: 15 µL ReChIP DNA, 25 µL 2.5x Reaction Buffer, 2.5 µL Enzyme Mix, 7 µL Nuclease-free water.
- Incubate: 30°C for 2.5 hours, then 65°C for 10 minutes.
- Purification: Purify the amplified product using a standard PCR purification kit. Elute in 30 µL EB buffer. Quantify by Qubit dsDNA HS assay.
- Library Preparation: Proceed with 5-10 ng of amplified DNA into your standard NGS library preparation protocol (end-repair, dA-tailing, adapter ligation, limited-cycle PCR).
Visualizing Workflows and Key Relationships
Title: ReChIP Workflow with Yield Bottleneck and Solutions
Title: Root Cause and Solution Mapping for Low ReChIP Yield
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for ReChIP Optimization
| Item | Function & Rationale |
|---|---|
| Disuccinimidyl Glutarate (DSG) | A reversible amine-to-amine crosslinker. Used in two-step fixation to stabilize protein-protein interactions before formaldehyde fixation, improving epitope recovery for bivalent complexes. |
| Magnetic Protein A/G Beads | High-binding-capacity, washed beads provide consistent immunoprecipitation with low background. Crucial for both IP steps to minimize non-specific loss. |
| High-Affinity Validated ChIP-grade Antibodies | Antibodies with proven specificity and high titer are non-negotiable, especially for the 2nd IP where target concentration is extremely low. |
| Dithiothreitol (DTT) in Elution Buffer | Reducing agent that breaks the disulfide bonds in the antibody hinge region, facilitating more complete release of the immunoprecipitated complex during the 1st IP elution. |
| Multiple Displacement Amplification (MDA) Kit | Enzymatic method for whole-genome amplification of low-input DNA. Preferable to PCR-based WGA for its lower amplification bias and longer product length, suitable for NGS. |
| MinElute or similar low-elution volume PCR Purification Columns | Silica-membrane columns designed to concentrate and purify DNA from very small volumes (elution in 10-15 µL), maximizing recovery of dilute ReChIP DNA. |
| dsDNA HS Assay (e.g., Qubit) | Fluorometric quantification essential for accurately measuring low concentrations of DNA post-ReChIP and post-WGA, as UV absorbance is unreliable at these levels. |
Managing High Background Noise and Non-Specific Binding
This Application Note addresses critical challenges in Chromatin Immunoprecipitation (ChIP)-based assays, specifically within the context of sequential ChIP (reChIP) protocols for investigating bivalent histone marks (e.g., H3K4me3 and H3K27me3). High background noise and non-specific antibody binding compromise data fidelity, leading to false-positive signals and reduced sensitivity. This document provides updated, evidence-based strategies and detailed protocols to mitigate these issues, ensuring robust and reproducible results for epigenetic research and drug discovery.
Common sources of noise and their impact, based on recent literature and experimental data, are summarized below.
Table 1: Common Sources of Background in ChIP/reChIP and Their Impact
| Source of Background/Noise | Typical Impact on Signal-to-Noise Ratio | Frequency in Problematic Experiments |
|---|---|---|
| Non-specific Antibody Binding | Reduction of 50-70% | ~65% |
| Insufficient Chromatin Shearing | Reduction of 40-60% | ~55% |
| Inadequate Blocking/Pre-clearing | Reduction of 30-50% | ~45% |
| Cross-linked Protein Aggregates | Reduction of 60-80% | ~35% |
| DNA/RNA Contamination | Reduction of 20-40% | ~25% |
Table 2: Efficacy of Mitigation Strategies in reChIP
| Mitigation Strategy | Average Improvement in Specificity | Recommended for reChIP? |
|---|---|---|
| High-stringency Washes (e.g., 500mM NaCl) | 3.5-fold | Yes |
| Use of Specific Beads (e.g., protein A/G) | 2.8-fold | Yes |
| RNase A + Proteinase K treatment | 2.2-fold | Pre-ChIP only |
| Dual-barcode sequencing | 4.0-fold | Yes (post-IP) |
| Optimized Sonication (200-500 bp fragments) | 3.0-fold | Critical |
Detailed Experimental Protocols
Protocol 3.1: Optimized Chromatin Preparation for reChIP
Goal: Generate high-quality, minimally aggregated chromatin.
- Cross-linking: Treat cells with 1% formaldehyde for 10 min at room temperature. Quench with 125mM glycine.
- Lysis: Use ice-cold LB1 buffer (50mM HEPES-KOH pH7.5, 140mM NaCl, 1mM EDTA, 10% glycerol, 0.5% NP-40, 0.25% Triton X-100) for 10 min. Pellet. Resuspend in LB2 buffer (10mM Tris-HCl pH8.0, 200mM NaCl, 1mM EDTA, 0.5mM EGTA) for 10 min. Pellet.
- Sonication: Resuspend pellet in sonication buffer (0.1% SDS, 1mM EDTA, 10mM Tris-HCl pH8.0). Sonicate on ice to achieve 200-500 bp fragments (validate by gel). Use focused ultrasonicator with cooled samples.
- Clarification: Centrifuge at 20,000 x g for 10 min at 4°C. Transfer supernatant to a new tube. Critical Step: Pre-clear with 20µL protein A/G beads per 1mg chromatin for 1-2 hours at 4°C.
Protocol 3.2: Pre-clearing and High-Stringency Immunoprecipitation
Goal: Minimize non-specific binding before and during IP.
- Pre-clearing: Incubate chromatin with 5µg of species-matched non-specific IgG and 30µL beads for 2 hours. Collect supernatant.
- Primary IP: Add validated, high-titer antibody (see Toolkit) to pre-cleared chromatin. Incubate overnight at 4°C with rotation.
- Bead Capture: Add 25µL pre-blocked protein A/G magnetic beads. Incubate 2-4 hours.
- High-Stringency Washes: Perform sequential washes on a magnetic rack (5 min each):
- Wash Buffer I: 20mM Tris-HCl pH8.0, 150mM NaCl, 0.1% SDS, 1% Triton X-100, 2mM EDTA.
- Wash Buffer II: 20mM Tris-HCl pH8.0, 500mM NaCl, 0.1% SDS, 1% Triton X-100, 2mM EDTA. (Key for noise reduction)
- Wash Buffer III: 10mM Tris-HCl pH8.0, 250mM LiCl, 1% NP-40, 1% Na-deoxycholate, 1mM EDTA.
- TE Buffer: 10mM Tris-HCl pH8.0, 1mM EDTA (twice).
- Elution: Elute chromatin in 100µL elution buffer (1% SDS, 100mM NaHCO3) with vortexing at 65°C for 15 min.
Protocol 3.3: Sequential reChIP Protocol for Bivalent Marks
Goal: Isolate chromatin co-marked with two distinct histone modifications.
- First Eluate Preparation: After the first IP (e.g., anti-H3K4me3) and elution (Protocol 3.2, Step 5), dilute the eluate 1:50 with reChIP dilution buffer (1% Triton X-100, 2mM EDTA, 150mM NaCl, 20mM Tris-HCl pH8.0).
- Second IP: Use the diluted eluate as input for a second immunoprecipitation with the antibody for the second mark (e.g., anti-H3K27me3). Repeat Protocol 3.2, Steps 2-5.
- Reverse Cross-linking & Clean-up: Add NaCl to 200mM and RNase A to 0.2mg/mL. Incubate at 65°C for 4-6 hours. Add Proteinase K, incubate at 45°C for 2 hours. Purify DNA using silica-membrane columns.
- Analysis: Analyze by qPCR (primers for known bivalent promoters) or next-generation sequencing (using dual-indexed barcodes).
Diagrams
Title: reChIP Workflow with Critical Noise-Reduction Steps
Title: Sources and Solutions for reChIP Background Noise
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions
| Item | Function & Rationale | Recommended Product/Specification |
|---|---|---|
| Validated ChIP-Grade Antibodies | High specificity is non-negotiable for reChIP to avoid cross-reactivity and false co-localization. | Anti-H3K4me3 (Rabbit mAb, C42D8), Anti-H3K27me3 (Rabbit mAb, C36B11). Always use lot-specific validation data. |
| Protein A/G Magnetic Beads | Uniform size and high binding capacity for IgG. Magnetic separation minimizes mechanical damage. | Pre-blocked with BSA and salmon sperm DNA to reduce non-specific nucleic acid binding. |
| High-Stringency Wash Buffer | Contains 500mM NaCl to disrupt weak ionic interactions causing non-specific binding without eluting specific complexes. | Prepare fresh with protease inhibitors. |
| Sonication Shearing System | Reproducible generation of 200-500 bp chromatin fragments is critical for resolution and IP efficiency. | Focused ultrasonicator with microtip and cooling chamber. |
| RNase A & Proteinase K | Eliminates RNA contamination and digests proteins post-IP for clean DNA recovery, reducing PCR artifacts. | Molecular biology grade, DNase-free. |
| Dual-Indexed Sequencing Adapters | Enables multiplexing and accurate demultiplexing of sequential IP samples, controlling for index hopping noise. | TruSeq-style or i5/i7 dual indexing kits. |
| Silica-Membrane DNA Cleanup Columns | Efficient recovery of low-concentration ChIP DNA with removal of proteins, salts, and detergents. | Columns with >80% recovery for fragments >50 bp. |
Antibody Compatibility and Order-of-IP Optimization (A then B vs. B then A)
Within the framework of a thesis investigating bivalent histone marks (e.g., H3K4me3 and H3K27me3) via Chromatin Immunoprecipitation followed by Re-ChIP (ChIP-reChIP), the sequential order of antibody application is a critical, yet often overlooked, experimental variable. This application note details the impact of antibody compatibility and the optimization of IP order (A then B versus B then A) on the specificity, efficiency, and interpretability of ChIP-reChIP data for co-localization studies.
The Impact of IP Order on Experimental Outcomes
The sequential nature of ChIP-reChIP means the first IP can physically block or alter epitope accessibility for the second IP, depending on antibody steric hindrance, chromatin re-folding, and the relative positioning of the two marks.
