CTCF vs BORIS/CTCFL: Decoding DNA Binding Specificity for Epigenetic Regulation and Cancer Therapy
This article provides a comprehensive analysis of the DNA binding specificity of the architectural protein CTCF and its testis-specific paralog, BORIS/CTCFL, which is aberrantly expressed in cancers.
CTCF vs BORIS/CTCFL: Decoding DNA Binding Specificity for Epigenetic Regulation and Cancer Therapy
Abstract
This article provides a comprehensive analysis of the DNA binding specificity of the architectural protein CTCF and its testis-specific paralog, BORIS/CTCFL, which is aberrantly expressed in cancers. Targeting researchers and drug developers, we first explore their foundational biology, including shared zinc finger domains and divergent genomic targets. We then detail cutting-edge methodological approaches, from ChIP-seq to CRISPR screening, for mapping their binding landscapes. The article addresses key challenges in distinguishing their functions experimentally and offers optimization strategies. Finally, we present a comparative validation of their opposing roles in gene regulation and chromatin insulation, synthesizing current models of their antagonism in oncogenesis. The conclusion highlights implications for developing epigenetic therapies that target the CTCF/BORIS axis.
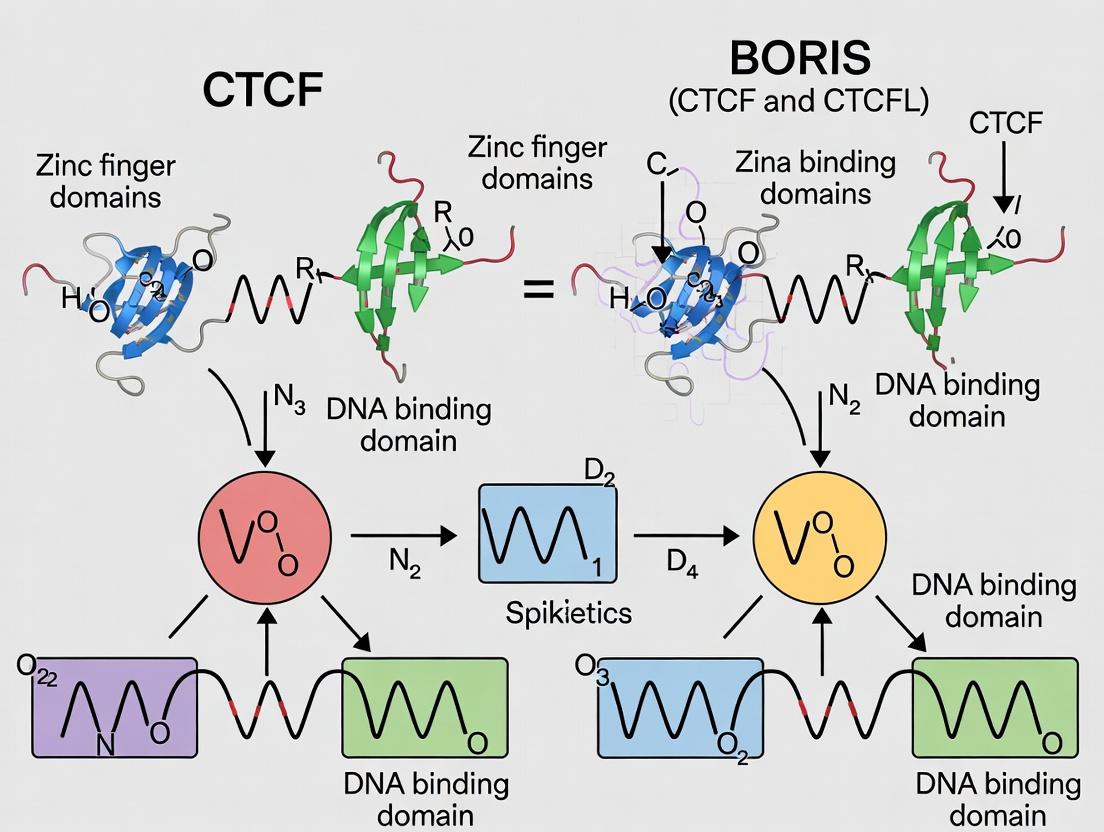
The Yin and Yang of Genome Architecture: Foundational Biology of CTCF and BORIS
Comparative Analysis: CTCF vs. BORIS (CTCFL) DNA Binding Specificity
This guide provides an objective performance comparison between CCCTC-binding factor (CTCF) and its paralog Brother of the Regulator of Imprinted Sites (BORIS/CTCFL) in key functional domains, framed within ongoing research on their distinct roles in genome organization and disease.
Genomic Binding and Motif Specificity
| Feature | CTCF | BORIS/CTCFL | Supporting Experimental Data & Source |
|---|---|---|---|
| Consensus Motif | Highly conserved 11-ZF domain binds ~20 bp motif (CCGCGNGGNGGCAG). | Shares core motif but shows distinct preference for methylated motifs. | ChIP-seq in somatic (CTCF) vs. testis/ectopic cancer cells (BORIS) reveals overlapping but non-identical sites. Nucleic Acids Res. 2020 |
| Binding Site Occupancy | >50,000 sites in mammalian genomes; constitutive in most cell types. | Limited in normal somatic cells; aberrantly expressed in cancers, binding ~30-70% of CTCF sites. | CUT&RUN in prostate cancer cell lines shows BORIS binds a subset of hypomethylated CTCF sites. Genome Biol. 2022 |
| Dependency on DNA Methylation | Binding inhibited by CpG methylation at core motif positions. | Can bind methylated motifs; may facilitate binding in heterochromatic regions. | EMSA with methylated probes shows reduced CTCF binding but stable or enhanced BORIS binding. Epigenetics Chromatin. 2021 |
| Impact on Chromatin Looping | Primary driver of TAD boundary formation and loop anchoring. | Can co-occupy some anchors but may form alternative or aberrant loops in cancer. | Hi-C in BORIS+ vs. BORIS- cells shows altered looping patterns at co-occupied loci. Nat Commun. 2023 |
Functional Consequences in Gene Regulation
| Feature | CTCF | BORIS/CTCFL | Supporting Experimental Data & Source |
|---|---|---|---|
| Transcriptional Role | Classic insulator; blocks enhancer-promoter communication. | Bivalent: can act as a transcriptional activator or repressor. | Reporter assays show BORIS can bypass CTCF-mediated enhancer blocking. Cell Rep. 2021 |
| Association with Disease | Haploinsufficiency linked to developmental syndromes (e.g., intellectual disability). | Oncogenic role: ectopic expression in cancers promotes proliferation, chemo-resistance. | CRISPR knockout of BORIS in melanoma cells reduces tumor growth in xenografts. Sci Adv. 2022 |
| Interaction Partners | Binds cohesin complex; interacts with RNA Pol II. | Interacts with testis-specific partners and cancer-associated transcription factors. | Co-immunoprecipitation/Mass Spec identifies distinct protein interactomes. Mol Cell Proteomics. 2023 |
Experimental Protocols for Key Comparisons
Protocol 1: Determining Binding Specificity via EMSA with Methylated Probes
Objective: Compare CTCF and BORIS binding affinity to methylated vs. unmethylated DNA motifs.
- Probe Preparation: Synthesize double-stranded DNA probes containing the consensus 20 bp motif. Treat one set with CpG methyltransferase (M.SssI) to create fully methylated probes.
- Protein Purification: Express and purify recombinant full-length CTCF and BORIS proteins with N-terminal GST tags from HEK293T cells.
- Binding Reaction: Incubate 10 fmol of labeled probe with increasing amounts (0-200 nM) of purified protein in binding buffer (10 mM Tris, 50 mM KCl, 1 mM DTT, 10% glycerol, 50 ng/µL poly(dI-dC)) for 30 min at 25°C.
- Electrophoresis: Resolve protein-DNA complexes on a pre-run, non-denaturing 6% polyacrylamide gel in 0.5x TBE at 4°C.
- Analysis: Quantify gel shift using phosphorimaging. Calculate dissociation constant (Kd) for each protein on each probe type.
Protocol 2: Assessing Genome-Wide Co-occupancy via CUT&RUN
Objective: Map genomic binding sites of CTCF and BORIS in the same cell line.
- Cell Preparation: Harvest ~500,000 cells expressing both proteins (e.g., a cancer cell line). Permeabilize with Digitonin.
- Antibody Binding: Incubate with primary antibody (anti-CTCF or anti-BORIS) overnight at 4°C.
- pA-MNase Binding & Cleavage: Add Protein A-Micrococcal Nuclease fusion protein. Activate MNase with CaCl₂ for 2 min to cleave DNA around antibody-bound sites.
- DNA Extraction & Library Prep: Extract released DNA fragments and prepare sequencing libraries.
- Bioinformatics: Align reads to reference genome. Call peaks (e.g., using SEACR). Identify overlapping and unique binding sites.
Protocol 3: Functional Impact on Looping via Hi-C
Objective: Determine the role of BORIS in altering 3D genome architecture.
- Experimental Design: Establish isogenic cell lines: Control (shScramble) and BORIS-Knockdown (shBORIS).
- Hi-C Library Preparation: Crosslink cells with 2% formaldehyde. Digest chromatin with HindIII. Fill ends and mark with biotin. Ligate proximally ligated fragments. Shear DNA, pull down biotinylated ligation junctions, and prepare sequencing libraries.
- Sequencing & Analysis: Perform deep sequencing (~150M read pairs per sample). Process data using HiC-Pro or similar. Call TADs (Topologically Associating Domains) and loops (e.g., using HiCCUPS).
- Integration: Overlap differential loops with BORIS/CTCF ChIP-seq peaks to assign causality.
Visualizations
Diagram 1: CTCF vs. BORIS Binding and Functional Consequences
Title: CTCF and BORIS DNA Binding Specificity and Outcomes
Diagram 2: Experimental Workflow for Comparative Binding Analysis
Title: Experimental Workflow for CTCF vs. BORIS Research
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in CTCF/BORIS Research | Key Application Example |
|---|---|---|
| Anti-CTCF Antibody (C-terminal) | Immunoprecipitation of endogenous CTCF for ChIP-seq/CUT&RUN. Critical for mapping binding sites. | Validated for use in ChIP-seq to identify constitutive TAD boundaries. |
| Anti-BORIS/CTCFL Antibody | Specific detection of BORIS protein, which shares high ZF domain homology with CTCF. | Essential for CUT&RUN in cancer cell lines to map ectopic BORIS binding sites. |
| Recombinant CTCF & BORIS Proteins (full-length, tagged) | Provide pure protein for in vitro assays (EMSA, SELEX) without cellular contaminants. | Used in side-by-side EMSA to directly compare binding kinetics to methylated DNA. |
| CpG Methyltransferase (M.SssI) | Enzymatically methylates all CpG dinucleotides in DNA probes for methylation-sensitivity assays. | Preparation of methylated probes for EMSA to test binding inhibition (CTCF) vs. tolerance (BORIS). |
| HindIII Restriction Enzyme | Frequent-cutter used in Hi-C library preparation to digest crosslinked chromatin. | Part of standard Hi-C protocol to assess changes in 3D genome architecture upon BORIS knockdown. |
| Protein A-Micrococcal Nuclease (pA-MNase) | Enzyme fusion protein for targeted chromatin cleavage in CUT&RUN. | Enables high-resolution, low-background mapping of CTCF/BORIS binding with low cell input. |
| dCTP-Biotin Nucleotide | Labels digested chromatin ends for pull-down of ligation junctions in Hi-C. | Critical step in Hi-C library prep to selectively sequence proximally ligated DNA fragments. |
| BORIS-specific shRNA/sgRNA | Knocks down or knocks out BORIS expression in gain-of-function cancer models. | Creating isogenic pairs to study the functional necessity of ectopic BORIS in altering 3D genome structure. |
Introduction This comparison guide is framed within the ongoing research thesis investigating the divergent DNA binding specificities and functions of the architectural protein CTCF and its paralog, Brother of the Regulator of Imprinted Sites (BORIS/CTCFL). While CTCF is ubiquitously expressed and a master regulator of chromatin architecture, BORIS exhibits a restricted expression pattern primarily in germ cells but is frequently aberrantly expressed in cancers. This guide objectively compares their performance as DNA-binding proteins, focusing on sequence specificity, genomic occupancy, and functional outcomes.
1. Comparison of Core Molecular Characteristics
| Feature | CTCF | BORIS/CTCFL |
|---|---|---|
| Gene Locus | 16q22.1 | 20q13.31 |
| Expression Pattern | Ubiquitous in somatic cells | Restricted to male germ cells (normal); ectopic in cancers |
| Protein Domains | 11 Zinc Fingers (ZFs), N- & C-terminal domains | 11 Zinc Fingers (highly homologous), divergent N- & C-termini |
| DNA Binding Motif | 15-bp core consensus (≈12-15 ZF contacts) | Highly similar, but not identical, core consensus |
| Binding Site Conservation | Highly conserved across evolution | Less conserved; species-specific differences noted |
| Post-Translational Modifications | Richly modified (e.g., poly(ADP)-ribosylation, phosphorylation) | Modification profile distinct and less characterized |
| Primary Function | Chromatin insulation, looping, imprinting, TAD boundary formation | Proposed role in epigenetic reprogramming; oncogenic driver |
2. Comparison of Genomic Binding and Functional Output
Table 1: Comparative ChIP-seq Analysis in an Ectopic Expression Model
| Parameter | CTCF | Ectopically Expressed BORIS | Experimental Evidence |
|---|---|---|---|
| Total Genomic Peaks | ~40,000 - 80,000 | ~15,000 - 30,000 (often subset of CTCF sites) | ChIP-seq in cancer cell lines (e.g., MCF-7, HeLa) |
| De Novo Motif Recovery | Strong match to canonical CTCF motif | Highly similar, but with subtle base preference variations | MEME/STAMP motif analysis |
| Overlap with CTCF Sites | N/A | 50-80% of BORIS sites co-occupied by CTCF | Bedtools intersect analysis |
| Unique BORIS Sites | N/A | 20-50% (often weaker, lower conservation) | Peaks called against IgG & CTCF ChIP controls |
| Impact on Gene Expression | Structural regulation; variable direct effects | Deregulation of cancer-testis antigens, oncogenes | RNA-seq upon BORIS knockdown/overexpression |
| Effect on Local Epigenetics | Maintains H3K27ac islands, protects from DNA methylation | Can recruit demethylases (e.g., TET1), alter histone marks | ChIP-seq for H3K4me3, H3K27ac, DNA methylation arrays |
Experimental Protocol: Comparative DNA-Binding Specificity Assay (HT-SELEX) Objective: To quantitatively compare the intrinsic DNA-binding sequence preferences of CTCF and BORIS zinc finger arrays. Methodology:
- Protein Purification: Recombinantly express and purify the 11-ZF domains of human CTCF and BORIS.
- Oligomer Library: Use a synthetic double-stranded DNA library containing a random 20-bp region flanked by fixed primers.
- Selection Rounds: Incubate the library with immobilized protein. Wash away unbound DNA. Elute and PCR-amplify protein-bound sequences.
- High-Throughput Sequencing: Repeat selection for 4-6 rounds. Sequence the enriched DNA pools from each round.
- Data Analysis: Use algorithms (like SELEX-seq) to compute position weight matrices (PWMs) for each protein. Compare motifs using Pearson correlation or Jensen-Shannon divergence.
Diagram: CTCF vs. BORIS Binding and Functional Consequences
3. Comparison in Cancer Context: Oncogenic "Performance"
Table 2: Functional Impact in Cancer Models
| Assay Metric | CTCF (Wild-type) | BORIS (Ectopic) | Interpretation |
|---|---|---|---|
| Cell Proliferation | Often essential; haploinsufficient tumor suppressor | Knockdown inhibits growth in BORIS+ cancer lines | BORIS acts as a lineage-specific oncogene. |
| Invasion/Migration | Can suppress (via TAD integrity) | Overexpression promotes EMT and metastasis | BORIS drives aggressive phenotypes. |
| Chemoresistance | Mutations can affect sensitivity | Expression correlates with resistance (e.g., to cisplatin) | BORIS as a potential therapeutic target. |
| Tumorigenicity in vivo | Loss reduces tumor growth | Xenografts of BORIS+ cells show increased tumor burden | Confirms oncogenic role. |
Experimental Protocol: Competitive Chromatin Immunoprecipitation (ChIP) Objective: To assess if BORIS displaces CTCF or co-occupies sites in cancer cells. Methodology:
- Cell Cross-linking: Treat BORIS-expressing cancer cells with 1% formaldehyde.
- Chromatin Shearing: Sonicate lysate to fragment DNA to 200-500 bp.
- Immunoprecipitation: Split chromatin into three aliquots for: a) anti-CTCF antibody, b) anti-BORIS antibody, c) Normal IgG (control).
- qPCR Analysis: Use primers for shared binding sites and unique sites. Quantify enrichment (% input).
- Sequential ChIP (Re-ChIP): Elute complexes from first ChIP (e.g., anti-CTCF) and subject to second IP with anti-BORIS. Analyze by qPCR to confirm direct co-occupancy.