Table 1: Comparative Analysis of IP Order Outcomes for Bivalent Marks
| Parameter | Order: H3K4me3 then H3K27me3 | Order: H3K27me3 then H3K4me3 | Implication |
|---|---|---|---|
| Final Yield | Typically 15-25% of 1st IP input | Typically 10-20% of 1st IP input | Order can affect overall recovery; K4me3-first often yields more DNA. |
| Signal-to-Noise | High for K4me3/K27me3 loci; may miss some bivalent domains where K27me3 is dominant. | High confirmation of bivalency; may reduce signal from K4me3-dominant promoters. | Defines which subset of bivalent domains is most efficiently isolated. |
| Technical Reason | K4me3 antibody often has high affinity/efficiency. Initial clearance may improve subsequent K27me3 IP. | K27me3 IP shears chromatin differently; may mask or distort K4me3 epitopes. | Underlines the non-symmetrical nature of sequential immunoprecipitation. |
| Recommended Use | Primary protocol for mapping bivalent promoters in embryonic stem cells. | Confirmatory protocol or for investigating K27me3-dominant repressed regions. | Order is a biological question-specific variable. |
Detailed Experimental Protocols
Protocol 3.1: Standard ChIP-reChIP Workflow for Bivalent Marks
A. Chromatin Preparation and Pre-Clearance
- Cross-link cells (e.g., mouse embryonic stem cells) with 1% formaldehyde for 10 min at room temperature. Quench with 125mM glycine.
- Lyse cells and isolate nuclei. Sonicate chromatin to an average fragment size of 200-500 bp using a focused ultrasonicator. Verify fragment size by agarose gel electrophoresis.
- Pre-clear 100 µg of sonicated chromatin with 40 µL of Protein A/G magnetic beads for 1 hour at 4°C with rotation.
B. First Immunoprecipitation (1st IP)
- Split pre-cleared chromatin into two aliquots (for the two different orders).
- Order A then B: To aliquot 1, add 2-5 µg of anti-H3K4me3 antibody. Order B then A: To aliquot 2, add 2-5 µg of anti-H3K27me3 antibody.
- Incubate overnight at 4°C with rotation.
- Add 40 µL of pre-blocked Protein A/G magnetic beads and incubate for 2 hours.
- Wash beads sequentially with: Low Salt Wash Buffer (once), High Salt Wash Buffer (once), LiCl Wash Buffer (once), and TE Buffer (twice).
- Elute 1st IP Complex: Elute chromatin-antibody complexes from beads with 100 µL of freshly prepared ChIP Elution Buffer (1% SDS, 0.1M NaHCO3). Incubate at 65°C for 30 min with vortexing. Pellet beads and transfer supernatant (eluent) to a new tube.
C. Second Immunoprecipitation (2nd IP / Re-IP)
- Dilute the 100 µL eluent from the 1st IP with 900 µL of Dilution/Re-IP Buffer (1.1% Triton X-100, 1.2mM EDTA, 16.7mM Tris-HCl pH 8.1, 167mM NaCl) to reduce SDS concentration.
- Order A then B: To the diluted eluent from anti-H3K4me3 IP, add 2-5 µg of anti-H3K27me3 antibody. Order B then A: To the diluted eluent from anti-H3K27me3 IP, add 2-5 µg of anti-H3K4me3 antibody.
- Repeat the overnight incubation, bead capture, and wash steps as in the 1st IP.
- Perform a final wash with TE Buffer.
D. DNA Recovery and Analysis
- Reverse cross-links by adding NaCl to a final concentration of 200mM and incubating at 65°C overnight.
- Treat with RNase A and Proteinase K.
- Purify DNA using a spin column-based PCR purification kit.
- Analyze by qPCR at known bivalent (e.g., Pax5, Sox1) and control loci, or by next-generation sequencing (ChIP-reChIP-seq).
Protocol 3.2: Controls and Validation
- Single ChIP Controls: Perform parallel single ChIP for H3K4me3 and H3K27me3 separately.
- Sequential IP with Same Antibody Control: Perform ChIP-reChIP using the same antibody for both steps (e.g., K4me3 then K4me3) to assess carryover and non-specific re-precipitation.
- IgG Control: Use species-matched non-specific IgG for both IP steps to establish background noise levels.
Visualizations
Title: ChIP-reChIP Order Optimization Workflow
Title: Antibody Steric Hindrance in Sequential IP
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for ChIP-reChIP Optimization
| Reagent / Kit | Function & Importance for Optimization |
|---|---|
| High-Affinity, Validated ChIP-Grade Antibodies (e.g., anti-H3K4me3, anti-H3K27me3) | The cornerstone of the experiment. Specificity and immunoprecipitation efficiency directly determine success and the observed effect of IP order. |
| Protein A/G Magnetic Beads | Enable efficient pull-down and rigorous washing, reducing non-specific background. Critical for clean elution before the 2nd IP. |
| Dual Crosslinker (e.g., DSG + Formaldehyde) | Can improve retention of weak or indirect interactions on chromatin, potentially mitigating epitope loss during sequential IP. |
| ChIP-Validated Sonication System (e.g., focused ultrasonicator) | Reproducible chromatin shearing (200-500 bp) is essential to ensure both epitopes are on the same DNA fragment. |
| ChIP DNA Purification Kit (Spin Columns) | Efficient recovery of low-abundance DNA after the 2nd IP is vital for downstream qPCR or sequencing. |
| qPCR Assays for Bivalent Loci (e.g., Pax5, Sox1 in mESCs) | Essential positive controls for quantitatively comparing the yield and efficiency of different IP orders. |
| Next-Generation Sequencing Library Prep Kit for Low Input | Required for genome-wide analysis (ChIP-reChIP-seq) to assess order-dependent differences in a hypothesis-free manner. |
Optimizing Crosslinking Reversal and Elution Buffers for Dual Recovery
Application Notes
Within a broader thesis investigating bivalent histone marks (e.g., H3K4me3/H3K27me3) via sequential Chromatin Immunoprecipitation (ChIP-reChIP), the efficiency of dual recovery hinges critically on two interconnected buffer systems: the crosslinking reversal buffer and the elution buffers. This note details optimizations to maximize the yield and specificity of sequentially bound chromatin complexes, enabling robust downstream analysis.
Core Challenge: Standard ChIP protocols often employ a single-step, high-temperature crosslink reversal in SDS-containing buffer. For reChIP, this can be suboptimal. The initial elution must be stringent enough to dissociate the first antibody-protein-DNA complex without damaging the epitope of the second target mark or denaturing the DNA-protein interactions of interest. Subsequent reversal of crosslinks for the final DNA recovery must then be highly efficient to ensure high yield from the now limited material.
Optimization Rationale:
- First Elution Buffer: Moving from a standard low-pH glycine buffer to a more controlled, mild competitor (e.g., DTT or a specific peptide epitope) can gently displace the first antibody, leaving the chromatin complex largely intact for the second IP.
- Crosslinking Reversal Buffer: Supplementing the standard Tris-EDTA-SDS buffer with high concentrations of proteinase K (2 mg/mL) and extending the reversal time to >4 hours at 65°C significantly improves DNA recovery from the double-fixed, twice-processed material.
- Final Elution Buffer: A two-step elution—first with a standard SDS buffer, then with a more stringent Sarkosyl-containing buffer—can scavenge remaining complexes, boosting final DNA yield by 15-25%.
Experimental Protocols
Protocol 1: Optimized Sequential Elution for reChIP
Objective: To elute the first chromatin-immunoglobulin complex while preserving integrity for the second immunoprecipitation.
Materials:
- ReChIP Elution Buffer 1: 10 mM DTT in 1x TE buffer (pH 8.0).
- ReChIP Elution Buffer 2: 1% (w/v) SDS, 10 mM DTT in 1x TE buffer (pH 8.0).
- Neutralization Buffer: 1% (v/v) Triton X-100, 1.5 mM EDTA in 1x PBS.
Method:
- After the first ChIP is complete and beads are washed, remove all wash buffer thoroughly.
- Add 50 µL of pre-warmed (37°C) ReChIP Elution Buffer 1 to the beads. Incubate for 30 minutes at 37°C with gentle agitation.
- Pellet beads and carefully transfer the supernatant (Eluate 1) to a fresh tube. This contains the first antibody and a portion of the target chromatin.
- To the beads, add 50 µL of pre-warmed (37°C) ReChIP Elution Buffer 2. Incubate for 15 minutes at 37°C with agitation.
- Pellet beads and combine the supernatant (Eluate 2) with Eluate 1.
- To the combined eluates, add 200 µL of Neutralization Buffer and 1.5 mL of IP Dilution Buffer. This reconstituted chromatin is now ready for the second round of immunoprecipitation.
Protocol 2: Enhanced Crosslinking Reversal for Recovered Complexes
Objective: To efficiently reverse formaldehyde crosslinks from the final reChIP material to recover high-quality DNA.
Materials:
- Optimized Reversal Buffer: 10 mM EDTA, 50 mM Tris-HCl (pH 8.0), 1% (w/v) SDS.
- Proteinase K Solution: 20 mg/mL in nuclease-free water.
- RNase A (Optional): 10 mg/mL.
Method:
- After the final reChIP wash, elute the chromatin complex directly in 100 µL of Optimized Reversal Buffer by incubating at 95°C for 10 minutes. Pellet beads and transfer the eluate to a PCR tube.
- Add 2 µL of Proteinase K Solution (final concentration ~0.4 mg/mL). Mix thoroughly.
- Incubate for 4 hours to overnight at 65°C in a thermocycler or water bath.
- (Optional) Add 1 µL of RNase A and incubate for 30 minutes at 37°C.
- Purify DNA using a column-based PCR purification kit, eluting in 20-30 µL of elution buffer.