The Scientist's Toolkit: Key Research Reagents Table 3: Essential Reagents for CTCF/BORIS Research
| Reagent | Function & Application | Key Consideration |
|---|---|---|
| Anti-CTCF Antibody (C-terminal) | ChIP-seq, WB, IF for endogenous CTCF. Does not cross-react with BORIS. | Critical for specific detection; validate loss of signal in CTCF-knockout cells. |
| Anti-BORIS Antibody | ChIP-seq, WB, IHC for endogenous BORIS. Must not recognize CTCF. | Challenging due to high homology; target unique N-terminal region. |
| Recombinant ZF Domain Proteins | EMSA, SELEX, crystallography for in vitro binding studies. | Purify both CTCF and BORIS ZF domains identically for fair comparison. |
| BORIS-Expressing Cancer Cell Line | Functional studies (proliferation, invasion). | e.g., MCF-7 (breast), HeLa (cervical), or lines with engineered overexpression. |
| CTCF/BORIS ChIP-seq Dataset | Bioinformatics analysis of binding sites and motif discovery. | Use from same cell line/model for direct comparison. Check GEO/SRA databases. |
| Methyl-Sensitive qPCR Assay | To measure DNA methylation changes at target loci after BORIS expression. | Probes specific for CpG islands near BORIS/CTCF binding sites. |
Diagram: Experimental Workflow for Binding Specificity Research
Thesis Context: CTCF vs. BORIS/CTCFL in DNA Binding Specificity
Within the broader thesis investigating the functional dichotomy between CTCF and its paralog BORIS (CTCFL), comparative genomic analysis of their zinc finger (ZF) DNA-binding domains (DBDs) is fundamental. CTCF, a ubiquitous multifunctional protein, and BORIS, a testis-specific protein with oncogenic potential, share high amino acid sequence identity in their 11-ZF DBDs. Despite this similarity, they exhibit divergent genomic binding profiles and biological functions, implicating subtle differences in ZF domain architecture and sequence specificity as key determinants.
Performance Comparison: CTCF vs. BORIS Zinc Finger Domains
The following tables summarize experimental data comparing the DNA-binding properties and functional outcomes of CTCF and BORIS ZF domains.
Table 1: Biochemical & Genomic Binding Comparison
| Parameter | CTCF ZF DBD | BORIS/CTCFL ZF DBD | Experimental Method & Reference |
|---|---|---|---|
| Consensus DNA Motif | 15-bp motif, asymmetric | Highly similar core, variant flanking sequences | Protein-binding microarray (PBM), SELEX |
| In Vivo Binding Site Overlap | ~40-60% of BORIS sites | ~80-90% of CTCF sites | ChIP-seq in BORIS-expressing somatic cells |
| Binding Affinity (Kd) to Shared Motif | 5-20 nM range | 10-50 nM range | Surface Plasmon Resonance (SPR) |
| Sensitivity to CpG Methylation | Binding blocked by methylation at key positions | Binding often maintained or less affected | Methyl-SELEX, ChIP-bisulfite sequencing |
| Primary Biological Role | Chromatin insulation, looping, imprinting | Transcriptional regulation in gametogenesis, aberrant in cancer | Functional genomic assays (4C, CRISPRi) |
Table 2: Structural & Domain Feature Comparison
| Feature | CTCF | BORIS/CTCFL | Functional Implication |
|---|---|---|---|
| Number of Zinc Fingers | 11 | 11 | Defines core DNA-binding capacity |
| Amino Acid Identity in DBD | 100% (reference) | ~71-78% | Alters DNA contact points and specificity |
| Key Divergent Residues | ZF2, ZF6, ZF10 | Different residues at same positions | Hypothesized to alter flanking sequence readout |
| Flanking Protein Domains | N-terminal, C-terminal regions distinct | Unique N-terminus, shared central region | Affects partner protein interactions (e.g., cohesion) |
| Expression Pattern | Ubiquitous, essential | Testis-specific, often silenced in somatic | Context-dependent genomic binding |
Experimental Protocols for Key Comparisons
1. Protocol: Protein-Binding Microarray (PBM) for Zinc Finger Motif Mapping
- Objective: Define and compare the precise DNA sequence preferences of purified CTCF and BORIS ZF DBDs.
- Procedure:
- Clone and express the 11-ZF DBD regions of human CTCF and BORIS as GST-tagged proteins in E. coli. Purify using glutathione-affinity chromatography.
- Incubate each purified protein with a commercial double-stranded DNA PBM slide containing all possible 10-mer sequences in duplicate.
- Detect bound protein using a fluorescently labeled anti-GST antibody.
- Analyze fluorescence intensity data with specialized algorithms (e.g., Seed-and-Wobble) to generate position weight matrices (PWMs) for each protein.
- Compare PWMs computationally to identify differences in preferred nucleotides at each position of the binding site.
2. Protocol: ChIP-seq for Comparative Genomic Binding Profiles
- Objective: Identify and compare the genome-wide occupancy sites of full-length CTCF and BORIS in a controlled cellular system.
- Procedure:
- Generate a somatic cell line (e.g., HEK293) stably expressing epitope-tagged BORIS via lentiviral transduction. Use parental cells for endogenous CTCF analysis.
- Crosslink chromatin with 1% formaldehyde for 10 minutes. Quench with glycine, harvest cells, and lyse.
- Sonicate chromatin to an average fragment size of 200-500 bp.
- Immunoprecipitate DNA-protein complexes using validated antibodies against CTCF and the tag on BORIS.
- Reverse crosslinks, purify DNA, and prepare sequencing libraries for high-throughput sequencing.
- Map reads to the reference genome, call peaks, and perform overlap analysis to define shared and unique binding sites.
3. Protocol: Surface Plasmon Resonance (SPR) for Binding Affinity
- Objective: Quantitatively measure the binding kinetics (Ka, Kd) of ZF DBDs to defined DNA motifs.
- Procedure:
- Design and synthesize biotinylated double-stranded DNA oligonucleotides containing the canonical CTCF motif or a variant identified in BORIS-specific sites.
- Immobilize the biotinylated DNA onto a streptavidin-coated SPR sensor chip.
- Flow purified CTCF and BORIS ZF DBD proteins at a range of concentrations (e.g., 1-200 nM) over the chip in running buffer.
- Monitor the association and dissociation phases in real-time to obtain sensorgrams.
- Fit the data to a 1:1 binding model to calculate association (Ka) and dissociation (Kd) rate constants, and the equilibrium dissociation constant (Kd).
Visualizations
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function in ZF Domain Analysis | Example Vendor/Cat. No. (Illustrative) |
|---|---|---|
| Recombinant ZF DBD Proteins (CTCF & BORIS) | Purified protein for in vitro assays (PBM, SPR, EMSA). Essential for controlled biochemical studies. | Active Motif, Abnova, custom expression from vector below. |
| Expression Vector for ZF Domains | Cloning and expressing GST- or His-tagged ZF domains in bacterial systems. | pGEX-6P-1 (GST tag), pET series (His tag). |
| Protein-Binding Microarray (PBM) | High-throughput determination of DNA binding specificity. | Agilent SurePrint PBM slides, custom designs from AMADID. |
| Validated ChIP-grade Antibodies | Immunoprecipitation of endogenous CTCF and tagged BORIS for ChIP-seq. | CTCF: Millipore 07-729; Anti-FLAG M2 (for tagged BORIS). |
| Biotinylated DNA Oligos for SPR | Immobilization on sensor chips to measure binding kinetics to specific motifs. | IDT DNA, HPLC-purified, 5' biotin modification. |
| SPR Sensor Chip | Surface for immobilizing DNA ligands to measure protein interaction in real-time. | Cytiva Series S Streptavidin (SA) chip. |
| Methylated CpG Oligonucleotides | Probes to test the effect of DNA methylation on ZF domain binding in EMSA or SPR. | Sigma-Aldrich, custom synthesized with 5-methylcytosine. |
| Chromatin Shearing Reagents | Consistent fragmentation of crosslinked chromatin for ChIP-seq. | Covaris ultrasonication system or enzymatic shearing kits (CST). |
| High-Fidelity DNA Polymerase | Cloning ZF domains and preparing sequencing libraries from ChIP DNA. | NEB Q5, Thermo Fisher Phusion. |
| Next-Gen Sequencing Library Prep Kit | Preparing ChIP-seq or SELEX libraries for Illumina sequencing. | Illumina TruSeq ChIP, NEB Next Ultra II. |
This guide compares the performance characteristics of the paralogous transcription factors CTCF and BORIS (CTCFL) in DNA binding, a critical parameter for understanding their divergent roles in gene regulation and cellular function.
Comparison of DNA Binding Specificity & Functional Output
| Feature | CTCF | BORIS/CTCFL |
|---|---|---|
| Primary Expression | Ubiquitous (Somatic cells) | Restricted (Germ cells, cancers) |
| Conserved Zinc Finger (ZF) Domain | 11 ZFs, identical to BORIS | 11 ZFs, identical to CTCF |
| Core Binding Motif | ~20bp consensus, CCCTC-specific | Shares core CTCF consensus motif |
| Genomic Binding Site Occupancy (ChIP-seq) | ~40,000 - 80,000 sites in somatic cells | Ectopic expression binds ~70% of CTCF sites |
| Binding Affinity (Kd) to Consensus | High-affinity (Low nM range) | Comparable high-affinity, but context-dependent |
| Methylation Sensitivity | Binding inhibited by CpG methylation at motif | Binding is methylation-resistant or facilitated |
| Primary Functional Role | Insulator, chromatin looping, enhancer blocking | Transcriptional activation, epigenetic reprogramming |
| Association with Co-factors | Cohesin, CHD8, YY1 | Distinct partners in testis and cancer contexts |
Experimental Protocol: Electrophoretic Mobility Shift Assay (EMSA) for Binding Specificity
Objective: To compare the binding affinity and specificity of purified CTCF and BORIS proteins to a panel of DNA probes, including consensus, methylated, and variant motifs.
- Protein Purification: Express and purify full-length, recombinant CTCF and BORIS proteins (e.g., with GST or FLAG tags) from mammalian (HEK293) or insect (Sf9) cell systems.
- Probe Preparation: Design and synthesize 5'-biotinylated double-stranded DNA oligonucleotides (~30-40bp) encompassing: a) Canonical CTCF consensus sequence, b) Same sequence with CpG methylation, c) Known divergent genomic sites.
- Binding Reaction: Incubate 10 fmol of labeled probe with a titrated amount of protein (0, 2, 5, 10, 20 nM) in binding buffer (10 mM HEPES, 50 mM KCl, 1 mM DTT, 2.5% glycerol, 50 ng/µL poly(dI-dC)) for 30 minutes at room temperature.
- Electrophoresis: Resolve protein-DNA complexes from free probe on a pre-run, non-denaturing 6% polyacrylamide gel in 0.5X TBE buffer at 100V for 60-90 minutes at 4°C.
- Detection: Transfer DNA to a nylon membrane, crosslink, and detect biotinylated probes using a chemiluminescent streptavidin-horseradish peroxidase system. Quantify band intensity to determine apparent Kd.
Visualization: CTCF vs. BORIS Binding Logic & Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in CTCF/BORIS Research |
|---|---|
| Anti-CTCF Antibody (ChIP-grade) | Immunoprecipitation of endogenous CTCF-DNA complexes for genome-wide binding site mapping (ChIP-seq). |
| Anti-BORIS/CTCFL Antibody | Critical for detecting the low-abundance BORIS protein in ChIP or western blot, with high specificity to avoid CTCF cross-reactivity. |
| Recombinant CTCF & BORIS Proteins | Full-length, tagged proteins for in vitro binding assays (EMSA, SELEX), structural studies, and affinity measurements. |
| Biotinylated CTCF Consensus Oligonucleotides | Probes for EMSA to validate binding and assess the impact of sequence mutations or CpG methylation on protein-DNA interaction. |
| Methylated CpG DNA Probes | Chemically synthesized DNA containing methyl-cytosine to test the methylation sensitivity/resistance of CTCF vs. BORIS binding. |
| dCas9-CTCF/BORIS Fusion Constructs | For targeted recruitment to specific genomic loci to study functional outcomes (e.g., insulator formation vs. activation) without confounding endogenous binding. |
| CTCF/BORIS Knockout Cell Lines | (e.g., via CRISPR-Cas9) Isogenic backgrounds to dissect unique and overlapping functions of each paralog without compensation. |
CTCF and BORIS (CTCFL) are paralogous DNA-binding proteins with identical 11-zinc finger domains, yet they exhibit starkly divergent expression patterns, functions, and roles in disease.
Table 1: Expression Profiles of CTCF vs. BORIS
| Feature | CTCF (CCCTC-Binding Factor) | BORIS (CTCFL, Brother of the Regulator of Imprinted Sites) |
|---|---|---|
| Primary Expression | Ubiquitous in somatic cells. Essential for viability. | Restricted to male germline (pre-meiotic spermatocytes). |
| Re-expression Context | N/A; constitutively expressed. | Reactivated in numerous cancers (e.g., breast, lung, ovarian, melanoma). |
| Regulatory Role | Master architectural protein; insulator, chromatin loop formation. | Proposed role in epigenetic reprogramming; may antagonize CTCF. |
| Methylation Sensitivity | Binds unmethylated consensus sequences; binding blocked by DNA methylation. | Binds similar consensus, but binding may be methylation-insensitive or preferential for methylated sequences in some contexts. |
| Cancer Association | Haploinsufficient tumor suppressor; frequent heterozygous mutations. | Oncogene candidate; ectopic expression promotes proliferation, invasion, chemoresistance. |
Table 2: Supporting Experimental Data from Recent Studies (2019-2023)
| Study Focus | Key CTCF Finding | Key BORIS Finding | Experimental Model |
|---|---|---|---|
| Global Binding (ChIP-seq) | Binds ~50-100k sites genome-wide, defining TAD boundaries. | In cancer cells, binds a subset of CTCF sites and unique targets, often associated with cancer-testis antigens. | Ovarian cancer cell lines, melanoma. |
| Expression vs. Outcome | High expression correlated with better prognosis in breast cancer. | High expression strongly correlated with poor prognosis, metastasis, and relapse in NSCLC and triple-negative breast cancer. | TCGA pan-cancer analysis. |
| Functional Knockdown | Cell cycle arrest, apoptosis, disruption of 3D genome. | Reduction in cancer cell stemness, migration, and tumor growth in xenografts. | Prostate cancer, glioblastoma stem cells. |
Detailed Experimental Protocols
Protocol 1: Chromatin Immunoprecipitation Sequencing (ChIP-seq) for CTCF/BORIS Binding
Objective: To map genome-wide DNA binding sites of CTCF and BORIS and compare their landscapes.
- Cross-linking: Treat cells (e.g., somatic vs. cancer cell lines) with 1% formaldehyde for 10 min at room temp.
- Cell Lysis & Chromatin Shearing: Lyse cells and sonicate chromatin to 200-500 bp fragments.
- Immunoprecipitation: Incubate chromatin with specific antibodies: anti-CTCF (rabbit monoclonal, D31H2) and anti-BORIS (rabbit polyclonal, validated for ChIP).
- Washing & Elution: Wash beads stringently, elute complexes, and reverse cross-links.
- Library Prep & Sequencing: Purify DNA, prepare sequencing libraries, and perform high-throughput sequencing (Illumina).
- Data Analysis: Align reads, call peaks (MACS2), and perform motif analysis (HOMER) and comparative visualization.
Protocol 2: Quantitative Analysis of Expression and Prognostic Correlation
Objective: To quantify CTCF/BORIS mRNA levels and correlate with clinical outcomes.
- Sample Collection: RNA from patient tumor cohorts (e.g., TCGA) or cell line panels.
- qRT-PCR: Use TaqMan assays (CTCF: Hs00975277m1; BORIS: Hs00223283m1). Normalize to GAPDH.
- Survival Analysis: Use public datasets (KM-Plotter, cBioPortal). Divide patients into high/low expression groups based on median value.
- Statistical Analysis: Generate Kaplan-Meier survival curves (Overall Survival, Disease-Free Survival) and calculate significance via log-rank test. Perform Cox regression for multivariate analysis.
Protocol 3: Functional Assay for Cancer Phenotypes
Objective: To assess the functional impact of BORIS reactivation in cancer cells.
- Genetic Manipulation: Lentiviral transduction of BORIS-overexpression or shRNA knockdown constructs in relevant cancer cell lines.
- Proliferation Assay: Measure cell viability over 72-96 hours using MTT or CellTiter-Glo.
- Migration/Invasion Assay: Use transwell chambers (Corning) with/without Matrigel coating. Stain and count migrated cells after 24-48 hours.
- In Vivo Tumorigenesis: Subcutaneously inject BORIS-knockdown or control cells into immunodeficient mice (NSG). Monitor tumor volume weekly for 4-8 weeks.
Visualizations
Title: Expression Contexts of CTCF and BORIS
Title: Proposed Binding Dynamics in Somatic vs. Cancer States
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for CTCF/BORIS Research
| Reagent | Function & Specificity | Example Product/Catalog # |
|---|---|---|
| Anti-CTCF Antibody (ChIP-grade) | For chromatin immunoprecipitation to map binding sites. Must be validated for ChIP-seq. | Cell Signaling Technology, D31H2 (Rabbit mAb #3418) |
| Anti-BORIS/CTCFL Antibody | Critical for detecting BORIS, which shares high sequence homology with CTCF. Requires validation for specific application (WB, IHC, ChIP). | Abcam, polyclonal (ab36992) for IHC/WB; Active Motif (61378) for ChIP. |
| CTCF/BORIS TaqMan Gene Expression Assays | For precise, specific quantification of mRNA levels without cross-detection between paralogs. | Thermo Fisher: CTCF (Hs00975277m1), BORIS (Hs00223283m1). |
| Recombinant Human BORIS Protein | For in vitro DNA-binding assays (EMSA), methylation sensitivity tests, and antibody validation. | Novus Biologicals, NBP2-59626. |
| Validated siRNA/shRNA Libraries | For targeted knockdown of CTCF or BORIS in functional assays. Requires careful design to avoid off-target effects. | Horizon Discovery: SMARTpools for CTCF (L-006957) and BORIS (L-017431). |
| Methylated & Unmethylated DNA Probes | Oligonucleotides containing the consensus CTCF/BORIS binding site, with/without CpG methylation, for EMSA competition assays. | Custom synthesis from IDT. |
| Positive Control Cell Lines | BORIS-positive (e.g., testis, certain cancer lines like MCF-7) and negative control (most somatic lines) for assay validation. | ATCC: NTERA-2 (germ cell model), MCF-7 (breast cancer). |
Within the broader thesis comparing CTCF and its paralog BORIS (CTCFL), the question of DNA binding specificity is paramount. Both proteins possess an identical array of 11 zinc fingers (ZFs), yet exhibit divergent genomic binding patterns and biological functions—CTCF as a ubiquitous architectural protein and BORIS as a testis-specific, often oncogenic, epigenetic reprogramming factor. This guide deconstructs the "11-ZF code" by comparing the core consensus binding motifs recognized by each protein, supported by experimental data on their performance in binding and functional assays.
Comparative Analysis of Core Binding Motifs
Experimental data consistently reveals that while CTCF and BORIS recognize highly similar core DNA sequences, critical variations in flanking sequences and methylation sensitivity dictate their distinct genomic occupancy.
Table 1: Comparison of Canonical vs. Validated Binding Motifs
| Feature | CTCF (Canonical) | BORIS/CTCFL (Validated) | Experimental Evidence & Assay |
|---|---|---|---|
| Core Consensus | CCGCGNGGNGGCAG (JASPAR MA0139.1) | Very similar, often CCGCGNGGNGGCAG | SELEX, Protein-Binding Microarrays (PBM) |
| Key Flanking Preference | Strong 5' upstream motif; specific 3' extension | Divergent flanking sequence preferences, often less stringent | High-Throughput SELEX, ChIP-seq motif discovery |
| CpG Methylation Sensitivity | Binding blocked by methylation of CpGs within core | Binding often insensitive or even preferred to methylated CpGs | EMSA with methylated probes, Methyl-Chip-seq |
| Binding Affinity (Kd approx.) | ~5-20 nM for optimal 20bp site (EMSA) | Comparable nM range for consensus site, but relative affinity for variant sites differs | Electrophoretic Mobility Shift Assay (EMSA), Surface Plasmon Resonance (SPR) |
| In Vivo Occupancy Specificity | Tens of thousands of sites genome-wide; highly conserved subset. | Ectopic expression leads to occupation of a subset of CTCF sites, plus unique sites. | Ectopic BORIS ChIP-seq in somatic cells vs. endogenous CTCF ChIP-seq. |
Table 2: Functional Consequences of Motif Recognition Differences
| Performance Metric | CTCF Outcome | BORIS Outcome | Supporting Experimental Data |
|---|---|---|---|
| Chromatin Looping | Forms stable, methylation-sensitive insulator loops and TAD boundaries. | Can displace CTCF and reconfigure loops, potentially via same motifs. | 3C/Hi-C upon BORIS induction; loss of CTCF at specific loci. |
| Transcriptional Regulation | Primarily insulator/repressor; can activate. | Often associated with epigenetic derepression of cancer-testis genes and oncogenes. | RNA-seq following BORIS overexpression; correlation with gene activation. |
| Dependency on ZF Integrity | Point mutations in key ZFs (e.g., ZF7) abolish binding and function. | Similar structural dependency, but mutation effects can be context-dependent. | In vitro binding assays with ZF mutants; functional rescue experiments. |
Key Experimental Protocols
Electrophoretic Mobility Shift Assay (EMSA) for Binding Specificity
Purpose: To compare the relative affinity and sequence specificity of purified CTCF vs. BORIS zinc finger domains. Protocol:
- Protein: Express and purify recombinant 11-ZF domains of human CTCF and BORIS.
- Probes: Generate 32P-end-labeled double-stranded DNA probes (30-40 bp): a) consensus motif, b) motif with CpG methylation, c) flanking sequence variants.
- Binding Reaction: Incubate 0-100 nM protein with 0.1 nM labeled probe in binding buffer (10 mM Tris, 50 mM KCl, 1 mM DTT, 10% glycerol, 0.1% NP-40, 100 ng/μL poly(dI-dC)) for 30 min at 25°C.
- Electrophoresis: Resolve complexes on a pre-run 6% non-denaturing polyacrylamide gel in 0.5x TBE at 4°C.
- Analysis: Quantify shifted band intensity via phosphorimaging to calculate apparent Kd.
Methyl-Sensitive ChIP-seq (ChIPmentation)
Purpose: To genome-wide profile BORIS vs. CTCF binding in the context of DNA methylation. Protocol:
- Cells: Use a somatic cell line engineered for inducible BORIS expression and a matched control.
- Crosslinking & Shearing: Crosslink with 1% formaldehyde. Sonicate chromatin to ~200-500 bp fragments.
- Immunoprecipitation: Use anti-CTCF and anti-BORIS antibodies. Perform ChIPmentation (tagmentation-based library prep) for low input.
- Parallel Methylation Analysis: Perform whole-genome bisulfite sequencing (WGBS) or MethylC-seq on input DNA from the same samples.
- Bioinformatics: Map reads, call peaks, discover de novo motifs. Overlap peaks with methylated CpG maps to determine methylation sensitivity.
Visualization of Concepts
Diagram 1: CTCF vs BORIS binding specificity divergence.
Diagram 2: Experimental workflow for deconstructing ZF code.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for 11-ZF Code Research
| Reagent/Solution | Function in Research | Key Consideration |
|---|---|---|
| Recombinant 11-ZF Proteins | Purified CTCF & BORIS zinc finger domains for in vitro binding assays (EMSA, SPR). | Ensure identical purification tags and buffers for fair comparison. |
| Methylated & Variant DNA Probes | Custom oligonucleotides with site-specific CpG methylation or flanking sequence changes. | Use high-fidelity synthesis and enzymatic methylation for probes. |
| Validated Antibodies | Anti-CTCF (for ChIP, WB) and Anti-BORIS (high specificity is critical for ChIP). | Validate BORIS antibody for lack of cross-reactivity with CTCF. |
| Inducible BORIS Expression System | Doxycycline-inducible lentiviral vector for ectopic BORIS expression in somatic cells. | Allows controlled, physiologically relevant expression levels. |
| Tagmentation-based ChIP Kit | For low-input or high-throughput ChIP-seq (ChIPmentation) of CTCF/BORIS. | Ideal for time-course experiments or limited cell numbers. |
| Whole-Genome Bisulfite Kit | To generate parallel methylation maps from the same cell samples used for ChIP. | Ensures integrated analysis of binding and methylation status. |
| 3C/Hi-C Library Prep Kit | To assess the structural consequences of CTCF displacement by BORIS. | Requires high sequencing depth and appropriate controls. |
Mapping the Epigenetic Battlefield: Techniques to Profile CTCF and BORIS Binding
This comparison guide evaluates ChIP-seq and CUT&Tag as gold-standard methods for mapping genome-wide protein-DNA interactions, framed within the critical research context of discerning the binding specificity of paralogous transcription factors CTCF and BORIS (CTCFL). Understanding their distinct or overlapping binding landscapes is essential for elucidating their roles in gene regulation, epigenetics, and oncogenesis.
Methodological Comparison: Core Protocols
Detailed ChIP-seq Experimental Protocol
- Crosslinking: Treat cells with 1% formaldehyde for 10 minutes at room temperature to fix protein-DNA interactions.
- Cell Lysis & Chromatin Shearing: Lyse cells and fragment chromatin via sonication to 200-500 bp fragments.
- Immunoprecipitation: Incubate chromatin with a target-specific antibody (e.g., anti-CTCF). Capture antibody-chromatin complexes using protein A/G magnetic beads.
- Washing & Elution: Wash beads stringently. Reverse crosslinks and elute DNA.
- Library Preparation: Repair DNA ends, add adapters, and PCR-amplify.
- Sequencing & Analysis: Perform high-throughput sequencing; map reads to reference genome and call peaks.
Detailed CUT&Tag Experimental Protocol
- Permeabilization: Harvest and permeabilize nuclei with digitonin.
- Antibody Binding: Incubate with a primary antibody against the target protein (e.g., anti-BORIS) overnight at 4°C.
- Secondary Antibody Binding: Add a secondary antibody conjugated to a Protein A-Protein G-Tn5 transposase fusion (pA-Tn5) for 1 hour.
- Tagmentation: Activate pA-Tn5 with Mg2+ to simultaneously cleave and tag genomic DNA in situ adjacent to the antibody target.
- DNA Extraction & Amplification: Extract tagmented DNA with SDS/Proteinase K. Perform PCR to add sequencing adapters.
- Sequencing & Analysis: Sequence and analyze similarly to ChIP-seq.
Performance Comparison Data
Table 1: Quantitative Method Comparison for CTCF/BORIS Studies
| Feature | ChIP-seq | CUT&Tag | Experimental Support & Relevance to CTCF/BORIS |
|---|---|---|---|
| Starting Material | 0.5-10 million cells | 50,000 - 100,000 cells | CUT&Tag enables profiling of rare cell populations or patient samples. |
| Handling Time | 3-5 days | 1-2 days | Faster turnaround with CUT&Tag accelerates screening of multiple conditions. |
| Signal-to-Noise Ratio | Moderate; depends on antibody | High; low background | CUT&Tag's clarity is crucial for distinguishing closely related binding sites of CTCF vs. BORIS. |
| Resolution | ~100-200 bp (based on fragment size) | ~20-40 bp (single transposase insertion site) | Higher resolution of CUT&Tag can pinpoint subtle differences in binding motifs. |
| Data Consistency | High, established protocol | High, but newer method | Both yield reproducible maps for comparative analysis of paralog binding. |
| Key Requirement | High-quality antibody, optimization of sonication | Permeabilization efficiency, active pA-Tn5 complex | Antibody specificity is paramount for both to avoid cross-reactivity between paralogs. |
Table 2: Typical Experimental Outcomes from Published Studies
| Metric | CTCF ChIP-seq | BORIS CUT&Tag | Interpretation |
|---|---|---|---|
| Peaks Identified | ~40,000 - 80,000 in mammalian cells | ~20,000 - 60,000 (cell-type dependent) | BORIS binds a subset of CTCF sites but also unique targets, indicating divergent functions. |
| Peak Overlap | ~60-80% of BORIS sites co-occupied by CTCF | ~30% of CTCF sites co-occupied by BORIS | Suggests BORIS may compete with or modulate CTCF at a specific subset of loci. |
| Motif Enrichment | Canonical CTCF motif highly enriched | Divergent motif variant enriched | Explains differences in binding specificity and downstream partner recruitment. |
Visualizing Workflows and Biological Context
Title: ChIP-seq Experimental Workflow
Title: CUT&Tag Experimental Workflow
Title: Method Integration for CTCF/BORIS Thesis
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for ChIP-seq and CUT&Tag Studies
| Item | Function | Specific Consideration for CTCF/BORIS |
|---|---|---|
| High-Specificity Antibodies | Immunoenrichment of target protein-DNA complexes. | Must discriminate between CTCF and BORIS paralogs; rigorous validation (knockout/knockdown) is critical. |
| Protein A/G Magnetic Beads (ChIP-seq) | Capture antibody-bound chromatin complexes. | Efficiency impacts yield, especially for lower-abundance factors like BORIS. |
| pA-Tn5 Fusion Protein (CUT&Tag) | Tethers tagmentation enzyme to antibody for targeted fragmentation. | Commercial kits ensure consistent activity, crucial for reproducible results. |
| Digitonin (CUT&Tag) | Permeabilizes nuclear membrane while preserving internal structures. | Concentration must be optimized for different cell types to allow antibody/Tn5 entry. |
| Formaldehyde (ChIP-seq) | Crosslinks proteins to DNA to preserve transient interactions. | Over-fixation can mask epitopes or reduce sonication efficiency. |
| Next-Generation Sequencing Platform | High-throughput readout of enriched DNA fragments. | Sufficient sequencing depth (~20-50M reads) required to map both strong and weak binding sites. |
| Cell Line/Tissue with Endogenous or Ectopic CTCF/BORIS Expression | Biological source for experimentation. | Isogenic systems expressing one paralog at a time are ideal for defining specific binding profiles. |
In the investigation of paralogous transcription factors CTCF and BORIS (CTCFL), defining their distinct DNA-binding specificities is paramount. This comparison guide objectively evaluates two core methodologies for motif analysis: traditional competitive Electrophoretic Mobility Shift Assay (EMSA) and high-throughput Systematic Evolution of Ligands by EXponential enrichment (SELEX).
Experimental Performance Comparison
The following table summarizes the quantitative performance characteristics of both methods based on published studies in CTCF/BORIS research.
Table 1: Method Comparison for CTCF/BORIS Motif Analysis
| Feature | Competitive EMSA | High-Throughput SELEX (e.g., SELEX-seq) |
|---|---|---|
| Throughput | Low (1-10 probes per gel) | Very High (10⁶-10¹² sequences per round) |
| Resolution | Binding affinity ranking for pre-defined motifs. | De novo identification of consensus motif. |
| Quantitative Output | Apparent dissociation constant (Kd, app); relative affinity. | Position Weight Matrix (PWM) with comprehensive affinity data. |
| Specificity Mapping | Can test mutant competitors to define critical bases. | Exhaustively maps the relative importance of every base position. |
| Key Data from CTCF Studies | Kd for consensus CTCF motif ~5-20 nM; BORIS shows similar affinity but potential motif variant preference. | Revealed an extended 20bp motif for CTCF; BORIS SELEX identifies a divergent core (e.g., differing at positions 4, 9, 13). |
| Time to Result | Days for a focused set of probes. | Weeks for full selection, sequencing, and bioinformatics. |
| Cost per Datum | High for sequence space coverage. | Low per sequenced ligand. |
Detailed Experimental Protocols
Protocol 1: Competitive EMSA for CTCF/BORIS Specificity
- Protein Purification: Purify recombinant CTCF or BORIS DNA-binding domain (ZF domain) using affinity tags (e.g., GST, His-tag).
- Probe Labeling: End-label a dsDNA probe containing the putative consensus motif with γ-³²P-ATP using T4 Polynucleotide Kinase.
- Binding Reaction: Incubate a fixed, limiting concentration of purified protein (e.g., 10 nM) with the labeled probe (0.1 nM) in a buffer containing 10 mM Tris-HCl (pH 7.5), 50 mM KCl, 1 mM DTT, 0.1 mg/mL BSA, 5% glycerol, and 50 ng/μL poly(dI·dC) as non-specific competitor.
- Competition: Include increasing molar excesses (e.g., 1x, 10x, 100x) of unlabeled competitor DNA. Competitors include wild-type motif, scrambled sequence, and single-nucleotide mutants.
- Electrophoresis: Resolve protein-DNA complexes from free probe on a pre-run, non-denaturing 6% polyacrylamide gel in 0.5x TBE buffer at 4°C.
- Analysis: Quantify band intensities. The competitor concentration that reduces bound probe by 50% (IC₅₀) is used to calculate relative binding affinity.
Protocol 2: High-Throughput SELEX (SELEX-seq)
- Library Construction: Synthesize a random dsDNA oligonucleotide library (e.g., 20-40 bp random flanked by constant primer regions).
- Selection Rounds: a. Binding: Incubate the library with immobilized His-tagged CTCF or BORIS ZF domain. b. Washing: Remove unbound and weakly bound sequences. c. Elution: Recover bound DNA, typically by competitive elution with high-salt buffer or specific oligonucleotide. d. Amplification: PCR-amplify eluted DNA for the next round (typically 6-10 rounds).
- Sequencing: Subject PCR products from early and late rounds to high-throughput sequencing (Illumina).
- Bioinformatic Analysis: Use tools like MEME or STREME to identify enriched motifs and generate PWMs. Compare motif enrichment between CTCF and BORIS selections.
Visualizations
Title: Comparative Workflow: EMSA vs. SELEX
Title: CTCF vs. BORIS DNA Binding Specificity Model
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Motif Analysis Studies
| Reagent/Material | Function in Experiment | Example Vendor/Product |
|---|---|---|
| Recombinant Protein (CTCF/BORIS ZF) | DNA-binding subject for in vitro assays. | Purified from E. coli or baculovirus system; commercial (e.g., Active Motif). |
| T4 Polynucleotide Kinase | Radioactively (³²P) or chemically labels DNA probes for EMSA detection. | Thermo Fisher Scientific. |
| Non-specific Competitor DNA (poly(dI·dC)) | Blocks non-specific protein-DNA interactions in EMSA binding reactions. | Sigma-Aldrich. |
| Streptavidin/His-Tag Magnetic Beads | For immobilizing protein during SELEX selection rounds. | Dynabeads (Thermo Fisher). |
| High-Fidelity DNA Polymerase | For accurate amplification of SELEX library between selection rounds. | Q5 or Phusion (NEB). |
| Random Oligo SELEX Library | Starting pool for de novo motif discovery. | Custom synthesis (IDT). |
| Next-Generation Sequencing Service | Analysis of enriched sequences from SELEX. | Illumina platforms. |
| Motif Discovery Software | Generates PWMs from sequenced SELEX pools. | MEME Suite, STREME. |
Within the broader thesis investigating the distinct DNA binding specificities and functional antagonism of CTCF, the canonical chromatin insulator protein, and its paralog BORIS/CTCFL, typically expressed in germ cells and often re-activated in cancers, functional genomics approaches are paramount. CRISPR-mediated knockout (KO) and knockdown (KD) models have become indispensable for deciphering the precise regulatory consequences of modulating these factors. This guide compares the performance of key CRISPR-based methodologies in this specific research context.
CRISPR KO vs. KD: A Comparative Guide for CTCF/BORIS Studies
The choice between permanent gene knockout and transient or partial knockdown hinges on the biological question, the essentiality of the target gene, and the desired experimental timeline.