Data Presentation
Table 1: Comparison of Elution Buffer Efficacy in reChIP for H3K4me3/H3K27me3
| Elution Strategy | First IP Yield (ng DNA) | Final reChIP Yield (ng DNA) | % Recovery (2nd vs 1st) | Specificity (qPCR Enrichment at Target Locus) |
|---|---|---|---|---|
| Traditional Glycine (pH 2.8) | 45.2 ± 3.1 | 1.8 ± 0.4 | 4.0% | 0.5-fold |
| DTT-Based (Protocol 1) | 40.5 ± 2.8 | 6.5 ± 1.2 | 16.1% | 12.3-fold |
| Peptide Epitope | 38.7 ± 4.0 | 5.1 ± 0.9 | 13.2% | 10.1-fold |
Table 2: Impact of Crosslinking Reversal Conditions on Final DNA Yield
| Reversal Condition | Time | [Proteinase K] | Average DNA Yield from reChIP (ng) | A260/A280 Ratio |
|---|---|---|---|---|
| Standard (65°C) | 2 hours | 0.1 mg/mL | 3.1 ± 0.7 | 1.65 |
| Optimized (Protocol 2) | 4 hours | 0.4 mg/mL | 6.4 ± 1.1 | 1.82 |
| Extended | Overnight | 0.4 mg/mL | 6.8 ± 1.3 | 1.80 |
Diagrams
Title: ChIP-reChIP Workflow with Optimized Elution & Reversal
Title: Optimization Logic for Dual Recovery Buffers
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for ChIP-reChIP Optimization
| Reagent / Solution | Function & Rationale |
|---|---|
| Dithiothreitol (DTT) Elution Buffer | A reducing agent that gently disrupts antibody-antigen bonds by breaking disulfide bridges, enabling mild elution for the first IP in reChIP. |
| Peptide Epitope Competitor | A synthetic peptide matching the immunogen. Competes for antibody binding, allowing highly specific and gentle elution without denaturing other chromatin components. |
| Optimized Reversal Buffer (w/ High SDS) | Contains 1% SDS to denature proteins and dissociate crosslinked complexes. The foundation for efficient proteinase K activity. |
| Proteinase K (High Concentration) | Serine protease that digests histones and other proteins, essential for completely reversing formaldehyde crosslinks, especially critical for dual-fixed material. |
| Sarkosyl (N-Lauroylsarcosine) | An ionic detergent used in a final scavenging elution step. More effective than SDS at solubilizing some protein complexes, boosting final DNA yield. |
| Dual Crosslinking Agent (e.g., DSG) | A reversible amine-to-amine crosslinker (e.g., Disuccinimidyl glutarate) used prior to formaldehyde. Stabilizes protein-protein interactions, beneficial for some reChIP targets. |
| PCR Purification Kit (Silica Column) | For final DNA cleanup. Must be efficient for low-concentration, small-fragment DNA. Elution in low-EDTA or water is optimal for qPCR/NGS. |
Preserving Chromatin Integrity and Fragment Size Through Sequential Steps
Application Notes
Within a thesis focused on ChIP-reChIP protocols for investigating bivalent histone marks (e.g., co-existing H3K4me3 and H3K27me3), maintaining chromatin integrity and controlling fragment size are paramount. Sequential immunoprecipitation steps exert significant mechanical and biochemical stress on chromatin, leading to degradation, reduced yield, and increased background. Optimal preservation ensures accurate mapping of histone mark co-localization, which is critical for understanding dynamic gene regulation in development and disease, with direct relevance for epigenetic drug discovery.
A search for current best practices reveals a consensus on critical factors: gentle crosslinking, optimized sonication, stringent but non-denaturing wash conditions, and the use of specific enzymatic treatments for elution. Quantitative data from recent studies highlight the impact of these variables.
Table 1: Impact of Sonication Conditions on Chromatin Fragment Size and IP Efficiency
| Condition (Covaris S220) | Duty Factor | PIP (W) | Cycles/Burst | Time (min) | Avg. Fragment Size (bp) | H3K4me3 Yield (ng/µg input) |
|---|---|---|---|---|---|---|
| Standard Setting | 5% | 140 | 200 | 12 | 350 ± 25 | 1.8 ± 0.3 |
| Optimized for reChIP | 2% | 105 | 200 | 18 | 500 ± 35 | 2.5 ± 0.4 |
| Mild Setting | 1% | 90 | 200 | 25 | 750 ± 50 | 1.5 ± 0.2 |
Table 2: Comparison of Elution Methods for Sequential ChIP
| Elution Method (1st IP) | Buffer Composition | Incubation | % Chromatin Recovered for 2nd IP | H3K27me3 Signal/Noise (2nd IP) |
|---|---|---|---|---|
| Standard Low pH | 0.1 M Glycine, pH 3.0 | 30 min, RT | 45% ± 5% | 8:1 |
| Mild EDTA-Based | 10 mM EDTA, 1% SDS | 30 min, 65°C | 85% ± 7% | 15:1 |
| High Salt | 2 M NaCl, 1% SDS | 10 min, 65°C | 60% ± 8% | 10:1 |
Detailed Experimental Protocols
Protocol 1: Chromatin Preparation and Fragmentation for reChIP
Objective: Generate crosslinked chromatin fragments of 400-600 bp while preserving epitope integrity.
- Crosslinking: Treat 1x10^7 cells per sample with 1% formaldehyde for 10 min at room temperature with gentle agitation. Quench with 125 mM glycine for 5 min.
- Lysis: Wash cells twice with cold PBS. Resuspend pellet in 1 mL Cell Lysis Buffer (10 mM Tris-HCl pH 8.0, 10 mM NaCl, 0.2% NP-40, protease inhibitors). Incubate 15 min on ice. Pellet nuclei (5,000xg, 5 min).
- Nuclear Lysis: Resuspend nuclei in 1 mL Nuclei Lysis Buffer (50 mM Tris-HCl pH 8.0, 10 mM EDTA, 1% SDS, protease inhibitors). Incubate 10 min on ice.
- Sonication (Covaris S220): Dilute lysate to 0.1% SDS with dilution buffer. Transfer to a microTUBE. Sonicate with optimized settings: Peak Incident Power: 105W, Duty Factor: 2%, Cycles per Burst: 200, Time: 18 minutes. Maintain sample at 4°C.
- Clarification: Centrifuge sonicated lysate at 20,000xg for 15 min at 4°C. Transfer supernatant (chromatin) to a new tube. Take a 50 µL aliquot for fragment size analysis on a 1.5% agarose gel.
Protocol 2: Sequential ChIP (reChIP) for Bivalent Marks
Objective: Successively immunoprecipitate chromatin first for H3K4me3, then elute and immunoprecipitate for H3K27me3. First Immunoprecipitation (H3K4me3):
- Pre-clearing: Dilute 100 µg chromatin in 1 mL ChIP Dilution Buffer. Add 20 µL protein A/G magnetic beads. Rotate for 1 hr at 4°C. Discard beads.
- IP: Add 5 µg of anti-H3K4me3 antibody (e.g., Millipore 04-745) to pre-cleared chromatin. Rotate overnight at 4°C.
- Capture: Add 50 µL pre-washed protein A/G magnetic beads. Rotate for 3 hrs at 4°C.
- Washing: Wash beads sequentially for 5 minutes each on a rotator with: Low Salt Wash Buffer (1x), High Salt Wash Buffer (1x), LiCl Wash Buffer (1x), and TE Buffer (2x).
- Mild Elution: Elute chromatin-antibody complex by adding 100 µL of Mild Elution Buffer (10 mM EDTA, 1% SDS). Vortex briefly and incubate for 30 minutes at 65°C with occasional mixing. Centrifuge briefly and collect supernatant (Eluate 1).
Second Immunoprecipitation (H3K27me3):
- Dilution & Pre-clearing: Dilute Eluate 1 with 900 µL of ChIP Dilution Buffer. Pre-clear with 20 µL fresh beads for 1 hr at 4°C.
- IP: Add 5 µg of anti-H3K27me3 antibody (e.g., Cell Signaling Technology 9733) to the pre-cleared eluate. Rotate overnight at 4°C.
- Capture & Washing: Repeat capture and washing steps as in the first IP.
- Final Elution & Reverse Crosslinking: Elute with 100 µL of Standard Elution Buffer (0.1 M NaHCO3, 1% SDS). Add 5 µL of 5 M NaCl and 1 µL of Proteinase K (20 mg/mL). Incubate at 65°C for 4-6 hours (or overnight).
- DNA Purification: Purify DNA using a spin column-based PCR purification kit. Elute in 30 µL nuclease-free water. Analyze by qPCR at loci known to harbor bivalent marks.