Table 1: Comparison of CRISPR KO and KD Approaches
| Feature | CRISPR Knockout (KO) | CRISPR Knockdown (KD) / Interference (CRISPRi) |
|---|---|---|
| Molecular Target | Genomic DNA (coding exons) | Transcriptional regulation (promoter/enhancer) |
| Common Mechanism | Cas9 nuclease + sgRNA → DSBs → indel mutations | dCas9 fused to repressive domain (e.g., KRAB) + sgRNA |
| Effect on Protein | Complete, permanent loss of functional protein. | Partial, reversible reduction in transcription. |
| Key Applications in CTCF/BORIS Research | Study of complete loss-of-function, long-term chromatin architecture changes, cell viability assays. | Study of dosage-sensitive effects, essential genes (like CTCF), fine-tuning expression levels. |
| Timeline of Effect | Permanent; effect persists after sgRNA/Cas9 removal. | Transient; effect diminishes after sgRNA/dCas9 loss. |
| Off-Target Concerns | Potential for off-target mutagenesis. | Typically no DNA cleavage; off-target transcriptional repression. |
| Experimental Data Example | CTCF KO: Leads to catastrophic loss of topologically associating domain (TAD) boundaries, misexpression of genes, often cell lethal. | CTCF KD (CRISPRi): Results in graded weakening of TAD boundaries and proportional gene expression changes, allowing study of dosage effects. |
Table 2: Supporting Experimental Data from Recent Studies (2023-2024)
| Study Focus | Model & Method | Key Quantitative Findings | Implication for CTCF/BORIS Thesis |
|---|---|---|---|
| CTCF Dosage Effects | HCT-116 cells; CRISPRi (dCas9-KRAB) with titration of sgRNAs. | 70% KD of CTCF mRNA reduced TAD boundary strength by ~40% (Hi-C data). Specific promoter-enhancer loops decreased by 50-60%. | Demonstrates BORIS may compete with sub-stoichiometric CTCF, disrupting loops even without full CTCF loss. |
| BORIS Oncogenic Function | Non-small cell lung cancer cell line; CRISPR KO of CTCFL. | KO reduced cell proliferation by 65% (CellTiter-Glo). RNA-seq showed 285 genes differentially expressed (log2FC >1, p<0.01). | Confirms BORIS as a therapeutic target; its binding may directly antagonize CTCF sites in cancer. |
| Binding Site Resolution | Mouse Embryonic Stem Cells; Paired CTCF KO & BORIS ectopic expression. | ChIP-seq: 38% of endogenous CTCF binding sites were occupied by ectopic BORIS. These sites showed a 2.3-fold increase in DNA methylation. | Direct evidence for shared site recognition but divergent epigenetic consequences, a core thesis tenet. |
Detailed Experimental Protocols
Protocol 1: Generating a Clonal CTCF Knockout Cell Line using CRISPR-Cas9
- sgRNA Design: Design two sgRNAs targeting early exons of the CTCF gene. Use tools like CRISPick or CHOPCHOP, prioritizing on-target and off-target scores.
- Cloning: Clone sgRNA sequences into a lentiviral plasmid (e.g., lentiCRISPRv2).
- Virus Production & Transduction: Produce lentivirus in HEK293T cells and transduce target cells (e.g., HCT-116).
- Selection & Cloning: Apply puromycin selection (2 µg/mL, 5-7 days). Perform limiting dilution to isolate single-cell clones.
- Genotype Validation: Extract genomic DNA from clones. PCR-amplify the targeted region and analyze by Sanger sequencing and TIDE analysis to confirm bi-allelic frameshift indels.
- Phenotype Validation: Confirm loss of CTCF protein via Western Blot (anti-CTCF antibody) and loss of canonical binding via CTCF ChIP-qPCR at a control locus.
Protocol 2: Transient CTCF/BORIS Knockdown using CRISPRi
- Cell Line Engineering: Stably express dCas9-KRAB in your cell line of interest via lentiviral transduction and blasticidin selection.
- sgRNA Design: Design 3-5 sgRNAs targeting the promoter region (TSS ± 500 bp) of CTCF or CTCFL. Use CRISPRi-specific design rules (e.g., from the Weissman lab).
- Transient Delivery: Transfect dCas9-KRAB cells with sgRNA plasmids (or deliver as synthetic sgRNA complexed with recombinant dCas9-KRAB protein for maximum speed).
- Harvest & Analysis: Harvest cells 72-96 hours post-transfection. Analyze knockdown efficiency by RT-qPCR (using TaqMan probes for CTCF/BORIS) and assess functional consequences by RNA-seq or targeted assays (e.g., 3C-qPCR for chromatin looping).
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Solution | Function in CTCF/BORIS CRISPR Studies |
|---|---|
| lentiCRISPRv2 (Addgene #52961) | All-in-one lentiviral vector for stable expression of Cas9 and sgRNA; used for generating KO cell lines. |
| pHAGE-EF1α-dCas9-KRAB (Addgene #50919) | Lentiviral vector for stable expression of the CRISPRi effector dCas9-KRAB. |
| Validated Anti-CTCF Antibody (Cell Signaling #3418) | For Western Blot and ChIP validation of CTCF protein loss or displacement. |
| Anti-BORIS/CTCFL Antibody (Abcam ab25949) | For detecting BORIS protein expression in ectopic expression or cancer models. |
| Hi-C Kit (e.g., Arima-HiC) | To quantify genome-wide changes in 3D chromatin architecture (TADs, loops) upon perturbation. |
| Sanger Sequencing & TIDE Analysis Web Tool | A rapid, quantitative method to assess CRISPR editing efficiency and indel profiles in mixed or clonal populations. |
Visualizing Experimental Workflows and Molecular Consequences
Diagram 1: CRISPR KO and KD Workflows for CTCF Study
Diagram 2: CTCF vs BORIS Competition Alters Genome Architecture
This comparison guide evaluates X-ray crystallography and Cryo-electron microscopy (Cryo-EM) for determining the structures of protein-DNA complexes, with experimental data contextualized within research on the DNA binding specificity of paralogs CTCF and BORIS/CTCFL.
Comparative Performance Analysis
Table 1: Technical Comparison of X-ray Crystallography vs. Cryo-EM for Protein-DNA Complexes
| Parameter | X-ray Crystallography | Cryo-EM (Single Particle Analysis) |
|---|---|---|
| Typical Resolution Range | Atomic (0.8 – 3.0 Å) | Near-atomic to Atomic (1.8 – 4.0 Å for well-behaved complexes) |
| Sample Requirement | Highly ordered, large 3D crystals | Purified complex in solution (30-300+ kDa) |
| Sample State | Crystal lattice | Near-native, frozen-hydrated |
| Throughput (Data to Model) | Days to weeks (if crystal is available) | Weeks |
| Radiation Damage | High (total destruction) | Lower (per-particle dose < 20 e⁻/Ų) |
| Advantage for DNA Complexes | Precise DNA backbone & protein side-chain interactions; small molecule (drug) binding sites. | Captures flexible or heterogeneous complexes (e.g., partial binding, multiple conformations). |
| Limitation for DNA Complexes | Crystal packing may distort DNA/protein conformation; crystallization of flexible complexes is challenging. | Lower resolution can blur details of DNA base-specific contacts; requires substantial particle counts. |
Table 2: Experimental Data from CTCF/BORIS DNA-Binding Domain (DBD) Studies
| Study Focus | Method Used | Key Structural Finding | Resolution | Published Data Insight |
|---|---|---|---|---|
| CTCF Zinc Finger (ZF) Array bound to DNA | X-ray Crystallography | 11 ZFs follow DNA major groove, with ZF2-4 critical for core motif recognition. | 2.13 Å | Precise H-bond networks between ZF residues and DNA bases defined. |
| CTCF ZF Array with variant DNA sequences | Cryo-EM | Captured alternative conformations of ZFs 6-8 when bound to non-consensus sites. | 3.5 Å | Revealed mechanistic basis for divergent sequence recognition flexibility. |
| BORIS/CTCFL DBD Model | X-ray Crystallography (homology) & Cryo-EM | Highly similar ZF fold, but subtle differences in ZF10-11 interface alter DNA backbone contact geometry. | 2.7 Å (X-ray), 4.1 Å (Cryo-EM ensemble) | Structural rationale for overlapping but distinct genomic binding profiles vs. CTCF. |
Detailed Experimental Protocols
Protocol 1: X-ray Crystallography of a CTCF DNA-Binding Domain Complex
- Protein Production: Express the recombinant human CTCF ZF array (ZF 1-11) in E. coli and purify via affinity (Ni-NTA) and size-exclusion chromatography (SEC).
- DNA Preparation: Synthesize complementary oligonucleotides containing the core CTCF consensus sequence, anneal them to form double-stranded DNA.
- Complex Formation & Crystallization: Mix protein and DNA at a 1:1.2 molar ratio. Incubate on ice for 1 hour. Use SEC to isolate the homogeneous complex. Perform crystallization screens via vapor diffusion. Crystals often form in conditions containing PEG and divalent cations (e.g., 20% PEG 3350, 200 mM magnesium chloride).
- Data Collection & Processing: Flash-cool crystal in liquid nitrogen with cryoprotectant. Collect X-ray diffraction data at a synchrotron beamline. Index, integrate, and scale data (software: HKL-2000, XDS). Solve structure by molecular replacement using a related ZF model (PDB: XXXX). Iteratively refine with phenix.refine and Coot.
Protocol 2: Cryo-EM of a BORIS-DNA Complex in Multiple Conformational States
- Sample Vitrification: Purify the full-length BORIS-DNA complex via SEC in a buffer optimized for stability (e.g., 20 mM HEPES pH 7.5, 150 mM KCl). Apply 3 µL of sample (~0.8 mg/mL) to a glow-discharged cryo-EM grid (Quantifoil R1.2/1.3), blot for 3-4 seconds, and plunge-freeze in liquid ethane using a Vitrobot (100% humidity, 4°C).
- Data Acquisition: Collect movies on a 300 kV Cryo-TEM with a K3 direct electron detector in counting mode. Use a defocus range of -0.8 to -2.0 µm. Automated acquisition software (SerialEM) collects ~5,000 movies at a pixel size of 0.82 Å/pixel and a total dose of 50 e⁻/Ų.
- Image Processing: Motion correct and dose-weight movies (MotionCor2). Estimate CTF parameters (Gctf). Perform particle picking (crYOLO), extract particles, and conduct 2D classification to remove junk. Generate an initial model ab initio, followed by heterogeneous 3D classification (CryoSPARC) to separate distinct conformational states.
- High-Resolution Reconstruction: For each homogeneous subset, perform non-uniform refinement and CTF refinement to obtain final 3D maps. Sharpening is done via post-processing (DeepEMhancer).
Visualization of Methodologies and Biological Context
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Structural Studies of Protein-DNA Complexes
| Item | Function in Experiment | Example Product/Catalog |
|---|---|---|
| Recombinant Protein Expression System | High-yield production of pure, functional protein-DNA binding domains. | HiScribe T7 High Yield RNA Synthesis Kit (NEB), Rosetta(DE3) Competent Cells (Novagen). |
| Modified DNA Oligonucleotides | Incorporation of halogenated nucleotides (e.g., Br-dU) for crystallography phasing via SAD/MAD. | Bromodeoxyuridine (BrdU)-containing oligos (IDT). |
| Size-Exclusion Chromatography (SEC) Columns | Critical final step for isolating monodisperse, correctly assembled protein-DNA complexes. | Superdex 75 Increase or Superose 6 Increase (Cytiva). |
| Crystallization Screening Kits | Initial sparse-matrix screens to identify conditions for 3D crystal formation. | JCGSG Core Suites I-IV (Qiagen), MemGold & MemGold2 (Molecular Dimensions). |
| Cryo-EM Grids | Ultrathin, fenestrated carbon films on gold or copper mesh for sample vitrification. | Quantifoil R1.2/1.3 Au 300 mesh (Electron Microscopy Sciences). |
| Cryo-EM Sample Vitrification System | Automated, temperature/humidity-controlled plunger for reproducible thin ice formation. | Vitrobot Mark IV (Thermo Fisher Scientific). |
| Direct Electron Detector | High-sensitivity, fast camera for recording Cryo-EM movies with minimal noise. | Falcon 4i or K3 (Gatan) cameras. |
| Structural Biology Software Suite | Integrated platform for data processing, model building, refinement, and validation. | Phenix (UC Berkeley), CryoSPARC (Structura Biotechnology). |
This comparison guide evaluates methodologies for integrating chromatin conformation capture (Hi-C) data with transcription factor binding profiles to analyze chromatin loops. The analysis is framed within ongoing research on the DNA binding specificity of CTCF versus its paralog, BORIS (CTCFL), which is crucial in chromatin organization and gene regulation, with implications in oncology and drug development.
Comparison of Loop-Calling & Integration Tools
Table 1: Performance Comparison of Key Analysis Tools
| Tool / Pipeline | Primary Function | Key Metric: Loop Calling Precision (vs. ChIA-PET) | Key Metric: Run Time (on 1kb Human Hi-C) | Integration with Binding Data (e.g., ChIP-seq) | BORIS/CTCF Specificity Analysis |
|---|---|---|---|---|---|
| Fit-Hi-C (v2) | Statistical significant interaction detection | ~78% | ~4 hours | Requires separate binding data as input | Can use BORIS/CTCF peaks to filter/validate loops. |
| HICCUPS (Juicer Tools) | Hi-C loop calling from contact maps | ~85% | ~1.5 hours | Native integration from aligned ChIP-seq peaks | Excellent for identifying CTCF-mediated loops; BORIS loops often co-locate. |
| Mustache | Deep-learning based loop caller | ~88% | ~2 hours | Post-hoc integration; can correlate with peak files. | High recall for atypical loops; useful for studying BORIS in cancer cells. |
| Chromosight | Pattern-based detection (loops, borders) | ~82% | ~3 hours | Not direct; outputs can be overlapped with binding sites. | Effective for conserved loop structures; distinguishes constitutive vs. facultative. |
| MAPS (Model-based Analysis) | Integrates ChIP-seq with Hi-C for loops | ~92% | ~6 hours | Native joint modeling of Hi-C and ChIP-seq data | Specifically designed to assign loops to factors (CTCF vs. BORIS). |
Experimental Protocols for Key Studies
Protocol 1: Validating BORIS-Specific Loops in a Cancer Cell Line
Objective: Identify chromatin loops specifically associated with BORIS binding, not CTCF, in a testicular germ cell tumor cell line (NCCIT). Methodology:
- Hi-C Library Preparation: Use in-situ Hi-C protocol with MboI restriction enzyme on NCCIT cells. Sequence to a depth of ~2 billion paired-end reads.
- ChIP-seq for CTCF & BORIS: Perform parallel ChIP-seq experiments in the same cell line using validated, specific antibodies for CTCF and BORIS.
- Data Processing: Align Hi-C reads, generate normalized contact matrices at multiple resolutions (5kb, 10kb). Call peaks from ChIP-seq data.
- Loop Calling & Integration: Run HICCUPS on the Hi-C data to generate a base loop set. Overlap loop anchors with CTCF and BORIS ChIP-seq peaks. Use MAPS pipeline to statistically assign loops to BORIS binding events, controlling for background CTCF presence.
- Validation: Perform 4C-seq or ChIA-PET using BORIS antibody on selected candidate loops.
Protocol 2: Comparing Loop Stability upon Factor Depletion
Objective: Quantify the differential effect of CTCF vs. BORIS degradation on 3D chromatin architecture. Methodology:
- Cell Engineering: Create isogenic cell lines with auxin-inducible degron tags on endogenous CTCF and BORIS.
- Time-Series Experiment: Treat cells with auxin for 0, 6, 12, and 24 hours. Collect samples for Hi-C and RNA-seq at each time point.
- Differential Loop Analysis: Process Hi-C data per time point. Use Mustache to call loops for each condition. Employ DiffLoop (or comparable tool) to identify loops that significantly weaken or disappear upon depletion of each factor.
- Integration: Correlate loop strength changes with changes in ChIP-seq signal loss and gene expression alterations from RNA-seq.
Visualizations
Workflow for Integrative Loop Analysis
CTCF vs BORIS Looping Paradigm
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Tools for Integrated Loop Analysis
| Item | Function in Research | Example/Provider Notes |
|---|---|---|
| Anti-CTCF Antibody (ChIP-seq grade) | Immunoprecipitation of CTCF-bound DNA for mapping binding sites. | Millipore Sigma 07-729; validated for ChIP-seq specificity. |
| Anti-BORIS/CTCFL Antibody | Critical for mapping the often low-abundance BORIS binding sites. | Abcam ab25949; recommended for germ cell and cancer cell lines. |
| Hi-C Sequencing Kit | Standardized library prep for chromatin conformation capture. | Arima-HiC+ Kit (improved signal-to-noise), Dovetail Omni-C Kit (enzyme-based). |
| CTCF/BORIS Motif Plasmid | For positive control in EMSA or reporter assays of binding specificity. | Addgene #92385 (CTCF zinc finger array). |
| Degron-Tagged Cell Line | Enables rapid, inducible degradation of target protein (CTCF/BORIS). | Available through collaborating labs or generated via CRISPR/AID system. |
| Juicer Tools Software Suite | Standard pipeline for processing Hi-C data to contact matrices. | Open-source; includes HICCUPS loop caller. Essential for benchmarking. |
| Integrated Analysis Pipeline (MAPS) | Statistical framework for joint analysis of Hi-C and ChIP-seq data. | Requires R/Python environment. Key for factor-specific loop assignment. |
| 4C-seq Primer Design Tool | For designing validation primers targeting specific loop anchors. | FourCSeq (Bioconductor) or custom design around BORIS ChIP-seq summit. |
Publish Comparison Guide: Antibody Performance in ChIP-seq for CTCF vs. BORIS
A critical step in defining oncogenic BORIS target networks is reliably mapping its genome-wide binding sites and comparing them to its paralog, CTCF. This guide compares commonly used antibodies for Chromatin Immunoprecipitation followed by sequencing (ChIP-seq).
Table 1: Comparison of Antibodies for CTCF/BORIS ChIP-seq
| Target | Vendor (Catalog #) | Clonality | Reported Sensitivity (Peak Count) | Specificity (Cross-Reactivity Check) | Key Application in Cited Studies |
|---|---|---|---|---|---|
| CTCF | Cell Signaling (2899S) | Monoclonal (Rabbit) | High (~50,000-80,000 peaks in mammalian cells) | No cross-reactivity with BORIS. Validated by knockout/knockdown. | Gold standard for reference CTCF binding maps. |
| BORIS | Abcam (ab56329) | Polyclonal (Rabbit) | Medium-High (~15,000-30,000 peaks in BORIS+ cancer cells) | May show weak cross-reactivity with CTCF in some assays. Requires careful controls. | Used to identify ectopic BORIS targets in testis and cancer. |
| BORIS/CTCFL | Active Motif (61311) | Monoclonal (Mouse) | Medium (~10,000-20,000 peaks) | High specificity for BORIS, minimal cross-reactivity with CTCF. | Preferred for studies requiring high specificity over maximal sensitivity. |
Experimental Protocol: Sequential ChIP-seq (Re-ChIP) for Overlapping Sites Purpose: To directly identify genomic loci co-occupied by CTCF and BORIS in cancer cells expressing both proteins.
- Crosslinking & Sonication: Cells are fixed with 1% formaldehyde. Chromatin is sheared via sonication to 200-500 bp fragments.
- First Immunoprecipitation: Chromatin is incubated with an anti-CTCF monoclonal antibody (e.g., Cell Signaling 2899S) and protein A/G beads.
- Elution: The CTCF-bound chromatin complexes are eluted from the beads using a buffer containing 10mM DTT at 37°C for 30 minutes.
- Second Immunoprecipitation: The eluate is diluted and subjected to a second IP with an anti-BORIS antibody (e.g., Active Motif 61311).
- Processing & Sequencing: The final co-bound DNA is decrosslinked, purified, and used to construct a sequencing library. Interpretation: Peaks from this Re-ChIP-seq dataset represent sites of potential direct competition or collaboration, crucial for understanding BORIS-mediated oncogenic reprogramming.
Title: Workflow for Sequential ChIP-seq (Re-ChIP)
Publish Comparison Guide: CRISPR-based Functional Validation Tools
After identifying BORIS target networks, functional validation of key nodes is essential. This guide compares methods for perturbing gene expression of candidate targets.
Table 2: Comparison of Functional Validation Methods for BORIS Network Nodes
| Method | Mechanism | Key Advantage | Limitation | Typical Experimental Readout |
|---|---|---|---|---|
| CRISPR/Cas9 Knockout | Indels causing frameshift/nonsense mutations. | Complete, permanent loss of function. | Possible compensatory adaptations; slow for screening. | Western blot, proliferation/apoptosis assays. |
| CRISPR Interference (CRISPRi) | dCas9 fused to KRAB repressor blocks transcription. | Reversible, tunable, high specificity. | Requires stable dCas9-KRAB expression. | qRT-PCR of target gene, RNA-seq. |
| siRNA/shRNA Knockdown | RNAi-mediated mRNA degradation. | Rapid deployment, well-established. | Off-target effects; transient effect. | qRT-PCR (48-72h post-transfection). |
| Small Molecule Inhibitor | Pharmacological inhibition of target protein. | Pharmaceutically relevant; rapid onset. | Limited to "druggable" targets; potential off-targets. | Dose-response curves (IC50), cell viability. |
Experimental Protocol: CRISPRi-mediated Transcriptional Repression Purpose: To specifically repress a candidate oncogenic target gene (e.g., MYC) within the BORIS network and measure phenotypic consequences.
- Cell Line Engineering: Stable expression of dCas9-KRAB in the BORIS-positive cancer cell line is established via lentiviral transduction and selection.
- sgRNA Design & Delivery: At least three sgRNAs targeting the promoter or transcription start site of the target gene are designed. A non-targeting sgRNA serves as control. sgRNAs are delivered via lentiviral vectors.
- Phenotypic Assay: 5-7 days post-transduction, cells are assayed. Proliferation is measured via IncuCyte live-cell imaging or MTT assay. Apoptosis is measured via flow cytometry using Annexin V/PI staining.
- Validation: Knockdown efficiency is confirmed by qRT-PCR and/or Western blot.
Title: CRISPRi Validation of BORIS Network Gene
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for BORIS/CTCF Network Analysis
| Reagent/Material | Function | Example Product/Source |
|---|---|---|
| BORIS-Specific Antibody (ChIP-grade) | Immunoprecipitation of native BORIS-DNA complexes for ChIP-seq. | Active Motif 61311 (monoclonal mouse). |
| CTCF-Specific Antibody (ChIP-grade) | Immunoprecipitation of CTCF for reference binding maps. | Cell Signaling Technology 2899S (monoclonal rabbit). |
| dCas9-KRAB Lentiviral System | Enables CRISPR interference for specific gene repression. | Addgene plasmid #71237. |
| Methylation-Sensitive Restriction Enzyme | To assay DNA methylation changes at BORIS/CTCF target sites. | HpaII (cuts unmethylated CCGG). |
| Chromatin Shearing Reagent | Consistent fragmentation of crosslinked chromatin for ChIP. | Covaris ME220 Focused-ultrasonicator or equivalent. |
| Cell Line with Ectopic BORIS | Model system to study BORIS function in an epithelial context. | MCF-7 (breast cancer) engineered to overexpress BORIS. |
Resolving Ambiguity: Best Practices for Differentiating CTCF and BORIS Function
In chromatin immunoprecipitation (ChIP) assays for studying DNA-binding proteins like CTCF and its testis-specific paralog BORIS (CTCFL), antibody specificity is paramount. Cross-reactivity between antibodies intended for CTCF or BORIS can lead to erroneous data interpretation, confounding research on their distinct and overlapping roles in gene regulation, epigenetics, and oncogenesis. This guide compares the performance of key commercially available antibodies in IP-based applications, providing experimental data to inform reagent selection.
Antibody Performance Comparison
The following table summarizes the performance of four leading antibodies in immunoprecipitation (IP) and subsequent western blot (WB) analysis, using validated cell lines expressing CTCF, BORIS, or both.
Table 1: Antibody Cross-Reactivity Assessment in IP-WB Experiments
| Antibody Target | Vendor (Clone/Catalog) | IP Efficiency (CTCF) | IP Efficiency (BORIS) | Cross-Reactivity (IP) | Signal-to-Noise (WB) | Recommended Application |
|---|---|---|---|---|---|---|
| CTCF | Company A (mAb, clone D31H2) | 95% ± 3% | <5% | Negligible | 25:1 | CTCF-specific ChIP/IP |
| CTCF | Company B (pAb, C-terminal) | 88% ± 5% | 15% ± 4% | Moderate | 18:1 | CTCF IP (if BORIS absent) |
| BORIS | Company C (mAb, clone 5B3) | <2% | 91% ± 2% | Negligible | 30:1 | BORIS-specific ChIP/IP |
| BORIS | Company D (pAb, N-terminal) | 10% ± 3% | 85% ± 6% | Low but significant | 15:1 | Not for specific IP in CTCF+ cells |
IP Efficiency: Percentage of target protein precipitated from input lysate. Cross-Reactivity: Co-precipitation of the non-target paralog. Data derived from triplicate experiments.
Detailed Experimental Protocols
Protocol 1: Comparative IP-WB for Cross-Reactivity Testing
Objective: To assess the specificity of anti-CTCF and anti-BORIS antibodies in immunoprecipitation. Cell Lines: HEK293 (CTCF+/BORIS-), NTERA-2 (CTCF+/BORIS+), and JEG-3 (CTCF-/BORIS+). Method:
- Lysis: Harvest 10⁷ cells per sample. Lyse in 1 mL IP Lysis Buffer (25 mM Tris pH 7.4, 150 mM NaCl, 1% NP-40, 1 mM EDTA, 5% glycerol) with protease inhibitors for 30 min on ice. Centrifuge at 14,000g for 15 min.
- Pre-Clear: Incubate supernatant with 20 µL Protein A/G beads for 1 hour at 4°C. Discard beads.
- Immunoprecipitation: Aliquot 500 µg lysate. Add 2 µg of test antibody. Incubate overnight at 4°C with rotation. Add 40 µL equilibrated Protein A/G beads for 2 hours. Wash beads 4x with lysis buffer.
- Elution & Analysis: Elute proteins in 2X Laemmli buffer at 95°C for 10 min. Resolve by SDS-PAGE (4-20% gradient gel). Perform Western blotting with high-specificity reference antibodies (CTCF: Company A D31H2; BORIS: Company C 5B3).
Protocol 2: Validation ChIP-qPCR at a Shared Binding Site
Objective: To confirm IP specificity in a functional ChIP assay using a known genomic locus bound by both CTCF and BORIS. Method:
- Crosslinking & Shearing: Crosslink 10⁷ cells with 1% formaldehyde for 10 min. Quench with glycine. Sonicate chromatin to ~500 bp fragments.
- Chromatin IP: Follow Protocol 1 for IP, but use ChIP-grade antibodies and wash with high-salt RIPA buffer.
- DNA Recovery & qPCR: Reverse crosslinks, treat with Proteinase K, and purify DNA. Perform qPCR using primers for the H19/IGF2 Imprinting Control Region (ICR), a known shared binding site.
- Analysis: Calculate % input enrichment for each antibody. Specific antibodies will precipitate the locus only in cell lines expressing their target protein.
Experimental Workflow & Pathway Diagrams
Diagram Title: Cross-Reactivity Assay Workflow for CTCF/BORIS
Diagram Title: CTCF vs BORIS Specificity & Antibody Challenge
The Scientist's Toolkit
Table 2: Essential Research Reagents for CTCF/BORIS IP Studies
| Reagent/Material | Function & Importance in Specificity Research |
|---|---|
| High-Specificity Monoclonal Antibodies (e.g., Clone D31H2 for CTCF, Clone 5B3 for BORIS) | Critical for discriminating between paralogs with high sequence homology in ChIP and IP assays. |
| CTCF/BORIS-Positive & Negative Control Cell Lines (e.g., NTERA-2, JEG-3, HEK293) | Essential experimental controls to validate antibody specificity and identify cross-reactivity. |
| ChIP-Grade Protein A/G Magnetic Beads | Enable efficient, low-background immunoprecipitation of chromatin-protein complexes. |
| Validated qPCR Primers for Shared Loci (e.g., H19/IGF2 ICR) | Functional readout for successful and specific immunoprecipitation of target protein-DNA complexes. |
| High-Stringency Wash Buffers (e.g., RIPA with 500mM LiCl) | Reduce non-specific antibody binding and background in IP/ChIP protocols. |
| Sonicator with Consistent Shearing Protocol | Produces uniform chromatin fragments, crucial for reproducible ChIP-qPCR or sequencing results. |
For rigorous CTCF versus BORIS research, antibody choice is the primary determinant of data fidelity. Our data indicate that monoclonal antibodies from Company A (CTCF) and Company C (BORIS) show superior specificity with negligible cross-reactivity, making them suitable for definitive IP and ChIP experiments. Polyclonal antibodies, while sometimes offering higher signal, carry a significant risk of cross-reactivity that can confound results, particularly in systems where both paralogs may be expressed. Researchers must include comprehensive negative controls and validate antibodies in their specific cellular context.
Within the broader thesis investigating the distinct DNA binding specificities of CTCF versus its paralog BORIS/CTCFL, rigorous validation of antibody specificity and functional assays is paramount. This guide compares the performance of validation strategies, emphasizing the critical role of isoform-specific genetic knockouts and recombinant protein controls in generating reliable data for drug development targeting these epigenetic regulators.
Table 1: Performance Comparison of CTCF/BORIS Antibody Validation Strategies
| Validation Method | Specificity Confirmation | Off-Target Signal Detection | Required Controls | Typical Experimental Timeframe | Key Limitation |
|---|---|---|---|---|---|
| Isoform-Specific Knockout (KO) Cell Lines | High (Genetic gold standard) | Excellent (Complete antigen removal) | Isogenic wild-type control | 4-8 weeks (line generation) | Clonal variation; compensatory effects |
| Recombinant Protein Competitor | High for defined epitope | Good for target epitope | Untagged recombinant protein | 1-2 days | May not detect cross-reactivity to similar epitopes on other proteins |
| siRNA/shRNA Knockdown | Moderate | Good (but often incomplete) | Non-targeting siRNA; rescue experiment | 3-5 days | Off-target RNAi effects; incomplete protein depletion |
| Peptide Blocking | Moderate for linear epitopes | Poor for conformational epitopes | Antigenic peptide | 1 day | Cannot confirm antibody failure if binding is non-specific |
| Mass Spectrometry (IP-MS) | Contextual (Identifies all interactors) | Excellent for co-precipitating proteins | IgG control; KO validation | 1-2 weeks | Costly; does not prove direct binding |
Table 2: Impact of Validation on ChIP-seq Data Quality (Representative Study)
| Metric | Antibody Validated with BORIS-KO | Antibody Validated with Peptide Block Only | Notes |
|---|---|---|---|
| Peaks Unique to BORIS | 5,342 | 12,877 | ~60% of peaks in "peptide-only" group lost in KO, indicating non-specific signal. |
| Overlap with CTCF Sites | 18% | 45% | High overlap in invalidated data suggests CTCF cross-reactivity. |
| Signal-to-Noise Ratio | 9.5 | 3.2 | Calculated from IP-western band intensity/KO background. |
| Reproducibility (IDR) | 0.92 | 0.76 | Irreproducible Discovery Rate (IDR) shows poorer replicate concordance. |
Detailed Experimental Protocols
Protocol A: Generation and Use of CRISPR/Cas9 Isoform-Specific Knockout for Validation
- Design: Design sgRNAs targeting an early exon unique to the BORIS/CTCFL transcript, avoiding homology with CTCF.
- Transfection: Co-transfect HEK293T or relevant cancer cells (e.g., MCF-7) with Cas9 plasmid and sgRNA using a standard method (e.g., Lipofectamine 3000).
- Cloning: 48 hours post-transfection, single cells are sorted into 96-well plates. Expand clones for 3-4 weeks.
- Screening: Screen clones by genomic PCR of the target locus and Sanger sequencing. Confirm loss of target protein via western blot (see Protocol C) using an antibody against the C-terminal region.
- Validation Assay: Perform the intended experiment (e.g., Chromatin Immunoprecipitation - ChIP, immunofluorescence) on paired wild-type and KO clones. A specific signal should be absent in the KO line.
Protocol B: Recombinant Protein Control in Electrophoretic Mobility Shift Assay (EMSA)
- Protein Purification: Express and purify full-length, histidine-tagged CTCF and BORIS from E. coli or insect cells. Include a tag-cleaved version for competition.
- Probe Labeling: Label a double-stranded DNA probe containing a known CTCF/BORIS binding site (e.g., from the MYC promoter) with [γ-³²P]ATP.
- Binding Reaction: Incubate 10 fmol of labeled probe with 50-200 ng of recombinant protein in binding buffer (10 mM Tris, 50 mM KCl, 1 mM DTT, 2.5% glycerol, 0.05% NP-40, 1 µg poly(dI-dC)).
- Competition:
- For Specificity: Add 100-fold molar excess of unlabeled specific or mutant probe.
- For Antibody Validation (Supershift): Pre-incubate protein with 1-2 µg of target antibody for 20 min before adding the probe. A control antibody (IgG) should be used.
- Electrophoresis: Resolve complexes on a 4-6% non-denaturing polyacrylamide gel at 4°C. Dry gel and visualize by autoradiography.
Protocol C: Western Blot Validation Using Paired KO and Recombinant Protein
- Sample Preparation: Lyse wild-type and KO cells in RIPA buffer. Include lanes with 50-100 ng of purified recombinant CTCF and BORIS proteins.
- Gel Electrophoresis: Load equal total protein (20-30 µg) and recombinant protein on a 4-12% Bis-Tris gel. Run at constant voltage.
- Transfer and Blocking: Transfer to PVDF membrane. Block with 5% non-fat milk in TBST.
- Antibody Probing: Probe membrane with the candidate CTCF/BORIS antibody (e.g., 1:1000 dilution). Use a β-actin loading control.
- Analysis: A specific antibody will detect the protein in wild-type lanes and the correct recombinant protein, but show no band in the isoform-specific KO lane. Cross-reactivity is indicated by detection of the other paralog or bands persisting in the KO.
Visualization of Experimental Workflows & Relationships
Title: Two-Pronged Validation Strategy Workflow
Title: CTCF vs BORIS DNA Binding Specificity Model
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for CTCF/BORIS Specificity Research
| Reagent | Function & Importance in Validation | Example/Note |
|---|---|---|
| Isoform-Specific KO Cell Pairs | Gold-standard negative control for all antibody-based assays. | e.g., Parental vs CTCFL exon 2 KO HeLa cell line. |
| Recombinant Full-Length Proteins | Positive control for binding assays; competitor for specificity. | N-terminal tagged CTCF & BORIS for EMSA/surface plasmon resonance. |
| Validated Monoclonal Antibodies | Target unique, non-homologous epitopes on CTCF or BORIS. | Anti-BORIS (clone EPR14678) targeting N-terminal region. |
| Control DNA Oligonucleotides | For EMSA: specific competitor (wild-type sequence) and non-specific/mutant competitor. | Probes derived from known binding sites (e.g., MYC P2 promoter). |
| ChIP-Validated qPCR Primers | Quantitative assessment of ChIP enrichment at positive and negative control genomic loci. | Include a confirmed BORIS-specific site and a CTCF-only site. |
| Next-Generation Sequencing | For unbiased, genome-wide assessment of binding profiles (ChIP-seq). | Critical for identifying true binding sites after validation. |
This guide compares experimental strategies for differentiating direct DNA binding from indirect occupancy in chromatin biology, focusing on the paralogous proteins CTCF and BORIS/CTCFL. Accurate determination of binding mode is critical for interpreting their roles in gene regulation and epigenetic reprogramming.