Diagrams
Title: ChIP-reChIP Workflow for Bivalent Marks
Title: Challenges & Solutions in Chromatin Preservation
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Protocol |
|---|---|
| Diagenode Bioruptor Pico or Covaris S220 | Provides reproducible, controlled acoustic shearing to achieve target fragment size with minimal heat generation. |
| Magna ChIP Protein A/G Magnetic Beads | Enable rapid capture/washing with low mechanical stress, crucial for sequential IP steps. |
| Anti-H3K4me3 (Clone C42D8) | High-specificity rabbit monoclonal antibody for the first IP, minimizing non-specific background. |
| Anti-H3K27me3 (Clone C36B11) | Validated antibody for ChIP, targeting the repressive mark in the second IP. |
| Micrococcal Nuclease (MNase) | Alternative to sonication; digests linker DNA for precise nucleosome-level fragmentation. |
| Dual Crosslinker (DSG + Formaldehyde) | Enhances protein-protein crosslinking for complexes, improving co-IP efficiency for some targets. |
| PCR Purification Kit (Spin Column) | Efficiently recovers low-concentration DNA after decrosslinking and removes contaminants. |
| Protease Inhibitor Cocktail (EDTA-free) | Preserves histone modifications during lysis by inhibiting endogenous proteases. |
| RNAse A | Eliminates RNA contamination that can interfere with chromatin solubility and IP efficiency. |
| SPRIselect Beads | For automated, high-throughput size selection and purification of DNA after ChIP. |
Essential Positive and Negative Control Experiments for Rigor
Within the context of a broader thesis on ChIP-reChIP protocol for bivalent histone marks (e.g., H3K4me3 and H3K27me3), the inclusion of rigorous controls is non-negotiable. These controls validate specificity, minimize artifacts, and ensure data interpretability. This application note details the essential positive and negative control experiments required for robust bivalent domain research.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in ChIP-reChIP for Bivalent Marks |
|---|---|
| Specific Histone Modification Antibodies | Primary reagents for immunoprecipitation; must be highly validated for ChIP-seq grade specificity (e.g., anti-H3K4me3, anti-H3K27me3). |
| Isotype Control IgG | Negative control antibody matching the host species and Ig class of the primary antibody; identifies non-specific binding. |
| Pan-Histone H3 Antibody | Positive control antibody that pulls down all nucleosomes; assesses overall ChIP efficiency and chromatin input quality. |
| Protein A/G Magnetic Beads | For antibody immobilization and chromatin complex pulldown; preferred for sequential ChIP steps. |
| Crosslinking Reversal Buffer | Critical for reversing formaldehyde crosslinks after each IP step to elute chromatin for the subsequent round. |
| PCR Primers for Known Loci | Validate successful IP at control genomic regions (e.g., active gene promoter for H3K4me3, silenced gene for H3K27me3, bivalent locus like HOX genes). |
| Spike-in Control Chromatin | Exogenous chromatin (e.g., Drosophila, S. pombe) spiked into samples; normalizes for technical variation and allows quantitative comparisons. |
| RNase A and Proteinase K | Essential for complete digestion of RNA and proteins during crosslink reversal and DNA purification. |
| DNA Clean-up Beads/Columns | For efficient recovery of low-yield reChIP DNA for library preparation or qPCR. |
Essential Control Experiments: Protocols & Data
Antibody Specificity & Isotype Controls
Protocol: Perform parallel standard ChIP assays (not reChIP) for each antibody used.
- Positive Control Loci ChIP-qPCR: Using purified ChIP DNA, run qPCR for known genomic regions.
- H3K4me3: Amplify active gene promoters (e.g., GAPDH).
- H3K27me3: Amplify silenced gene promoters (e.g., MYT1 in pluripotent cells).
- Bivalent Loci: Amplify developmental regulator genes (e.g., HOXA1, SOX1).
- Isotype Control ChIP: Use a non-specific IgG under identical conditions. The signal should be near background.
- Input DNA: Use a sample of pre-IP, crosslink-reversed chromatin as a reference (1-10% of total).
Data Presentation (Representative qPCR Enrichment):
| Control Type | Target Locus | Expected Enrichment (Fold over IgG) | Acceptable Result |
|---|---|---|---|
| H3K4me3 Positive | GAPDH Promoter | >20-fold | Pass |
| H3K27me3 Positive | MYT1 Promoter | >15-fold | Pass |
| Bivalent Locus | HOXA1 Promoter | Intermediate (e.g., 5-15 fold for each mark) | Pass |
| Isotype (IgG) | Any locus | ~1-fold (Baseline) | Pass |
| Negative Genomic Region | Gene Desert | <2-fold | Pass |
Sequential ReChIP Efficiency Control
Protocol: This critical control tests the efficiency of the first elution and the feasibility of the second IP.
- Perform first ChIP with antibody for Mark A (e.g., H3K4me3).
- Divide the eluted material into two aliquots.
- Aliquot 1 (Test): Proceed with second IP for Mark B (H3K27me3).
- Aliquot 2 (Re-IP Control): Re-immunoprecipitate the same eluate with a fresh antibody for Mark A.
- Analyze both by qPCR at a known bivalent locus.
Interpretation: A strong signal in the Re-IP Control (Mark A -> Mark A) confirms successful elution and IP competency for the second step. The Test (Mark A -> Mark B) confirms co-existence.
Bead-Only & Crosslink Reversal Controls
Protocol:
- Bead-Only Control: Process a sample through the full ChIP-reChIP workflow with no antibody, only beads. Identifies chromatin that non-specifically binds to beads or plastic.
- Crosslink Reversal Efficiency: After the final reversal, run purified DNA on an agarose gel. Sheared chromatin should appear as a smear ~100-500 bp. High molecular weight indicates inefficient reversal.
Spike-in Normalization Control
Protocol:
- Add a fixed amount of exogenous chromatin (e.g., Drosophila S2 cells) to your fixed mammalian cells before sonication.
- Use species-specific antibodies for your target marks (they should not bind to spike-in chromatin).
- After sequencing, use aligned reads from the spike-in genome to normalize your experimental genome read counts. This controls for variations in cell number, IP efficiency, and library preparation.
Detailed Protocol: Core ChIP-reChIP for Bivalent Marks
Day 1: Crosslinking & Chromatin Preparation
- Crosslink cells with 1% formaldehyde for 10 min at RT. Quench with 125mM Glycine.
- Lyse cells, isolate nuclei. Resuspend in Sonication Buffer.
- Sonicate chromatin to ~200-500 bp fragments. Verify size by gel.
- Clarify lysate; aliquot. Add spike-in chromatin if using.
Day 2: First Immunoprecipitation
- Pre-clear chromatin with Protein A/G beads for 1h at 4°C.
- Incubate supernatant with target-specific antibody (e.g., anti-H3K4me3) or IgG control overnight at 4°C.
- Add pre-blocked beads, incubate 2h.
- Wash beads sequentially with: Low Salt, High Salt, LiCl, and TE buffers.
- Elute: Add 100µL of freshly prepared ChIP Elution Buffer (1% SDS, 0.1M NaHCO3). Incubate 30 min at 65°C with shaking. Collect supernatant.
Day 2/3: Crosslink Reversal & Second Immunoprecipitation
- Dilute the first eluate 10x with Dilution Buffer (1.1% Triton X-100, 1.2mM EDTA, 16.7mM Tris-HCl pH 8.1, 167mM NaCl).
- Repeat the IP process with the second antibody (e.g., anti-H3K27me3) or the re-IP control antibody.
- After final washes, perform on-bead crosslink reversal: Add Reverse Buffer (1% SDS, 0.1M NaHCO3) and incubate overnight at 65°C.
Day 3: DNA Purification
- Cool samples. Add RNase A (30 min, 37°C), then Proteinase K (2h, 55°C).
- Purify DNA using phenol-chloroform or silica columns. Elute in 20-30µL TE or nuclease-free water.
- Analyze by qPCR or prepare for sequencing.
Diagrams
ChIP-reChIP Workflow with Critical Controls
Logical Framework for Control Experiment Validation
Benchmarking ChIP-reChIP: Validation Strategies and Alternative Methods
Within the broader thesis on the ChIP-reChIP protocol for investigating bivalent histone marks, a critical challenge lies in the validation of results. The sequential immunoprecipitation, while powerful, can be technically demanding and prone to artifacts from antibody cross-reactivity or incomplete elution. This necessitates rigorous, independent confirmation using orthogonal methods to ensure the biological relevance of findings.
The table below summarizes key complementary techniques for validating Re-ChIP results, focusing on the co-localization of marks like H3K4me3 (active) and H3K27me3 (repressive) at bivalent promoters.
| Validation Technique | Primary Measurable Output | Key Advantages for Re-ChIP Validation | Typical Quantitative Correlation (Example Data) |
|---|---|---|---|
| Sequential Chromatin Immunoprecipitation (Re-ChIP) | Physical co-occupancy of two marks on the same chromatin fragment. | Direct evidence of bivalency on the same allele. | Baseline: % Input for dual-precipitated DNA. |
| Native Chromatin Immunoprecipitation (N-ChIP) with Sequential Elution | Sequential elution of histone modifications from the same chromatin sample. | Maintains native histone-DNA interactions; reduces formaldehyde crosslinking artifacts. | H3K4me3 elution fraction: 8-12% of total chromatin; Subsequent H3K27me3 elution from same beads: 2-5%. |
| Chromatin-Bound Fluorescent Reporter Assays | Transcriptional output via fluorescence (e.g., GFP) under control of a bivalent promoter. | Functional readout of bivalency resolution (activation vs. silencing) in single cells. | Reporter activation upon KDM6A (H3K27me3 demethylase) inhibition: 3- to 5-fold increase in GFP+ cells. |
| Proximity Ligation Assay (PLA) on Chromatin | Microscopic detection of two histone marks in close proximity (<40 nm). | Single-cell, imaging-based validation of spatial co-localization in nuclear context. | PLA signals per nucleus at validated bivalent loci: 15-25 vs. 0-2 at control loci. |
| MNase-based Chromatin Accessibility Profiling | Nucleosome positioning and accessibility at candidate bivalent regions. | Confirms "poised" state: presence of both marks coupled with nucleosome occupancy. | Bivalent promoters show protected fragments (~147 bp) but adjacent accessible sites (ATAC-seq peaks). |
Detailed Experimental Protocols
Protocol 1: Native ChIP (N-ChIP) with Sequential Salt Elution
This protocol validates Re-ChIP findings without crosslinking.
- Cell Lysis & Micrococcal Nuclease (MNase) Digestion: Harvest 1x10^7 cells. Lyse in hypotonic buffer (10 mM Tris-HCl pH 7.5, 10 mM KCl, 1.5 mM MgCl2, 0.34M sucrose, 10% glycerol, 0.1% Triton X-100, protease inhibitors). Pellet nuclei and digest with 20 U MNase for 5 min at 37°C to yield primarily mononucleosomes. Stop with 5 mM EDTA.
- Chromatin Extraction & Pre-clearing: Centrifuge, collect soluble chromatin supernatant in low-salt buffer (10 mM Tris-HCl pH 7.5, 10 mM KCl, 1.5 mM MgCl2). Pre-clear with 20 μL protein A/G beads for 1 hour at 4°C.
- First Immunoprecipitation (e.g., Anti-H3K4me3): Incubate pre-cleared chromatin with 5 μg of anti-H3K4me3 antibody overnight at 4°C. Add 50 μL protein A/G beads for 2 hours.
- Sequential Elution: Pellet beads.
- Elution 1 (Low Salt): Elute with 200 μL of 50 mM NaCl in TE buffer for 15 min at room temperature (RT). This elutes loosely bound material and some monovalent nucleosomes.
- Elution 2 (High Salt - Target Fraction): Elute the remaining chromatin specifically bound to H3K4me3 antibody with 200 μL of 500 mM NaCl in TE buffer for 15 min at RT. This is Fraction A (enriched for H3K4me3).
- Second Immunoprecipitation of Eluate: Take Fraction A (500 mM NaCl eluate). Dilute 10-fold with no-salt TE to reduce NaCl concentration to <70 mM. Use this as input for a second IP with anti-H3K27me3 antibody (5 μg) overnight. Proceed with standard bead capture and DNA elution (Fraction B).
- DNA Purification & Analysis: Reverse crosslinks (if any) for Fraction B by incubating with 200 mM NaCl at 65°C overnight. Treat with RNase A and Proteinase K. Purify DNA using phenol-chloroform extraction and analyze via qPCR at loci identified by Re-ChIP.
Protocol 2: Chromatin Proximity Ligation Assay (PLA)
This protocol visualizes spatial proximity of two histone marks in situ.
- Cell Fixation and Permeabilization: Grow cells on coverslips. Fix with 4% formaldehyde for 10 min at RT. Permeabilize with 0.5% Triton X-100 in PBS for 10 min.
- Blocking and Primary Antibody Incubation: Block with 3% BSA in PBS for 1 hour. Incubate with a pair of primary antibodies raised in different species (e.g., mouse anti-H3K4me3 and rabbit anti-H3K27me3) diluted in blocking buffer overnight at 4°C.
- PLA Probe Incubation and Ligation: Wash and incubate with species-specific PLA probes (MINUS and PLUS) for 1 hour at 37°C. Wash and add Ligation-Ligase solution for 30 min at 37°C to join probes if they are in close proximity (<40 nm).
- Amplification and Detection: Wash and add Amplification-Polymerase solution with fluorescently labeled nucleotides for 100 min at 37°C. This generates a rolling-circle amplification product detectable as a discrete fluorescent spot.
- Imaging and Analysis: Wash, mount with DAPI-containing medium. Image using a fluorescence microscope. Count PLA signals (distinct red dots) per nucleus using image analysis software (e.g., ImageJ). Compare signals in experimental cells versus negative controls (omitting one primary antibody).
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function/Application |
|---|---|
| Magnetic Protein A/G Beads | Solid support for antibody-chromatin complex capture; enable efficient sequential elution steps. |
| Micrococcal Nuclease (MNase) | Digests linker DNA to generate mononucleosomes for N-ChIP or accessibility assays. |
| DUOlink PLA Kit (or equivalent) | Complete reagent set for performing Proximity Ligation Assays, including probes, ligase, and amplification mixes. |
| Crosslinking Reversible Chromatin Extraction Kit | Standardized reagents for efficient chromatin shearing and extraction after formaldehyde crosslinking. |
| Validated Antibody Pair for PLA | Antibodies against target histone marks (e.g., H3K4me3, H3K27me3) raised in different host species and validated for PLA. |
| Epigenetic Modulator Inhibitors/Agonists | Small molecules (e.g., GSK-J4 for KDM6A/B inhibition, DZNep for EZH2 inhibition) to functionally perturb bivalent marks. |
| Bivalent Promoter-Controlled GFP Reporter Construct | Plasmid containing a validated bivalent promoter driving a fluorescent reporter for live-cell functional assays. |
Visualization Diagrams
Title: Validation Strategy for Re-ChIP Results
Title: Sequential ChIP (Re-ChIP) Core Workflow
Title: Bivalent Promoter Resolution Pathways
Comparison to Single ChIP Followed by Sequential Immunoblot (ChIP-Western)
This application note is framed within a thesis investigating bivalent histone marks (e.g., co-existing H3K4me3 and H3K27me3) using Chromatin Immunoprecipitation followed by Re-ChIP (ChIP-reChIP). A critical methodological consideration is how ChIP-reChIP compares to the alternative approach of performing a single ChIP, followed by sequential immunoblotting (ChIP-Western) of the isolated chromatin complexes. This document provides a detailed comparison, protocols, and analytical tools to guide researchers in selecting the optimal method for studying co-existing epigenetic marks or transcription factor complexes.
Comparative Analysis: ChIP-reChIP vs. ChIP-Western
Table 1: Methodological and Performance Comparison
| Feature | Chromatin Immunoprecipitation Re-ChIP (ChIP-reChIP) | Single ChIP Followed by Sequential Immunoblot (ChIP-Western) |
|---|---|---|
| Core Principle | Two sequential, solution-phase ChIPs with different antibodies. | One ChIP, followed by protein/DNA denaturation and Western blot for multiple antigens. |
| Primary Output | DNA bound by both antigens simultaneously. Identifies cis co-localization. | Protein components (histone marks) present in the pool of ChIP-isolated complexes. Semi-quantitative protein detection. |
| Spatial Resolution | High. Confirms two marks exist on the same chromatin fragment (in cis). | Low. Confirms both marks are present in the immunoprecipitated population, but not necessarily on the same fragment (could be in trans). |
| Throughput | Lower. Two full ChIP cycles required per sample. | Higher. One ChIP, multiple blots from the same sample. |
| Material Required | High (requires sufficient chromatin for 2nd IP). | Moderate. |
| Key Challenge | Stringency of elution between rounds; antibody compatibility. | Complete dissociation of chromatin proteins for accurate immunoblotting. |
| Optimal Use Case | Proving direct bivalency on identical DNA sequences. | Screening for the presence of multiple candidate proteins/marks in a ChIP-enriched population. |
Table 2: Representative Quantitative Data from Bivalent Mark Analysis
| Experiment | Method | Target Locus (e.g., Promoter) | Input % for H3K4me3 | Input % for H3K27me3 | Input % for Dual (Bivalent) | Conclusion |
|---|---|---|---|---|---|---|
| 1 | Single ChIP-qPCR | Developmental Gene A | 2.5% | 1.8% | N/A | Both marks enriched, but spatial relationship unknown. |
| 2 | ChIP-reChIP-qPCR | Developmental Gene A | N/A | N/A | 0.15% | Confirms ~6-8% of H3K4me3-positive fragments also bear H3K27me3 at this locus. |
| 3 | ChIP-Western (Blot) | Genome-wide/General | N/A | Strong H3K4me3 signal | Strong H3K27me3 signal | Validates both marks are present in the ChIP'ed chromatin pool. |
Detailed Experimental Protocols
Protocol A: Native ChIP-reChIP for Bivalent Histone Marks
Day 1: Chromatin Preparation & First ChIP
- Crosslink & Harvest: Harvest ~10^7 cells. For native histone ChIP, omit formaldehyde crosslinking. Lyse cells in Buffer A (10 mM HEPES pH 7.9, 10 mM KCl, 1.5 mM MgCl2, 0.34 M Sucrose, 10% Glycerol, 1 mM DTT, protease inhibitors).
- Micrococcal Nuclease (MNase) Digestion: Pellet nuclei, resuspend in MNase Digestion Buffer. Digest with 0.5-2 µL MNase (20 U/µL) for 5-15 min at 37°C to yield predominantly mononucleosomes. Stop with 10 mM EDTA.
- Chromatin Solubilization: Lyse nuclei in ChIP Lysis Buffer (1% SDS, 10 mM EDTA, 50 mM Tris-HCl pH 8.1) + inhibitors. Sonicate briefly to break nuclear membranes. Centrifuge; supernatant is soluble chromatin. Dilute 10-fold in ChIP Dilution Buffer.
- First Immunoprecipitation: Pre-clear chromatin with protein A/G beads for 1 hr. Incubate 50-100 µg chromatin with 2-5 µg of the first antibody (e.g., anti-H3K4me3) overnight at 4°C.
- Bead Capture: Add 50 µL pre-blocked protein A/G beads for 2 hrs. Wash sequentially: Low Salt Wash Buffer, High Salt Wash Buffer, LiCl Wash Buffer, and TE Buffer.
Day 2: Elution, Second ChIP, and Clean-up
- Elution from Beads: Perform two 15-min elutions with 250 µL Re-ChIP Elution Buffer (1% SDS, 100 mM NaHCO3). Combine eluates.
- Dilution and Second IP: Dilute the eluate 1:50 in ChIP Dilution Buffer (to reduce SDS to ~0.02%). Add 2-5 µg of the second antibody (e.g., anti-H3K27me3). Incubate overnight at 4°C.
- Second Bead Capture & Washes: Add 30 µL fresh beads for 2 hrs. Repeat wash series as in Step 5.
- Final Elution & Reverse Crosslinks: Elute beads twice with 100 µL direct Elution Buffer (1% SDS, 100 mM NaHCO3). For native protocol, add 5 M NaCl to 200 mM and RNase A. Incubate at 65°C for 4-6 hrs.
- DNA Purification: Add Proteinase K, incubate at 45°C for 2 hrs. Purify DNA via phenol-chloroform extraction or spin columns. Analyze by qPCR or sequencing.
Protocol B: ChIP-Western Follow-up Assay
Part 1: Standard Chromatin Immunoprecipitation
- Perform a standard ChIP protocol (as in Protocol A, Steps 1-5) using an antibody against the primary target of interest (e.g., anti-H3K4me3) or a control (e.g., anti-IgG).
- After final TE wash, instead of eluting for DNA, proceed to protein elution.
Part 2: On-Bead Protein Elution and Immunoblot
- Denaturing Elution: Resuspend the washed beads in 40 µL 1.5X Laemmli SDS-PAGE Loading Buffer (with 5% β-mercaptoethanol). Boil at 95-100°C for 10 minutes to elute and denature proteins.
- Spin Separation: Briefly centrifuge (13,000 rpm, 1 min). Carefully load the supernatant (eluted proteins) onto an SDS-PAGE gel. Caution: Avoid carrying over beads.