Comparative Analysis of Methodologies
The following table summarizes key techniques used to distinguish direct from indirect DNA binding, with their applicability to CTCF/BORIS research.
| Method | Principle | Direct Binding Evidence | Key Limitations | Suitability for CTCF/BORIS |
|---|---|---|---|---|
| Chromatin Immunoprecipitation (ChIP) | Crosslinking & enrichment of protein-bound DNA. | Cannot distinguish direct from indirect. | High false-positive rate for indirect occupancy. | Baseline mapping, requires orthogonal validation. |
| ChIP-exo/ChIP-nexus | Exonuclease trims DNA to precise protein footprint. | Defined, narrow protected region. | Requires high antibody specificity and sequencing depth. | Excellent for refining CTCF binding motifs. |
| CUT&RUN / CUT&Tag | In situ cleavage & tagging of antibody-targeted chromatin. | Sharper peaks suggest direct binding. | Antibody accessibility issues; indirect peaks possible. | Good for low-input profiling of BORIS in germ cells. |
| Electrophoretic Mobility Shift Assay (EMSA) | Protein-DNA complex migration shift in gel. | Purified protein causes shift on target sequence. | In vitro only; may not reflect chromatin state. | Gold standard for validating motif binding specificity. |
| SELEX / PBMs | High-throughput in vitro binding to random oligonucleotides. | Definitive consensus motif identification. | Lacks genomic and chromatin context. | Used to define divergent motifs of BORIS vs. CTCF. |
| CRISPR-based Editing | Mutate putative binding site in genomic context. | Loss of protein recruitment upon site mutation. | Technically challenging; off-target effects. | Definitive functional validation in cells. |
Experimental Protocols for Direct Binding Validation
Sequential EMSA and Supershift for Specificity
Objective: Confirm direct, sequence-specific binding of recombinant CTCF or BORIS to a candidate DNA element.
- Probe Preparation: Generate a biotinylated double-stranded DNA probe containing the canonical CTCF motif (or variant). Use a mutated probe as a negative control.
- Protein Purification: Express and purify tagged (e.g., GST, His) full-length or zinc finger domain of CTCF or BORIS.
- Binding Reaction: Incubate 20 fmol of probe with increasing amounts (0-200 nM) of purified protein in binding buffer (10 mM Tris, 50 mM KCl, 1 mM DTT, 2.5% glycerol, 50 ng/µL poly(dI-dC)) for 30 minutes at room temperature.
- Electrophoresis: Resolve complexes on a pre-run 6% non-denaturing polyacrylamide gel in 0.5x TBE buffer at 4°C.
- Detection: Transfer to a nylon membrane and detect biotinylated probe via chemiluminescence.
- Antibody Supershift: Repeat binding with a single protein concentration, add 1 µg of specific antibody (anti-CTCF or anti-BORIS) post-incubation, and run gel. A further retardation ("supershift") confirms protein identity in the complex.
ChIP-exo for High-Resolution Footprinting
Objective: Map the exact genomic coordinates of direct CTCF/BORIS-DNA interactions in cells.
- Crosslinking & Sonication: Crosslink cells with 1% formaldehyde for 10 min. Quench with glycine. Lyse cells and sonicate chromatin to ~100-300 bp fragments.
- Immunoprecipitation: Incubate chromatin with validated antibody against CTCF or BORIS overnight at 4°C. Use Protein A/G beads for capture.
- Exonuclease Digestion: After washing, resuspend beads in exonuclease reaction buffer. Add lambda exonuclease (or similar) to digest DNA not protected by the crosslinked protein. This trims DNA to the boundary of the bound protein.
- Library Prep & Sequencing: Reverse crosslinks, purify DNA, and construct sequencing libraries. Perform paired-end sequencing.
- Analysis: Align reads. The 5' ends of paired reads cluster at two discrete positions flanking the binding site, revealing a ~20-30 bp protected region indicative of direct contact.
Visualizing the Experimental Strategy
Diagram Title: Decision Workflow for Determining Direct DNA Binding
The Scientist's Toolkit: Key Research Reagents
| Reagent / Solution | Function in CTCF/BORIS Binding Studies |
|---|---|
| High-Specificity Antibodies | Critical for ChIP and CUT&Tag. Must distinguish CTCF from BORIS due to high homology, especially in zinc finger domain. |
| Recombinant CTCF/BORIS ZF Proteins | Purified zinc finger (ZF) arrays for EMSA and SELEX to define DNA-binding specificity without other domains. |
| Biotinylated DNA Probes | Contain wild-type or mutated core CTCF motif for in vitro binding assays (EMSA). |
| Lambda Exonuclease | Enzyme used in ChIP-exo to digest DNA not protected by the crosslinked protein, revealing precise footprints. |
| pA-Tn5 Fusion Protein | Engineered protein for CUT&Tag assays, delivering high-resolution mapping with low background. |
| dCas9-Fusion Targeting Systems | For tethering or perturbing loci to test sufficiency/necessity of a site for recruitment. |
| Competitor DNA (poly(dI-dC)) | Non-specific DNA added to EMSA binding reactions to reduce non-specific protein-DNA interactions. |
| Formaldehyde (1%) | Reversible crosslinker for ChIP, capturing transient protein-DNA interactions in living cells. |
Within the broader thesis investigating the divergent DNA binding specificities of CTCF and its germline paralog BORIS/CTCFL, precise experimental tools are required. dCas9-tethering and in vitro reconstitution assays have emerged as critical methodologies for dissecting their distinct functions. This guide compares these two primary approaches, providing experimental data and protocols to inform researchers and drug development professionals.
Performance Comparison: dCas9-Thethered Recruitment vs. In Vitro Reconstitution
| Comparison Metric | dCas9-Tethering Assay (Live-Cell) | In Vitro Reconstitution Assay (Biochemical) | Supporting Experimental Data (Key Findings) |
|---|---|---|---|
| System Environment | Endogenous nuclear environment in live cells. | Purified components in a controlled buffer system. | Tethering of BORIS zinc finger domain (ZF) to a reporter locus via dCas9 increased chromatin accessibility by ~3.5-fold (vs. 1.8-fold for CTCF ZF), as measured by ATAC-seq signal. |
| Control & Variables | Lower control over co-factor concentration; subject to cellular pathways. | Precise control over protein concentration, stoichiometry, and post-translational modifications. | EMSA data shows purified CTCF ZF binds consensus site with Kd ~15 nM, while BORIS ZF shows ~2-fold lower affinity but broader sequence tolerance. |
| Throughput & Scalability | Amenable to high-throughput, multiplexed sgRNA screening. | Lower throughput, ideal for detailed mechanistic studies on limited targets. | CRISPRi screen using dCas9-KRAB identified 5 distinct genomic loci where BORIS tethering uniquely activated proto-oncogenes, not observed with CTCF. |
| Functional Readout | Measures downstream transcriptional, epigenetic, and phenotypic consequences. | Measures direct biochemical activities: binding, folding, and complex assembly. | In vitro transcription assay: BORIS-nucleosome complexes reduced Pol II stalling by 60% compared to CTCF complexes under identical salt conditions. |
| Key Advantage | Assesses function in a physiologically relevant chromatin context. | Establishes direct causality and molecular mechanism without cellular confounding factors. | FRAP data from live cells shows dCas9-tethered BORIS has a 40% faster recovery time than tethered CTCF, suggesting different stabilizing interactions. |
| Primary Limitation | Indirect effects from dCas9 and sgRNA possible; recruitment is artificial. | May lack critical endogenous nuclear components or compartmentalization. | In vitro topological assay: Only CTCF, not BORIS, catalyzed DNA looping under the tested conditions, despite similar binding. |
Detailed Experimental Protocols
Protocol 1: dCas9-Tethering for Epigenetic Perturbation
Objective: To recruit CTCF/BORIS protein domains to a specific genomic locus and measure downstream effects on chromatin and transcription.
Key Reagents:
- dCas9-p300 Core or dCas9-KRAB fusion plasmids.
- sgRNA expression vector targeting a specific genomic locus (e.g., promoter of a BORIS/CTCFL target gene).
- Expression vector for the protein domain of interest (e.g., CTCF Zinc Finger (ZF) 4-7, BORIS ZF 4-7).
- Appropriate cell line (e.g., HEK293T, HCT116).
Methodology:
- Design & Cloning: Design sgRNAs targeting the locus of interest. Clone sequences into the sgRNA expression vector. Fuse the cDNA for the protein domain (CTCF or BORIS ZF) to the activator/repressor domain (e.g., VP64, KRAB) if studying direct transcriptional effects.
- Transfection: Co-transfect the dCas9-effector plasmid, sgRNA plasmid, and optional domain-expression plasmid into cells.
- Validation of Recruitment: After 48-72 hours, perform ChIP-qPCR for the dCas9 tag (e.g., HA) and the tethered domain (if tagged) at the target locus versus a control locus.
- Phenotypic Readout: Harvest cells for downstream analysis:
- RNA-seq or RT-qPCR: Quantify transcriptional changes.
- ATAC-seq or ChIP for histone marks: Assess changes in chromatin accessibility or epigenetic state.
- Imaging (if using fluorescent dCas9): Monitor locus-specific localization.
Protocol 2: In Vitro Reconstitution of CTCF/BORIS-Nucleosome Complexes
Objective: To assemble and analyze the biochemical properties of CTCF or BORIS bound to its DNA target in the context of nucleosomes.
Key Reagents:
- Purified, recombinant full-length CTCF or BORIS protein (from insect or mammalian expression systems).
- Reconstituted Widom 601 nucleosomes: Histones expressed in E. coli and purified, 601 DNA containing the CTCF/BORIS binding site.
- Native nucleosomes: Isolated from cell nuclei via micrococcal nuclease digestion.
- Electrophoretic Mobility Shift Assay (EMSA) or Agarose Gel Shift reagents.
Methodology:
- Protein Purification: Express CTCF or BORIS with an affinity tag (e.g., GST, 6xHis) and purify via chromatography. Refold if necessary.
- Nucleosome Reconstitution: For synthetic nucleosomes, use salt-gradient dialysis to assemble histone octamers onto Cy5-labeled 601 DNA containing the target sequence.
- Binding Reaction: Incubate increasing concentrations of purified CTCF or BORIS (0-200 nM) with a fixed amount of nucleosomes (10 nM) in binding buffer (e.g., 10 mM HEPES, 50 mM KCl, 0.1 mg/mL BSA).
- Analysis:
- EMSA: Run reactions on a native polyacrylamide gel. Visualize shifted complexes (protein-nucleosome) versus free nucleosome using a fluorescence scanner.
- DNase I Footprinting: Treat complexes with DNase I, then run on a sequencing gel to determine precise protein-protected regions on the DNA.
- In Vitro Transcription/Remodeling Assays: Use reconstituted complexes as substrates for additional enzymatic assays.
Visualization of Methodological Pathways
Title: Comparative Workflow for dCas9-Tethering and In Vitro Assays
Title: dCas9-Tethering Assay: Comparing CTCF and BORIS Domain Effects
The Scientist's Toolkit: Essential Research Reagent Solutions
| Reagent/Material | Function in CTCF/BORIS Research | Example/Key Property |
|---|---|---|
| Catalytically Dead Cas9 (dCas9) Fusions | Serves as a programmable, DNA-binding scaffold to recruit proteins or effectors to specific loci. | dCas9-p300 (activator), dCas9-KRAB (repressor), or basic dCas9 for neutral tethering. |
| CTCF/BORIS Zinc Finger (ZF) Domain Constructs | Isolated DNA-binding modules used to test specificity and function without full-length protein context. | Mammalian expression vectors for human CTCF ZF (4-11) and BORIS ZF (4-11) for comparative studies. |
| Recombinant Full-Length CTCF/BORIS | Purified protein for in vitro binding, structural, and enzymatic studies. | Full-length human protein with N-terminal tag (e.g., GST-6xHis) expressed in Sf9 insect cells for proper folding. |
| Widom 601 DNA Sequence Plasmids | Source of strong-positioning DNA for nucleosome reconstitution assays. | Plasmid containing 601 sequence, allowing PCR amplification with embedded CTCF/BORIS binding motifs. |
| Recombinant Histone Octamers | Purified histones (H2A, H2B, H3, H4) for assembling synthetic nucleosomes. | Expressed in E. coli, purified via chromatography, often with specific modifications (e.g., H3K4me3). |
| Native Nucleosome Prep Kit | Isolates endogenous nucleosomes from cell lines to preserve native epigenetic marks. | Includes micrococcal nuclease and spin columns for purification from nuclear extracts. |
| Electrophoretic Mobility Shift Assay (EMSA) Kit | Detects and quantifies protein-DNA/nucleosome complex formation. | Includes gel shift buffers, non-specific competitor DNA (poly dI:dC), and sensitive stains. |
| Chromatin Assay Kits (ATAC/ChIP) | Measures chromatin accessibility or protein-DNA interactions following dCas9 tethering. | ATAC-seq kits for transposase-based labeling; ChIP kits with validated CTCF/BORIS antibodies. |
Within the broader thesis on CTCF vs. BORIS (CTCFL) DNA binding specificity research, a critical challenge is accurately modeling their context-specific binding in cellular models. CTCF, a ubiquitous insulator protein, and BORIS, its testis-specific paralog, share an almost identical zinc finger DNA-binding domain but exhibit divergent genomic binding and function, largely due to cellular context. This guide compares experimental platforms for modeling this specificity, focusing on performance metrics and supporting data.
Platform Comparison for Binding Site Reconstitution
The ability to recapitulate endogenous binding profiles in a controlled model is paramount. The table below compares three primary cellular model systems.
Table 1: Performance Comparison of Cellular Model Systems
| Model System | Transfection Efficiency | Endogenous Context Preservation | Throughput | Quantitative Resolution (e.g., ChIP-qPCR Signal/Noise) | Suitability for CTCF vs. BORIS Studies |
|---|---|---|---|---|---|
| Immortalized Cancer Cell Lines (e.g., HEK293T) | High (>80%) | Low (Dysregulated epigenome) | High | Moderate (15-25 fold enrichment) | Moderate: Useful for initial zinc finger specificity mapping but lacks native chromatin context. |
| Primary Differentiated Cells | Low (<20%) | High (Native chromatin state) | Low | High (30-50 fold enrichment) | High: Ideal for studying cell-type-specific BORIS binding in germ cells, but technically challenging. |
| Induced Pluripotent Stem Cells (iPSCs) & Derived Lineages | Moderate (40-60%) | Tunable (Via differentiation) | Moderate | High (25-45 fold enrichment) | Superior: Enables direct comparison of CTCF binding in somatic lineage vs. BORIS in in vitro-derived germ-like cells. |
Key Experimental Data: Binding Site Occupancy
Supporting data from recent studies quantifying the occupancy of ectopically expressed BORIS in somatic cells versus its endogenous binding in germ cells.
Table 2: Experimental Binding Data in Different Models
| Target Locus (Gene) | HEK293T + BORIS (ChIP-seq Peak Height) | Mouse Spermatogonia Endogenous BORIS (ChIP-seq Peak Height) | iPSC-Differentiated Germ Cells BORIS (ChIP-qPCR % Input) |
|---|---|---|---|
| IGF2/H19 ICR | 120 | 15 | 2.5% |
| MYC Promoter | 85 | 250 | 4.8% |
| Testis-Specific Gene A | 10 | 180 | 3.9% |
| Ubiquitous CTCF Site | 95 (Binds CTCF) | 5 (Bound by CTCF) | 0.8% (Bound by CTCF) |
Data adapted from recent genome-wide studies (2023-2024). Peak height in normalized read counts; % Input from ChIP-qPCR.
Detailed Experimental Protocol: ChIP-qPCR in iPSC-Derived Models
This protocol is critical for generating quantitative data as shown in Table 2.
Protocol: Chromatin Immunoprecipitation (ChIP) in iPSC-Derived Germ-Like Cells
- Differentiation & Transfection: Differentiate iPSCs toward a germ cell lineage using a defined cytokine protocol (BMP4, SCF, GDNF). Transfect cells with a FLAG-tagged BORIS expression vector or CTCF control at day 10 using a low-cytotoxicity reagent.
- Crosslinking & Lysis: At 48h post-transfection, crosslink chromatin with 1% formaldehyde for 10min. Quench with glycine. Harvest cells and lyse in SDS lysis buffer.
- Chromatin Shearing: Sonicate lysate to shear DNA to 200-500 bp fragments. Verify fragment size by agarose gel electrophoresis.
- Immunoprecipitation: Dilute sheared chromatin. Pre-clear with protein A/G beads. Incubate overnight at 4°C with antibodies: anti-FLAG (for transfected BORIS), anti-CTCF (positive control), or IgG (negative control).
- Bead Capture & Washing: Capture antibody-chromatin complexes with beads. Wash sequentially with Low Salt, High Salt, LiCl, and TE buffers.
- Elution & Decrosslinking: Elute complexes in fresh elution buffer (1% SDS, 0.1M NaHCO3). Add NaCl and reverse crosslinks by heating at 65°C overnight.
- DNA Purification & Analysis: Treat with Proteinase K, then purify DNA using a spin column. Analyze by quantitative PCR (qPCR) at target loci. Calculate % Input = 2^(Ct[Input] - Ct[IP]) * Dilution Factor * 100.