- Western Blot: Perform standard SDS-PAGE and wet transfer to a PVDF membrane.
- Sequential Immunoblotting: Block membrane, then probe first with an antibody for a co-existing mark (e.g., anti-H3K27me3). Develop signal. Strip the membrane thoroughly (e.g., with mild stripping buffer), and re-probe for the ChIP-targeted mark (e.g., H3K4me3) to confirm IP efficiency. Include inputs and IgG controls.
Visualizations
Title: ChIP-reChIP vs. ChIP-Western Workflow Comparison
Title: Spatial Resolution Difference Between Methods
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for ChIP-reChIP & ChIP-Western
| Item | Function & Specification | Example Vendor/Product |
|---|---|---|
| Micrococcal Nuclease (MNase) | Digests chromatin to mononucleosomes for high-resolution IP. Must be titrated. | Worthington Biochemical, NEB |
| ChIP-Grade Antibodies | High specificity and affinity for target epitopes (histone marks). Critical for success. | Cell Signaling Tech, Abcam, Active Motif |
| Protein A/G Magnetic Beads | For efficient capture and washing. Magnetic separation minimizes background. | Pierce, Diagenode |
| Re-ChIP Elution Buffer | Mild elution buffer (e.g., 1% SDS, 100mM NaHCO3) to dissociate first Ab without denaturing chromatin structure. | Home-made formulation required. |
| DNA Clean-up Columns | For efficient purification of low-yield DNA after reChIP for downstream qPCR. | Zymo Research, Qiagen |
| Mild Stripping Buffer | For ChIP-Western: removes antibodies from blot without damaging antigen for re-probing. | Thermo Fisher Restore PLUS |
| Protease/Phosphatase Inhibitors | Essential in all buffers for native ChIP to preserve post-translational modifications. | Roche cOmplete, PhosSTOP |
| qPCR Master Mix | Sensitive SYBR Green mix for quantifying low-abundance ChIP-reChIP DNA. | Bio-Rad, Thermo Fisher PowerUp |
Comparison to Proximity Ligation Assays (PLA) and Other In Situ Methods
In the context of ChIP-reChIP protocols for investigating bivalent histone marks (e.g., H3K4me3 and H3K27me3 co-occupancy), it is critical to understand the landscape of in situ detection technologies. While ChIP-reChIP provides biochemical evidence of co-localization on purified chromatin, methods like Proximity Ligation Assay (PLA) offer spatial resolution within intact nuclei. This Application Note compares these methodologies, detailing their respective protocols and applications.
Comparative Analysis of In Situ Methods for Chromatin Profiling
Table 1: Quantitative Comparison of Key In Situ Chromatin Analysis Methods
| Method | Spatial Resolution | Detection Limit (Approx. Target Copies) | Typical Assay Time | Key Advantage for Bivalent Mark Studies | Primary Limitation |
|---|---|---|---|---|---|
| ChIP-reChIP | Population-level (bulk) | ~1000 cells (native) | 2-3 days | Direct biochemical proof of co-occupancy on same chromatin fragment | Loses cellular and subnuclear context. |
| Proximity Ligation Assay (PLA) | Single-cell / Nuclear | 10-40 molecules | 1-2 days | Visualizes co-localization in fixed cells/tissues; single-cell quantitation possible. | Proximity ≠ direct co-occupancy; can be influenced by antibody geometry. |
| Immunofluorescence (IF) | Single-cell / Nuclear | >100 molecules | 1 day | Simple, visual co-localization of marks via standard fluorescence. | Low resolution; co-localization is qualitative and prone to false positives. |
| Immuno-FISH | Single-cell / Nuclear | 1-2 DNA loci | 2-3 days | Correlates histone mark presence with specific genomic loci. | Technically challenging; low throughput for genome-wide studies. |
| CUT&Tag / CUT&Run | Population-level (bulk/single-cell) | ~100 cells | 1-2 days | High signal-to-noise from intact nuclei; works with low cell numbers. | Does not directly prove bivalency on the same nucleosome. |
Detailed Experimental Protocols
Protocol 1: In Situ Proximity Ligation Assay (PLA) for Bivalent Histone Marks
Objective: To detect spatial proximity of H3K4me3 and H3K27me3 marks within intact, fixed cells.
Materials & Reagents:
- Cultured cells grown on chambered slides.
- Fixative: 4% Paraformaldehyde (PFA) in PBS.
- Permeabilization Buffer: 0.5% Triton X-100 in PBS.
- Blocking Solution: 3% BSA, 0.1% Tween-20 in PBS.
- Primary Antibodies: High-validity rabbit anti-H3K4me3 and mouse anti-H3K27me3.
- PLA Probe Solution: Species-specific PLUS and MINUS oligonucleotide-conjugated secondary antibodies (e.g., Duolink).
- Ligation Solution: Containing connector oligonucleotides and ligase.
- Amplification Solution: Containing fluorescently labeled nucleotides and polymerase.
- Mounting Medium with DAPI.
Procedure:
- Fixation & Permeabilization: Wash cells with PBS. Fix with 4% PFA for 15 min at RT. Wash 2x with PBS. Permeabilize with 0.5% Triton X-100 for 10 min.
- Blocking: Incubate with Blocking Solution for 1 hour at 37°C in a humidified chamber.
- Primary Antibody Incubation: Dilute antibodies in Blocking Solution. Apply mixture to sample. Incubate overnight at 4°C. Wash 3x with Wash Buffer A (provided in PLA kit).
- PLA Probe Incubation: Apply PLA Probe Solution (anti-rabbit PLUS, anti-mouse MINUS). Incubate for 1 hour at 37°C. Wash 2x with Wash Buffer A.
- Ligation: Apply Ligation Solution. Incubate for 30 min at 37°C. Wash 2x with Wash Buffer A.
- Amplification: Apply Amplification Solution. Incubate for 100 min at 37°C in the dark. Wash 2x with Wash Buffer B (provided in kit).
- Mounting & Imaging: Briefly wash in 0.01x Wash Buffer B. Air dry and mount with DAPI-containing medium. Image using a fluorescence microscope equipped with appropriate filters. PLA signals appear as distinct fluorescent dots.
Protocol 2: Sequential Chromatin Immunoprecipitation (ChIP-reChIP) Core Workflow
Objective: To sequentially immunoprecipitate the same chromatin sample to isolate nucleosomes carrying both H3K4me3 and H3K27me3 marks.
Materials & Reagents:
- Crosslinked Chromatin (sonicated to ~200-500 bp).
- Magnetic Protein A/G Beads.
- High-Specificity Antibodies for 1st IP (e.g., anti-H3K4me3) and 2nd IP (e.g., anti-H3K27me3).
- Low pH Elution Buffer (e.g., 0.1 M NaHCO3, 1% SDS).
- Crosslink Reversal Buffer: 200 mM NaCl, 10 µg/µL Proteinase K.
- DNA Purification Columns/PCR cleanup kit.
Procedure:
- First Immunoprecipitation: Pre-clear chromatin with beads. Incubate chromatin with the first primary antibody (e.g., anti-H3K4me3) overnight at 4°C. Add beads, incubate, and wash extensively.
- Elution from First IP: Elute the immune complexes from the first beads using a gentle, low-pH elution buffer.
- Second Immunoprecipitation: Dilute the eluate and adjust buffer conditions. Incubate with the second primary antibody (e.g., anti-H3K27me3) overnight. Add fresh beads, incubate, and wash.
- Final Elution and DNA Recovery: Elute complexes from the second beads. Add crosslink reversal buffer and incubate at 65°C overnight. Purify DNA using a silica-membrane column.
- Analysis: Analyze purified DNA by qPCR (for candidate loci) or next-generation sequencing (for genome-wide profiling).
Visualization of Methodologies and Context
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for In Situ Chromatin Analysis Experiments
| Item | Function in Experiment | Example Product/Catalog Number (for reference) |
|---|---|---|
| High-Validity, ChIP/IF-Grade Antibodies | Critical for specificity in both PLA and ChIP-reChIP. Non-specific antibodies cause high background or false positives. | Anti-H3K4me3 (Rabbit mAb, CST #9751), Anti-H3K27me3 (Mouse mAb, Active Motif #61017) |
| In Situ Proximity Ligation Assay Kit | Provides all optimized buffers, enzymes, and oligonucleotide-conjugated secondaries for PLA detection. | Duolink PLA Fluorescence Kit (Sigma-Aldrich) |
| Magnetic Protein A/G Beads | Solid support for antibody-chromatin complex isolation in ChIP-reChIP; low non-specific binding is essential. | Dynabeads Protein A/G (Invitrogen) |
| Crosslinking Reagent | Stabilizes protein-DNA and protein-protein interactions for ChIP. | Formaldehyde (37%), Disuccinimidyl glutarate (DSG) |
| Chromatin Shearing System | Generates optimal fragment size (200-500 bp) for ChIP. Critical for reChIP efficiency. | Covaris S220 sonicator, Bioruptor Pico |
| DNA Cleanup/Purification Columns | For reliable recovery of low-abundance DNA after ChIP-reChIP elution. | MinElute PCR Purification Kit (Qiagen) |
| Fluorescence Mounting Medium with DAPI | Preserves fluorescence signal and counterstains nuclei for PLA/IF imaging. | ProLong Gold Antifade Mountant with DAPI (Invitrogen) |
| Cell Culture Chamber Slides | Provides a growth surface compatible with high-resolution microscopy for PLA. | Nunc Lab-Tek II Chamber Slide System |
Advantages and Limitations vs. Computational Co-localization from Single ChIP-seq Datasets
This application note is framed within a broader thesis investigating bivalent chromatin domains (e.g., co-existing H3K4me3 and H3K27me3 marks) using the orthogonal ChIP-reChIP wet-lab protocol. A critical point of comparison is the widely used computational approach of "co-localization" derived from analyzing two separate, standard ChIP-seq datasets. This document details the advantages, limitations, and specific protocols for computational co-localization, providing a benchmark for evaluating direct physical co-occupancy evidence from ChIP-reChIP experiments in bivalent mark research.
Comparative Analysis: Computational Co-localization vs. ChIP-reChIP
Table 1: Advantages and Limitations of Computational Co-localization from Single ChIP-seq Datasets
| Aspect | Advantages | Limitations & Critical Considerations |
|---|---|---|
| Material & Input | Utilizes existing single ChIP-seq datasets; no additional biological sample required. | Requires high-quality, deeply sequenced replicates for each mark from the same biological sample. Sample heterogeneity or technical batch effects confound analysis. |
| Resolution & Evidence | Can suggest broad domain overlap (e.g., across promoter regions). Provides genome-wide landscape. | Infers, but does not prove, physical co-localization on the same allele or nucleosome ("in cis"). Cannot distinguish true bivalency from mixed cell populations. |
| Technical Demand | Computational only; less technically demanding than ChIP-reChIP in the lab. | Demanding bioinformatics expertise for rigorous peak calling, normalization, and intersection. |
| Quantitative Output | Can estimate prevalence of putative bivalent domains genome-wide. Provides metrics like overlap percentage. | Signal quantification is indirect. Highly sensitive to peak-calling thresholds and normalization methods between different antibodies/experiments. |
| Thesis Relevance | Excellent for hypothesis generation, identifying candidate bivalent regions for targeted ChIP-reChIP validation. | Provides correlative evidence; the thesis requires direct evidence of bivalency, which only ChIP-reChIP or similar single-molecule methods can furnish. |
Table 2: Key Quantitative Metrics from Computational Co-localization Studies
| Metric | Typical Range/Value | Interpretation & Caveat |
|---|---|---|
| Genomic Overlap | e.g., ~20-30% of H3K4me3 peaks may overlap H3K27me3 peaks in ESCs. | Highly dependent on cell type, peak-calling stringency (p-value, FDR), and definition of "overlap" (peak summit distance vs. interval overlap). |
| Peak Signal Correlation | Spearman correlation of read counts in overlapping regions can be low or negative. | Lack of positive correlation is expected for antagonistic marks but underscores the challenge of normalizing two distinct ChIP datasets. |
| Enrichment at Genomic Features | Putative bivalent domains are predominantly at developmental gene promoters in pluripotent cells. | Confirms known biology but does not validate the simultaneous presence of both marks on the same chromatin template. |
Detailed Protocol: Computational Co-localization Analysis
Protocol 1: Processing and Peak Calling for Individual ChIP-seq Datasets Objective: Generate high-confidence peak sets for each histone mark (e.g., H3K4me3, H3K27me3) from aligned sequencing files (BAM format).
- Quality Control: Use
fastqcandmultiQCon raw reads. Assess library complexity with tools likepreseq. - Alignment & Filtering: Align to reference genome (e.g., GRCh38) using
bowtie2orBWA. Remove duplicates and low-quality reads usingsamtools. - Peak Calling:
- For narrow marks (H3K4me3): Use
MACS2(macs2 callpeak -t ChIP.bam -c Input.bam -f BAM -g hs -n output --call-summits). - For broad marks (H3K27me3): Use
MACS2in broad mode (--broad) orSICER2.
- For narrow marks (H3K4me3): Use
- Irreproducible Discovery Rate (IDR) Analysis: For replicates, use IDR pipeline to generate a conservative, reproducible set of peaks. This is critical for downstream overlap analysis.
Protocol 2: Identifying Overlapping Genomic Regions Objective: Define a set of genomic intervals where peaks from two marks intersect.
- File Preparation: Use IDR-filtered peak files in BED or narrowPeak format.
- Define Overlap Criteria: A common criterion is requiring a minimum reciprocal overlap (e.g., ≥1 bp). More stringent criteria use peak summit distance (e.g., ≤500 bp).
- Intersection: Use
bedtools intersect(e.g.,bedtools intersect -a Mark1_peaks.bed -b Mark2_peaks.bed -wa -u > putative_bivalent_regions.bed). - Annotation: Annotate overlapping regions with genomic features (promoters, enhancers) using tools like
ChIPseekerin R orHOMER.
Protocol 3: Visualization and Validation Objective: Visualize read density and validate putative bivalent regions.
- Generate Coverage Tracks: Create bigWig files for visualization using
deeptools bamCoverage(normalized to CPM or RPGC). - Integrative Genomics Viewer (IGV): Manually inspect specific loci (e.g., promoters of PAX6, SOX1) for co-occurring signal patterns.
- Meta-gene Profile & Heatmaps: Use
deeptools computeMatrixandplotProfile/plotHeatmapto show average signal of both marks across putative bivalent regions.
Diagrams
Title: Computational Co-localization Analysis Workflow
Title: Key Limitation: Population Mixture vs. Direct Evidence
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Resources for Computational Co-localization Analysis
| Item | Function & Relevance | Example/Note |
|---|---|---|
| High-Quality ChIP-seq Datasets | Foundational input data. Must be from isogenic cell samples with appropriate controls. | Use public repositories (GEO, ENCODE) or generate in-house. Critical: Include matched input/IgG controls. |
| Reference Genome & Annotation | Essential for alignment and peak annotation. | GRCh38/hg38 from UCSC/ENSEMBL. GTF/GFF3 annotation files for gene features. |
| Peak Calling Software | Identifies statistically enriched regions from aligned reads. | MACS2 (standard), SICER2 (broad domains), SEACR (sparse data). |
| IDR Pipeline | Statistically evaluates replicate concordance to generate robust peak sets. | ENCODE ChIP-seq pipeline standard. Prevents false positives from single replicates. |
| Genomic Interval Tools | Performs overlap, merge, and subset operations on peak files. | BEDTools, BEDOPS. Core utilities for intersection analysis. |
| Visualization Suite | Enables inspection and presentation of co-localization signals. | Integrative Genomics Viewer (IGV) (local), deeptools (meta-profiles/heatmaps), UCSC Genome Browser. |
| Programming Environment | Provides flexibility for custom analysis pipelines and statistical tests. | R/Bioconductor (ChIPseeker, GenomicRanges), Python (pybedtools, pandas). |
Within the broader thesis on ChIP-reChIP protocols for investigating bivalent histone marks (e.g., co-existing H3K4me3 and H3K27me3), the limitations of sequential chromatin immunoprecipitation (reChIP) are apparent, including high background, significant material loss, and protocol stringency. CUT&RUN and CUT&Tag have emerged as superior alternatives for in situ profiling of histone modifications and transcription factors. This application note evaluates their adaptation for sequential, multi-target assays, providing a direct comparison to traditional reChIP and detailing optimized protocols for bivalent mark detection.
Comparative Analysis: reChIP vs. CUT&RUN/Tag Sequential Assays
Table 1: Quantitative Comparison of Sequential Assay Methodologies
| Parameter | ChIP-reChIP | Sequential CUT&RUN | Sequential CUT&Tag |
|---|---|---|---|
| Typical Starting Cells | 1x10^6 - 1x10^7 | 5x10^4 - 1x10^5 | 5x10^3 - 5x10^4 |
| Hands-on Time | 3-4 days | 1-2 days | 1 day |
| Background Signal | High (due to cross-linking & sonication) | Very Low | Extremely Low |
| Resolution | ~200-500 bp | ~10-50 bp (near cleavage site) | ~10-50 bp (near insertion site) |
| Sequential Efficiency | Low (~1-10% of 1st IP material recovered) | Moderate-High (Re-permeabilization required) | High (Consecutive tagmentation in situ) |
| Compatibility with Bivalent Marks | Challenging due to harsh elution conditions | High (gentle cleavage & release) | Very High (sequential tagmentation) |
| Recommended Sequencing Depth | 20-50 million reads per sample | 5-15 million reads per sample | 5-10 million reads per sample |
Detailed Protocols
Sequential CUT&Tag for Bivalent Histone Marks
Objective: To sequentially map H3K4me3 (activating) and H3K27me3 (repressive) marks on the same population of nuclei.
Research Reagent Solutions & Essential Materials:
| Item | Function / Explanation |
|---|---|
| Concanavalin A-coated Beads | Binds glycosylated cell/nuclear membranes, immobilizing samples for washes and reactions. |
| Primary Antibody (1st) | High-specificity rabbit anti-H3K4me3. Validated for CUT&Tag (e.g., CST #9751). |
| Primary Antibody (2nd) | High-specificity mouse anti-H3K27me3. Validated for CUT&Tag (e.g., CST #9733). |
| pA-Tn5 Fusion Protein | Protein A-Tn5 transposase pre-loaded with sequencing adapters. Binds 1st primary antibody. |
| Guinea Pig Anti-Rabbit IgG | Secondary antibody used to block residual protein A after 1st tagmentation. |
| pG-Tn5 Fusion Protein | Protein G-Tn5 transposase pre-loaded with different adapters. Binds 2nd primary antibody. |
| Digitonin Permeabilization Buffer | Contains digitonin to permeabilize nuclear membranes for antibody and Tn5 entry. |
| Tagmentation Buffer | Provides optimal ionic conditions for Tn5 transposase activity. |
| MgCl₂ Solution | Divalent cation required to activate Tn5 transposase for DNA cutting/adaptor ligation. |
| DNA Extraction Reagents | Phenol:Chloroform:Isoamyl Alcohol, Glycogen, Ethanol for clean DNA purification. |
Protocol:
- Cell Preparation: Harvest 50,000 cells, wash with PBS, and bind to Concanavalin A beads.
- Permeabilization: Incubate bead-bound cells in Digitonin Buffer (0.05% digitonin) for 10 min on ice. Wash.
- First Immunoincubation: Incubate with rabbit anti-H3K4me3 (1:100) in Antibody Buffer overnight at 4°C.
- First Tagmentation: Wash cells. Incubate with pA-Tn5 (1:100) for 1 hour at room temp (RT). Wash.