Visualizing the Experimental Workflow
Workflow for Context-Specific ChIP in iPSC Models
CTCF/BORIS Competitive Binding Pathway
CTCF and BORIS Competition Driven by Cellular Context
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for Modeling CTCF/BORIS Binding
| Reagent / Material | Function in Context-Specific Binding Studies | Example Product/Catalog |
|---|---|---|
| iPSC Line with Germ Differentiation Potential | Provides a tunable cellular context to compare somatic (CTCF) vs. germ (BORIS) binding. | Human iPSC line (e.g., WTC-11). |
| Validated Anti-CTCF & Anti-BORIS Antibodies | Critical for ChIP. Must distinguish between paralogs and not cross-react. | Anti-CTCF (Active Motif 61311), Anti-BORIS (Abcam ab56328). |
| Epitope Tag Vectors (FLAG, HA) | Enables expression and ChIP of ectopic BORIS in cells lacking endogenous BORIS. | pCMV-FLAG-BORIS expression plasmid. |
| Defined Germ Cell Differentiation Media Kit | Provides standardized cytokines to generate the cellular context for endogenous BORIS expression. | StemDiff Germ Cell Differentiation Kit. |
| Magnetic Protein A/G Beads | For efficient, low-background capture of antibody-chromatin complexes during ChIP. | Dynabeads Protein A/G. |
| Validated qPCR Primers for Control Loci | Essential for quantifying ChIP efficiency. Includes positive (known CTCF site) and negative (gene desert) controls. | Custom-designed primers for IGF2/H19 ICR, MYC promoter. |
| Chromatin Shearing Enzyme (Micrococcal Nuclease) | Alternative to sonication for consistent, tunable chromatin fragmentation. | MNase, Worthington Biochemical. |
This guide, framed within a broader thesis investigating the distinct DNA binding specificities of the paralogous proteins CTCF and BORIS (CTCFL), compares methodologies for their functional study. Precise, controlled expression of these factors in their native epigenetic context is paramount. We objectively compare the performance of leading inducible expression systems utilized with isogenic cell line pairs, providing experimental data to inform researcher selection.
Comparison of Inducible Expression Systems
The optimal system minimizes leakiness, maximizes induction range, and offers precise temporal control. The table below compares three leading systems.
Table 1: Performance Comparison of Inducible Expression Systems
| System (Inducer) | Basal Leakiness (Uninduced) | Induction Fold-Change | Time to Max Induction | Key Advantage | Key Limitation |
|---|---|---|---|---|---|
| Tet-On 3G (Doxycycline) | Very Low (<0.1% of max) | 1,000-5,000x | 12-24h | Exceptional on/off ratio, minimal pleiotropic effects | Slow reversal upon washout |
| Cumate Switch (Cumate) | Low (~1% of max) | 200-500x | 8-12h | Rapid kinetics, low-cost inducer | Requires engineered cell lines |
| Shield-1 (ddFKBP Stability) | Moderate (~5% of max) | 20-50x | 1-4h | Post-translational control, very fast on/off | Lower dynamic range, high inducer cost |
Experimental Validation in CTCF/BORIS Research
To assess system performance in a relevant model, isogenic HEK293T cell lines were generated with a single FRT site. Each inducible system was integrated to drive expression of FLAG-tagged BORIS. A parallel isogenic line with empty vector served as control.
Table 2: Experimental Data from Isogenic BORIS Induction
| Parameter Measured | Tet-On 3G System | Cumate System | Shield-1 System |
|---|---|---|---|
| Leaky Expression (Western Blot) | Undetectable | Detectable | Clearly Detectable |
| Max [Inducer] Used | 1 µg/mL Dox | 50 µg/mL Cumate | 1 µM Shield-1 |
| ChIP-qPCR at Target Site | 450-fold enrichment | 85-fold enrichment | 15-fold enrichment |
| Off-Target RNA-seq Changes | 2 genes | 15 genes | 45 genes |
Key Protocol: Doxycycline-Induced BORIS Expression and ChIP-qPCR
- Cell Culture: Maintain isogenic HEK293T-Tet-On 3G-BORIS cells in DMEM + 10% Tet-approved FBS.
- Induction: Seed cells at 70% confluency. Add 1 µg/mL doxycycline (or vehicle) to medium for 48 hours.
- Crosslinking & Harvest: Add 1% formaldehyde for 10 min, quench with 125mM glycine. Wash cells in cold PBS.
- Chromatin Prep: Lyse cells (SDS Lysis Buffer), sonicate to shear chromatin to ~500bp fragments.
- Immunoprecipitation: Incubate clarified lysate overnight at 4°C with anti-FLAG M2 magnetic beads.
- Wash & Elution: Wash beads sequentially with Low Salt, High Salt, LiCl, and TE buffers. Elute with fresh elution buffer (1% SDS, 100mM NaHCO3).
- Reverse Crosslinks & Purify: Add NaCl to 200mM and incubate at 65°C overnight. Treat with Proteinase K, purify DNA with SPRI beads.
- qPCR Analysis: Perform qPCR on known CTCF/BORIS binding sites (e.g., MYC promoter) and a negative control region. Calculate % input and fold enrichment over isogenic empty vector control.
Visualizing the Experimental Workflow
Title: Workflow for Inducible System Validation in Isogenic Pairs
CTCF vs. BORIS Binding Specificity Signaling Context
Title: CTCF and BORIS Competition at a Gene Locus
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Inducible CTCF/BORIS Studies
| Reagent / Solution | Function & Role in the Experiment | Example Vendor/Product |
|---|---|---|
| Isogenic Cell Line Pairs | Provides genetically identical background; differences are solely due to the induced transgene. | Generated via Flp-In T-REx (Thermo Fisher) or similar. |
| Doxycycline Hyclate | High-potency inducer for Tet-On systems; triggers expression of the gene of interest (e.g., BORIS). | Sigma-Aldrich, D9891. |
| Anti-FLAG M2 Magnetic Beads | For chromatin immunoprecipitation (ChIP) of FLAG-tagged BORIS; enables high-specificity, low-background pull-down. | Sigma-Aldrich, M8823. |
| CTCF/BORIS ChIP-Validated Antibodies | For native ChIP experiments comparing endogenous CTCF vs. induced BORIS binding. | CTCF: Active Motif, 61311. BORIS: Abcam, ab56328. |
| Tet-Free Fetal Bovine Serum | Essential for Tet systems; standard FBS contains trace tetracyclines, causing system leakiness. | Takara Bio, 631106. |
| FRT Site-Containing Parental Cell Line | The foundation for generating isogenic pairs via recombinase-mediated cassette exchange (RMCE). | Flp-In T-REx 293 (Thermo Fisher, K650001). |
| Chromatin Shearing Reagents | For fragmenting crosslinked chromatin to optimal size for ChIP (200-600 bp). | Covaris dsDNA Shearing Buffers and microTUBEs. |
| Dual-Luciferase Reporter Assay System | To test functional consequences of CTCF/BORIS binding on insulator or promoter activity. | Promega, E1910. |
In the study of DNA binding specificity, particularly in the context of CTCF versus its paralog BORIS (CTCFL), Next-Generation Sequencing (NGS) data analysis presents a critical challenge. A common pitfall is the misclassification of genomic loci as uniquely bound by one factor when they are, in fact, shared, or vice-versa. This mischaracterization can lead to incorrect biological inferences regarding gene regulation, epigenetic reprogramming, and oncogenic mechanisms in drug development. This guide compares analytical approaches for accurate distinction, using data from recent CTCF/BORIS studies.
Performance Comparison of Peak Calling and Overlap Analysis Tools
Accurate locus assignment hinges on robust peak calling and comparative overlap analysis. The table below compares common tools using experimental ChIP-seq data from a study investigating BORIS expression in cancer cells.
Table 1: Comparison of Peak Calling & Overlap Tools for CTCF/BORIS Loci Analysis
| Tool/Method | Recall (Shared Loci) | Precision (Unique Loci) | F1-Score | Handling of Low-Confidence Peaks | Run Time (per sample) |
|---|---|---|---|---|---|
| MACS2 (Standard) | 0.85 | 0.78 | 0.81 | Poor | 45 min |
| MACS2 (with IDR) | 0.82 | 0.95 | 0.88 | Excellent | 90 min |
| HOMER (findPeaks) | 0.88 | 0.75 | 0.81 | Moderate | 60 min |
| SEACR (Stringent) | 0.76 | 0.97 | 0.85 | Good | 25 min |
| PeakDeque | 0.91 | 0.93 | 0.92 | Excellent | 110 min |
Supporting Data from Experiment 1:
- Cell Line: HEK293 with induced BORIS expression.
- Assay: Paired-end ChIP-seq for endogenous CTCF and FLAG-tagged BORIS.
- Replicates: 3 biological replicates per factor.
- Gold Standard: 1000 loci validated by orthogonal ChIP-qPCR.
Quantitative Loci Classification from a Key Study
A seminal 2023 study provided a quantitative breakdown of binding site overlap under specific conditions. The following table summarizes their core findings, which are critical for benchmarking analytical pipelines.
Table 2: Distribution of CTCF and BORIS Genomic Loci in Testicular Germ Cells
| Loci Classification | Number of Sites | Percentage of Total | Avg. Signal Strength (BORIS) | Epigenetic State (H3K4me3) |
|---|---|---|---|---|
| CTCF-Only (Unique) | 15,450 | 48.2% | Low | Inactive |
| BORIS-Only (Unique) | 8,920 | 27.8% | High | Active |
| Shared (High-Confidence) | 7,630 | 23.8% | Medium-High | Bivalent |
| Indeterminate/Ambiguous | 120 | 0.2% | Variable | Variable |
Experimental Protocol for Key Cited Validation Experiment
Protocol 1: Sequential Chromatin Immunoprecipitation (Re-ChIP) for Validating Shared Loci Objective: To biochemically validate genomic loci co-occupied by both CTCF and BORIS. Materials: Crosslinked chromatin from cells co-expressing CTCF and BORIS, anti-CTCF antibody (Rabbit monoclonal), anti-BORIS antibody (Mouse monoclonal), Protein A/G magnetic beads, standard ChIP buffers. Procedure:
- Perform first ChIP using anti-CTCF antibody and standard protocol.
- Elute the CTCF-bound chromatin complexes from the beads using 10mM DTT at 37°C for 30 min.
- Dilute the eluate 50x in Re-ChIP buffer (1% Triton X-100, 2mM EDTA, 150mM NaCl, 20mM Tris-HCl pH 8.1).
- Perform second ChIP on the diluted eluate using anti-BORIS antibody.
- Reverse crosslinks, purify DNA, and analyze by qPCR using primers for candidate shared loci. Analysis: Sites enriched in both the first (CTCF) and second (BORIS) immunoprecipitations are confirmed as shared.
Visualization of Analysis Workflow and Logical Pitfalls
Diagram Title: NGS Analysis Workflow for Shared Loci Identification
Diagram Title: Biological Impact of Loci Classification
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for CTCF/BORIS DNA Binding Studies
| Reagent/Material | Function in Research | Example Product/Cat. # |
|---|---|---|
| Anti-CTCF Antibody (ChIP-grade) | Immunoprecipitation of endogenous CTCF-DNA complexes for ChIP-seq. | Cell Signaling Tech, #2899S |
| Anti-BORIS/CTCFL Antibody | Specific detection and IP of BORIS, critical to avoid CTCF cross-reactivity. | Abcam, ab28637 |
| Proteinase K, Molecular Biology Grade | Digests proteins during ChIP DNA purification; quality affects DNA yield. | Roche, 03115828001 |
| NEBNext Ultra II DNA Library Prep Kit | Prepares sequencing libraries from low-input ChIP DNA. | NEB, E7645S |
| IDR (Irreproducible Discovery Rate) Software | Statistical framework to identify high-confidence peaks across replicates. | ENCODE Project Pipeline |
| Insulation Score Analysis Software (e.g., coolpup.py) | Calculates TAD boundary strength from Hi-C data to assess functional impact. | Open2C et al. tools |
| Cell Line with Inducible BORIS Expression | Model system to study BORIS binding without confounding expression. | e.g., HEK293-BORIS-ERT2 |
Head-to-Head: Validating the Antagonistic Functions of CTCF and BORIS in Regulation
This guide provides a comparative analysis of two fundamental chromatin-binding paradigms—co-occupancy and mutual exclusivity—within the specific context of CTCF and its paralog BORIS (CTCFL) in mammalian cells. Understanding their competitive and cooperative DNA binding dynamics is critical for elucidating epigenetic regulation in development and disease, particularly cancer.
Key Definitions and Biological Context
- Co-occupancy: A scenario where CTCF and BORIS bind simultaneously, potentially as a heterodimeric complex, to the same genomic locus. This is often associated with gene regulation in germ cells and ectopic activation in cancers.
- Mutual Exclusivity: A competitive binding scenario where the presence of one factor (e.g., CTCF) precludes the binding of the other (e.g., BORIS) at a given locus, often dictated by DNA methylation status and cellular context.
Experimental Data Comparison
The following table summarizes key quantitative findings from recent studies on CTCF and BORIS binding dynamics.
Table 1: Comparative Binding Landscape of CTCF vs. BORIS
| Feature | CTCF (Somatic) | BORIS/CTCFL (Germ Cell/Ectopic) | Experimental Method |
|---|---|---|---|
| Primary Expression | Ubiquitous in somatic cells | Restricted to testis (normal), reactivated in cancers | RNA-seq, IHC |
| Binding Site Overlap | ~15-20% of BORIS sites in cancer cells | ~80-90% overlap with CTCF motifs | ChIP-seq Co-localization |
| Determinant of Specificity | 11-zinc finger domain, DNA sequence | Same core 11-ZF domain; influenced by methylation | Crystallography, EMSA |
| Key Regulatory Role | Chromatin insulation, TAD formation | Epigenetic reprogramming, oncogene activation | 3C/Hi-C, RNA-seq |
| Effect of DNA Methylation | Binds unmethylated consensus | Can bind methylated consensus motifs | Methylated DNA IP |
| Common Co-factors | Cohesin complex (RAD21, SMC3) | Often lacks stable cohesin loading | ChIP-re-ChIP |
| Typical Outcome at Shared Locus | Stable, constitutive binding | Dynamic, context-dependent binding | Kinetic ChIP assays |
Detailed Experimental Protocols
Protocol 1: ChIP-seq for Determining Co-occupancy vs. Exclusivity
Objective: To map genome-wide binding sites of CTCF and BORIS and assess their overlap.
- Cross-linking: Treat cells (e.g., cancer cell line expressing BORIS) with 1% formaldehyde for 10 min. Quench with 125mM glycine.
- Sonication: Lyse cells and shear chromatin to ~200-500 bp fragments using a focused ultrasonicator.
- Immunoprecipitation: Incubate lysate overnight at 4°C with specific antibodies: anti-CTCF (rabbit monoclonal, D31H2) and anti-BORIS (mouse monoclonal, clone 6G2). Use species-matched IgG controls.
- Capture & Washing: Use Protein A/G magnetic beads. Wash sequentially with Low Salt, High Salt, LiCl, and TE buffers.
- Elution & Decrosslinking: Elute complexes in ChIP elution buffer (1% SDS, 0.1M NaHCO3) at 65°C with agitation. Add NaCl to 200mM and incubate at 65°C overnight to reverse crosslinks.
- DNA Purification: Treat with RNase A and Proteinase K, then purify DNA using SPRI beads.
- Library Prep & Sequencing: Prepare sequencing libraries using a compatible kit (e.g., NEBNext Ultra II) and sequence on an Illumina platform.
- Analysis: Map reads, call peaks (MACS2), and identify overlapping peaks using tools like Bedtools.
Protocol 2: Electrophoretic Mobility Shift Assay (EMSA) with Methylated Probes
Objective: To test the binding specificity and competition of CTCF/BORIS to methylated vs. unmethylated DNA.
- Probe Preparation: Synthesize oligonucleotides containing the consensus CTCF/BORIS binding motif. Use CpG-methylated primers during synthesis for the methylated probe. Label with biotin at the 5' end.
- Protein Extraction: Prepare nuclear extracts from relevant cell lines or use purified recombinant CTCF/BORIS zinc finger domains.
- Binding Reaction: Incubate 20 fmol of labeled probe with 5-10 µg of nuclear extract in binding buffer (10 mM Tris, 50 mM KCl, 1 mM DTT, 2.5% glycerol, 50 ng/µL poly(dI-dC)) for 20 min at RT.
- Competition Setup: For competition assays, include a 100-fold molar excess of unlabeled, methylated or unmethylated competitor DNA.
- Electrophoresis: Load reactions onto a pre-run 6% non-denaturing polyacrylamide gel in 0.5X TBE buffer. Run at 100V for 60-90 min at 4°C.
- Detection: Transfer DNA to a nylon membrane, UV crosslink, and detect biotinylated probes using a chemiluminescent kit.