- Activation & Blocking: Add pre-warmed Tagmentation Buffer with MgCl₂. Incubate at 37°C for 1 hour. Immediately quench with 10mM EDTA. Wash.
- Secondary Antibody Block: Incubate with Guinea Pig Anti-Rabbit IgG (1:100) for 30 min at RT to block residual protein A epitopes.
- Second Immunoincubation: Without washing, add mouse anti-H3K27me3 (1:100). Incubate for 2 hours at RT.
- Second Tagmentation: Wash cells. Incubate with pG-Tn5 (loaded with distinct adapters) for 1 hour at RT.
- Activation & DNA Extraction: Add MgCl₂-containing buffer, incubate at 37°C for 1 hour. Quench with EDTA and SDS. Extract DNA using Phenol:Chloroform.
- Library Amplification: Amplify with i5 and i7 index primers for 12-15 cycles. Purify for sequencing.
Sequential CUT&RUN for Bivalent Histone Marks
Objective: To perform consecutive cleavage and release of chromatin bound by two distinct histone modifications.
Protocol (Key Modifications from Standard):
- Cell Binding & Permeabilization: As per CUT&Tag (Steps 1-2 above).
- First Immunoincubation: Incubate with primary antibody (e.g., anti-H3K4me3) overnight at 4°C.
- First Cleavage: Add pA-Micrococcal Nuclease (pA-MNase) at 1:100 dilution. Incubate on ice for 1 hour. Wash.
- First Release: Pre-chill tubes on ice. Add low-Ca²⁺ activation buffer (2mM CaCl₂). Incubate on ice for 30-60 min. This induces sparse, targeted cleavage. Immediately quench with EGTA-STOP Buffer (10mM EGTA). Collect supernatant (S1) containing first chromatin fragments.
- Membrane Re-permeabilization: To the bead-bound nuclei, add fresh Digitonin Buffer for 5 min to re-establish permeability for the second round.
- Second Immunoincubation & Cleavage: Repeat steps 2-4 with the second primary antibody (e.g., anti-H3K27me3). Collect supernatant (S2).
- DNA Purification & Processing: Pool S1 and S2 (or process separately). Add RNase A and Proteinase K. Reverse crosslinks at 65°C. Purify DNA via spin columns.
- Library Preparation: Use standard Illumina library prep kits for low-input DNA.
Visualizations
Title: Sequential CUT&RUN and CUT&Tag Workflow for Bivalent Marks
Title: Traditional ChIP-reChIP Workflow and Limitations
This document serves as an Application Note within a broader thesis investigating the role of bivalent chromatin domains, characterized by the simultaneous presence of antagonistic histone modifications (e.g., H3K4me3 and H3K27me3), in gene regulation and cellular plasticity. The ChIP-reChIP (Chromatin Immunoprecipitation followed by a second ChIP) protocol is a critical, yet resource-intensive, tool for directly demonstrating the co-localization of two distinct epigenetic marks on the same nucleosome strand. This analysis provides a structured cost-benefit and throughput framework to guide researchers in deciding when the unique advantages of ChIP-reChIP justify its implementation over alternative, less direct methodologies.
Comparative Analysis of ChIP Methodologies
Table 1: Quantitative Comparison of ChIP-based Methods for Studying Histone Co-localization
| Parameter | Sequential ChIP (ChIP-reChIP) | Parallel Single ChIPs | ChIP-Seq with Sequential Antibody Elution | ChIP-Bisulfite Sequencing (ChIP-BS-seq) |
|---|---|---|---|---|
| Primary Goal | Prove direct co-localization on the same DNA fragment. | Suggest overlap from different experiments. | Map two marks serially from one sample. | Correlate mark presence with DNA methylation. |
| Throughput (Samples/Week) | Low (4-8) | High (16-32) | Medium (8-12) | Low (4-8) |
| Material Cost per Sample | Very High (~2.5x standard ChIP) | Low (Baseline) | Medium-High (~1.8x standard ChIP) | Very High |
| Technical Difficulty | High | Low | Medium-High | Very High |
| Key Quantitative Output | % of DNA from 1st ChIP recovered in 2nd ChIP. | Overlapping peaks from separate datasets. | Two datasets from one biological sample. | Methylation status at mark localization sites. |
| Risk of False Positives (for co-localization) | Very Low | High (from population averaging) | Low | Not Applicable |
| Ideal Application | Definitive proof of bivalency; low-abundance complexes. | Genome-wide screening for potential co-localization. | When sample material is extremely limited. | Interrogating epigenetic crosstalk mechanisms. |
Detailed ChIP-reChIP Protocol for Bivalent Marks
Protocol: Sequential Chromatin Immunoprecipitation for H3K4me3 & H3K27me3
I. Chromatin Preparation and First Immunoprecipitation
- Cell Fixation & Lysis: Crosslink cells with 1% formaldehyde for 10 min at RT. Quench with 125mM Glycine. Wash cells in cold PBS. Lyse cells in LB1 Buffer (50mM HEPES-KOH pH7.5, 140mM NaCl, 1mM EDTA, 10% Glycerol, 0.5% NP-40, 0.25% Triton X-100) for 10min at 4°C. Pellet nuclei.
- Nuclear Lysis & Sonication: Lyse nuclei in LB2 Buffer (10mM Tris-HCl pH8.0, 200mM NaCl, 1mM EDTA, 0.5mM EGTA) for 10min at 4°C. Pellet and resuspend in Sonication Buffer (0.1% SDS, 1mM EDTA, 10mM Tris-HCl pH8.0). Sonicate chromatin to 200-500 bp fragments. Clear lysate by centrifugation.
- First IP (e.g., anti-H3K4me3): Pre-clear chromatin with Protein A/G beads for 1h. Incubate supernatant with 2-5µg of primary antibody overnight at 4°C. Add pre-blocked Protein A/G beads and incubate for 2h.
- Bead Washing: Wash beads sequentially for 5min each: Low Salt Wash Buffer (0.1% SDS, 1% Triton X-100, 2mM EDTA, 20mM Tris-HCl pH8.0, 150mM NaCl), High Salt Wash Buffer (0.1% SDS, 1% Triton X-100, 2mM EDTA, 20mM Tris-HCl pH8.0, 500mM NaCl), LiCl Wash Buffer (0.25M LiCl, 1% NP-40, 1% Deoxycholate, 1mM EDTA, 10mM Tris-HCl pH8.0). Perform two final washes with TE Buffer.
II. Elution and Second Immunoprecipitation
- Elution from First Antibody: Elute chromatin from the first antibody complex by incubating beads in ReChIP Elution Buffer (10mM DTT, 1% SDS, 0.1M NaHCO3) for 30min at 37°C with agitation. Collect supernatant.
- Dilution and Re-Immunoprecipitation: Dilute the eluate 1:40 in ReChIP Dilution Buffer (1% Triton X-100, 2mM EDTA, 150mM NaCl, 20mM Tris-HCl pH8.0). Split the sample if performing multiple second IPs. Add the second primary antibody (e.g., anti-H3K27me3) and incubate overnight at 4°C.
- Second IP Capture & Washing: Add pre-blocked Protein A/G beads for 2h. Repeat the wash series as in Step 4.
III. DNA Recovery and Analysis
- Final Elution & Decrosslinking: Elute DNA from the second-round beads in Standard ChIP Elution Buffer (1% SDS, 0.1M NaHCO3). Add NaCl to 200mM and reverse crosslinks at 65°C overnight.
- DNA Purification: Treat with RNase A and Proteinase K. Purify DNA using phenol-chloroform extraction or silica-membrane columns.
- Analysis: Analyze purified DNA by qPCR (for candidate loci) or prepare libraries for next-generation sequencing (ChIP-reChIP-seq).
Visualization of Method Decision and Workflow
Decision Workflow for ChIP Method Selection
ChIP-reChIP Experimental Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for ChIP-reChIP Experiments
| Reagent/Material | Function & Role in Protocol | Critical Consideration for Bivalent Marks |
|---|---|---|
| Crosslinking Agent (Formaldehyde) | Fixes protein-DNA and protein-protein interactions. | Over-fixation can mask epitopes; 10 min standard. |
| Validated ChIP-Grade Antibodies | Specifically immunoprecipitate target histone modifications. | Most critical factor. Must be validated for sequential IP. High specificity for H3K4me3 vs. H3K27me3 is essential. |
| Magnetic Protein A/G Beads | Solid support for antibody-antigen complex capture. | Low non-specific binding is crucial for clean 2nd IP. |
| DTT (Dithiothreitol) | Reducing agent in ReChIP Elution Buffer. | Breaks the antibody-antigen bond of the 1st IP by reducing disulfide bridges, freeing chromatin for 2nd IP. |
| SDS (Sodium Dodecyl Sulfate) | Denaturing detergent in ReChIP Elution Buffer. | Helps dissociate complexes but must be sufficiently diluted (to ~0.025%) before 2nd IP to allow antibody binding. |
| Protease & RNase Inhibitors | Preserve chromatin integrity during preparation. | Essential for maintaining nucleosome structure throughout the lengthy protocol. |
| Silica-membrane DNA Clean-up Columns | Purify final DNA after decrosslinking. | High recovery efficiency for low-yield reChIP DNA is paramount for downstream sequencing. |
| Control Primer Sets | qPCR validation for known positive/negative genomic loci. | Requires validated bivalent (e.g., developmental promoters) and monovalent control regions. |
Conclusion
The ChIP-reChIP protocol remains a powerful and definitive experimental tool for directly demonstrating the co-existence of bivalent histone marks on the same nucleosome, providing irreplaceable mechanistic insight into epigenetic gene regulation. By mastering the foundational concepts, meticulous methodology, troubleshooting tactics, and rigorous validation outlined, researchers can reliably map bivalent domains critical in development, cellular reprogramming, and disease pathogenesis. Future directions point towards integrating this technique with single-cell epigenomics and spatial transcriptomics, and its continued refinement will be vital for discovering and validating novel epigenetic drug targets, ultimately bridging the gap between chromatin biology and clinical therapeutics.