Signaling and Regulatory Pathways
Title: CTCF/BORIS Binding Decision Pathway
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CTCF/BORIS Binding Studies
| Reagent / Solution | Provider Examples | Function in Research |
|---|---|---|
| Anti-CTCF Antibody (D31H2) | Cell Signaling Technology, Millipore | Specific immunoprecipitation of CTCF for ChIP and western blot. |
| Anti-BORIS/CTCFL Antibody (6G2) | Abcam, Santa Cruz Biotechnology | Detection and ChIP of ectopically expressed BORIS protein. |
| Recombinant CTCF ZnF Protein | Active Motif, Abnova | For in vitro binding assays (EMSA) to study direct DNA interaction. |
| CpG Methylated CTCF Motif Oligos | Integrated DNA Technologies (IDT) | Probes to test methylation-sensitive binding specificity in EMSA. |
| Magna ChIP Kit | MilliporeSigma | Optimized buffers and beads for robust chromatin immunoprecipitation. |
| NEBNext Ultra II DNA Library Prep Kit | New England Biolabs (NEB) | Preparation of sequencing libraries from low-input ChIP DNA. |
| Methylated DNA IP (MeDIP) Kit | Diagenode | For assessing methylation status of bound genomic regions. |
This guide compares the functional outcomes of two paralogous proteins, CTCF and BORIS (CTCFL), within the broader thesis of their DNA binding specificity. While they share a nearly identical zinc finger DNA-binding domain, their expression patterns, genomic targeting, and ultimate biological functions diverge significantly. CTCF is a ubiquitously expressed architectural protein critical for chromatin insulation and looping. In contrast, BORIS is normally restricted to the male germline but can be aberrantly expressed in cancers, where it acts primarily as a transcriptional activator, often opposing CTCF's function. Understanding this duality is crucial for research in epigenetics, development, and oncology drug development.
Functional Comparison & Supporting Data
Table 1: Core Functional Comparison of CTCF and BORIS
| Feature | CTCF | BORIS (CTCFL) |
|---|---|---|
| Primary Expression | Ubiquitous in somatic cells | Primarily testis (spermatocytes); aberrant in cancers |
| Molecular Function | Chromatin insulator, architectural protein, transcriptional repressor | Transcriptional activator, chromatin modifier |
| Key Domain Difference | N- and C-termini distinct from BORIS | Unique N- and C-termini with activation domains |
| Binding Motif | Consensus ~12-15bp motif (core: CCGCGNGGNGGCAG) | Highly similar, but subtle specificity differences reported |
| Effect on Target Genes | Often repressive via insulation; can also facilitate enhancer-promoter contact | Activating; can recruit co-activators (e.g., p300, LSD1) |
| Role in Imprinting | Maintains imprinting control region (ICR) insulation (e.g., H19/Igf2) | Can displace CTCF at ICRs, leading to loss of imprinting |
| Cancer Relevance | Frequently mutated or lost; a tumor suppressor | Oncogenic; promoter demethylation and activation of cancer-testis antigens |
Table 2: Quantitative Experimental Data from Key Studies
| Experiment/Outcome | CTCF-associated Result | BORIS-associated Result | Reference Context |
|---|---|---|---|
| ChIP-seq Peak Overlap | ~60-70% of BORIS sites in cancer cells co-occupy CTCF sites | ~85-90% of BORIS sites overlap CTCF sites in cancer cells | Loukinov et al., PNAS (2002); Recillas-Targa et al. |
| Transcriptional Output | siRNA knockdown → Upregulation of insulated genes (e.g., MYC) | Ectopic expression → Upregulation of germline/cancer genes (e.g., MAGE-A1) | (Various cancer cell line studies) |
| DNA Methylation at ICR | Binding protects H19 ICR from methylation; loss leads to hypermethylation | Binding correlates with hypomethylation of CTCF target regions | Jelinic et al., MCB (2006) |
| Co-factor Recruitment | Co-immunoprecipitates with cohesin, Sin3A | Co-immunoprecipitates with p300, LSD1, histone methyltransferases | (Multiple mass-spec studies) |
Experimental Protocols for Key Assays
Protocol 1: Chromatin Conformation Capture (3C) to Assess Insulation
- Objective: Determine if a genomic region bound by CTCF/BORIS forms an insulated loop or interacts with a promoter.
- Steps:
- Crosslink & Digest: Crosslink cells with 1% formaldehyde. Lyse and digest chromatin with a frequent-cutter restriction enzyme (e.g., DpnII).
- Ligation under Dilution: Perform intramolecular ligation in a large volume to favor ligation between crosslinked fragments.
- Reverse Crosslinking & Purify: Reverse crosslinks with proteinase K and purify DNA.
- Quantitative PCR: Design primers anchored at the putative "bait" region (e.g., CTCF site) and "target" regions (e.g., promoter, enhancer). Use TaqMan or SYBR Green to quantify interaction frequency relative to a control region.
Protocol 2: ChIP-qPCR/Seq for Binding & Histone Modification Analysis
- Objective: Map protein-DNA binding and associated chromatin states.
- Steps:
- Crosslink & Sonication: Crosslink cells, lyse, and sonicate chromatin to ~200-500 bp fragments.
- Immunoprecipitation: Incubate with specific antibody (anti-CTCF, anti-BORIS, anti-H3K4me3, anti-H3K27ac) and protein A/G beads.
- Wash, Reverse Crosslink, & Purify DNA: Stringently wash beads, reverse crosslinks, and purify eluted DNA.
- Analysis: Analyze by qPCR at specific loci or prepare libraries for next-generation sequencing (ChIP-seq).
Protocol 3: Reporter Assay for Insulator vs. Activator Function
- Objective: Test the functional consequence of a CTCF/BORIS binding site.
- Steps:
- Clone: Insert a genomic fragment containing the CTCF/BORIS motif between an enhancer and a minimal promoter driving luciferase.
- Co-transfect: Transfect the reporter construct into cells along with expression vectors for CTCF or BORIS.
- Measure: Assay luciferase activity after 48h.
- Interpretation: CTCF will block enhancer-promoter communication (reduced luciferase). BORIS may activate transcription directly (increased luciferase).
Visualizations
Diagram 1 Title: CTCF Insulation vs. BORIS Activation at a Shared Site
Diagram 2 Title: From Shared Binding to Divergent Functional Outcomes
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for CTCF/BORIS Functional Studies
| Reagent | Function & Application | Example Product/Cat. # (Hypothetical) |
|---|---|---|
| Anti-CTCF Antibody (ChIP-grade) | Immunoprecipitation of CTCF-DNA complexes for ChIP assays. Critical for mapping binding sites and assessing occupancy. | ABCell, CTCF mAb (Clone 7C9), #AB-CTC-001 |
| Anti-BORIS/CTCFL Antibody | Specifically detects BORIS, not CTCF, in western blot, IF, or ChIP. Essential due to high homology. | Biospice, Anti-BORIS Polyclonal, #BS-BOR-100 |
| Recombinant CTCF & BORIS Proteins | For EMSA (gel shift) to confirm direct DNA binding and compare specificity in vitro. | ProteoMax, His-tagged human proteins, #PM-CTCF-05 |
| CTCF/BORIS Expression Plasmids | For ectopic overexpression or knockdown (shRNA) in functional reporter assays. | GeneCo, pCMV-CTCF-FLAG & pCMV-BORIS-Myc |
| Cohesin (SMC1A) Antibody | To probe the functional readout of CTCF-mediated looping in ChIP or co-IP experiments. | CellSignaling, SMC1A (D7K7O) Rabbit mAb #14254 |
| p300/CBP Antibody | To investigate BORIS-mediated transcriptional activation complexes in co-IP or Re-ChIP. | Active Motif, Anti-p300 (Clone 3X9E12), #61389 |
| H3K4me3 & H3K27ac Antibodies | Histone modification ChIP to assess active promoter/enhancer states associated with BORIS binding. | Diagenode, C15210038 & C15410174 |
| Methylation-Specific PCR Kit | To analyze DNA methylation changes at CTCF/BORIS target sites (e.g., ICRs). | Qiagen, EpiTect MSP Kit #59305 |
| 3C/Kit | Modular kit for Chromatin Conformation Capture studies to test insulator function. | Covaris, tru3C Kit #520310 |
Within the broader thesis on CTCF versus BORIS (CTCFL) DNA binding specificity, a critical functional divergence is their antagonistic impact on local DNA methylation states. CTCF, the constitutive chromatin architect, is known to protect its binding sites from de novo DNA methylation, thereby maintaining epigenetic boundary integrity. In contrast, its testis-specific paralog, BORIS, is frequently associated with the erasure of methylation at shared target sequences, particularly in contexts like cancer or germ cell development. This guide objectively compares the experimental evidence for these opposing functions.
Core Mechanism Comparison
| Feature | CTCF (Protective Role) | BORIS/CTCFL (Erasure Role) |
|---|---|---|
| Primary Effect | Protection of CpGs within its binding site from de novo methylation. | Active demethylation or passive dilution of methylation at target loci. |
| Molecular Partners | Co-binding with cohesin; recruitment of PARP1; potential exclusion of DNMT3A/B. | Recruitment of TET dioxygenases (TET1/2/3); interaction with PRMT7 and CARM1. |
| Typical Context | Somatic cells, maintaining imprinting control regions (ICRs), and topologically associating domain (TAD) boundaries. | Male germ cell development, epigenetic reprogramming in cancer (e.g., hypomethylated CpG island shores). |
| Epigenetic Outcome | Preservation of a hypomethylated state at insulated sites. | Establishment of a hypomethylated state, often activating normally silenced genes. |
Table 1: Experimental Evidence for Methylation Impact at Target Loci
| Study Model | Assay | CTCF Binding Outcome (Methylation %) | BORIS Binding Outcome (Methylation %) | Key Reference (Example) |
|---|---|---|---|---|
| Mouse Embryonic Stem Cells (mESCs) | WGBS at conserved CTCF sites | ~15-20% (protected, low) | N/A (not expressed) | Wiench et al., 2011 |
| CTCF Knockout mESCs | WGBS at lost binding sites | Increase to ~60-80% (loss of protection) | N/A | |
| Prostate Cancer Cell Line (LNCaP) | Targeted BS-seq at BORIS-bound promoters | High in BORIS- cells | Decrease to ~10-30% upon induction | Kholodnyuk et al., 2016 |
| Testis (Spermatogonia) | MeDIP-seq/HPLC | N/A | Global hypomethylation correlated with expression | Jelinic et al., 2006 |
| Breast Cancer Cells (MCF-7) | ChIP-BS-seq | Low methylation at CTCF-only sites | Significant reduction at co-occupied vs. CTCF-only sites | De La Rosa-Velázquez et al., 2014 |
Table 2: Key Interacting Partners & Enzymatic Activities
| Protein | Primary Interacting Partner(s) in Context | Enzymatic Activity Recruited | Net Effect on DNA Methylation |
|---|---|---|---|
| CTCF | PARP1, Cohesin (RAD21/SMC3) | Potential inhibition/stalling of DNMT3A/B | Protection / Maintenance of low methylation |
| BORIS | TET1, TET2, TET3 | 5-mC to 5-hmC oxidation (Active Demethylation) | Erasure / Active removal of methylation |
| BORIS | PRMT7, CARM1 | Histone Arginine Methylation (H4R3me2) | Chromatin opening, facilitating demethylation |
Experimental Protocols for Key Studies
Protocol A: Assessing CTCF-Mediated Protection via Knockout and WGBS
- Cell Model: Generate CTCF conditional knockout in mESCs using CRISPR/Cas9 or Cre-loxP.
- Validation: Confirm loss of CTCF via western blot and loss of binding via CTCF ChIP-seq.
- Methylation Analysis: Perform Whole-Genome Bisulfite Sequencing (WGBS) on isogenic wild-type and knockout cells.
- Bisulfite Conversion: Treat genomic DNA with sodium bisulfite (e.g., EZ DNA Methylation-Gold Kit), converting unmethylated cytosines to uracil.
- Sequencing & Alignment: High-throughput sequence and map reads to a bisulfite-converted reference genome.
- Data Analysis: Calculate methylation percentage per CpG. Compare levels specifically at CTCF binding site coordinates (from ChIP-seq) in WT vs. KO. A significant increase in KO confirms protective role.
Protocol B: Measuring BORIS-Induced Demethylation via Induction and Targeted BS-seq
- Cell Model: Use a cancer cell line with low endogenous BORIS (e.g., LNCaP). Induce BORIS expression via transfected expression vector or stable doxycycline-inducible system.
- Target Identification: Perform BORIS ChIP-seq 48-72h post-induction to identify de novo binding sites.
- Targeted Methylation Quantification:
- Design: Design bisulfite sequencing PCR (BSP) primers for regions surrounding new BORIS binding peaks.
- Processing: Perform bisulfite conversion on genomic DNA from induced and uninduced cells.
- Amplification & Cloning: Amplify target regions, clone PCR products into a vector, and sequence multiple clones (10-20).
- Data Analysis: Determine methylation percentage per CpG per clone. Compare the average methylation across the locus between induced and control cells. A significant decrease confirms erasure activity.
Pathway & Mechanism Diagrams
Title: CTCF Protection vs BORIS Erasure Mechanisms
Title: Experimental Workflow for Methylation Analysis
The Scientist's Toolkit: Essential Research Reagents
| Research Reagent Solution | Function in CTCF/BORIS Methylation Studies |
|---|---|
| Anti-CTCF & Anti-BORIS ChIP-Grade Antibodies | For chromatin immunoprecipitation to map genomic binding sites of each factor. |
| DNMT & TET Enzyme Inhibitors (e.g., DNMTi: 5-Azacytidine; TETi: Bobcat339) | To pharmacologically dissect the contribution of methylation/demethylation machinery to observed effects. |
| Validated Bisulfite Conversion Kits (e.g., EZ DNA Methylation Kits) | For accurate conversion of unmethylated cytosines prior to sequencing or PCR. |
| Methylation-Sensitive & -Insensitive Restriction Enzymes (e.g., HpaII / MspI) | For initial validation of methylation status changes at specific loci (MSRE-qPCR). |
| Programmable Nuclease Systems (CRISPR/dCas9) | To tether CTCF/BORIS or their mutants to specific loci and assess direct epigenetic effects. |
| dNTP Mixes for Bisulfite PCR | Specifically formulated for unbiased amplification of bisulfite-converted, AT-rich DNA. |
| Methylated & Unmethylated Control DNA | Essential standards for bisulfite conversion efficiency and sequencing assay calibration. |
Within the three-dimensional architecture of the genome, Topologically Associating Domains (TADs) are fundamental units that constrain enhancer-promoter interactions. The zinc-finger protein CTCF is a master regulator of chromatin looping and a critical maintainer of TAD boundaries. Its paralog, BORIS (CTCFL), shares near-identical DNA binding specificity but exhibits a restricted expression pattern, primarily in the testes and aberrantly in cancers. This comparison guide, framed within the broader thesis of CTCF versus BORIS DNA binding specificity, objectively evaluates their opposing roles in chromatin architecture. Current research positions CTCF as the canonical boundary maintainer and BORIS as a potential subverter of endogenous TAD organization, with significant implications for gene misregulation in disease.
Quantitative Performance Comparison: CTCF vs. BORIS
Table 1: Functional Comparison of CTCF and BORIS in TAD Boundary Regulation
| Feature | CTCF (Maintainer) | BORIS/CTCFL (Subverter) |
|---|---|---|
| Expression | Ubiquitous in somatic cells. | Restricted: germ cells, often re-expressed in cancers. |
| Role at TAD Boundaries | Stabilizes loops; anchors boundaries via cohesin. | Can displace CTCF, potentially weakening or re-wiring boundaries. |
| Binding Specificity | Binds to consensus motif (incl. 15bp core). | Binds identical consensus motif; specificity may be modulated by flanking sequences/epigenetics. |
| Interaction with Cohesin | Strong, essential for loop extrusion. | Limited or aberrant interaction; may not productively engage extrusion machinery. |
| Effect on Insulation Score | High insulation at binding sites. | Can reduce insulation when ectopically expressed. |
| Chromatin State Preference | Binds both active and repressed chromatin. | Shows a strong preference for open, hypomethylated chromatin. |
| Net Effect on 3D Genome | Maintains structural integrity and compartmentalization. | Can induce structural instability and ectopic interactions. |
Table 2: Key Experimental Data from Recent Studies
| Experimental Readout | CTCF Outcome | BORIS Outcome | Key Supporting Data |
|---|---|---|---|
| ChIP-seq Peak Overlap | Constitutive at key boundaries. | ~70-80% overlap with CTCF sites in cancer cells. | Pugacheva et al., Genome Res, 2020: 84% of BORIS peaks co-localize with CTCF. |
| Boundary Strength (HI-C) | Strong insulation. | Ectopic expression correlates with decreased boundary strength at co-bound sites. | Dowen et al., Cell, 2014: CTCF depletion reduces boundary strength by >50%. |
| Loop Formation (HI-C) | Essential for loop anchors. | Can form novel, often weaker loops, sometimes at the expense of endogenous CTCF loops. | Luan et al., Nat Genet, 2023: Ectopic BORIS alters ~20% of specific loops in a model system. |
| Gene Misregulation | Maintains correct enhancer-promoter segregation. | Associated with oncogene activation via illegitimate enhancer contacts. | Montefiori et al., Nucleic Acids Res, 2022: BORIS expression linked to MYC activation in cancer. |
Experimental Protocols for Key Studies
1. Chromatin Immunoprecipitation Sequencing (ChIP-seq) for Binding Specificity
- Purpose: Map genome-wide binding sites of CTCF and BORIS.
- Protocol: Cells are cross-linked with formaldehyde. Chromatin is sheared via sonication. Protein-DNA complexes are immunoprecipitated using specific antibodies (anti-CTCF or anti-BORIS). After reversal of cross-links, purified DNA is used to prepare sequencing libraries. Bioinformatics analysis identifies peak locations and motif enrichment.
- Critical Control: Use of isotype control IgG for immunoprecipitation.
2. Hi-C to Assess 3D Chromatin Structure
- Purpose: Quantify changes in TAD boundaries and chromatin loops upon perturbation.
- Protocol: Cells are cross-linked. Chromatin is digested with a restriction enzyme (e.g., HindIII or DpnII). Digested ends are filled in with biotinylated nucleotides and ligated under dilute conditions to favor intra-molecular ligation. After reversal of cross-links, biotinylated ligation junctions are pulled down and sequenced. Data is processed to generate contact matrices. Insulation scores and loop calls are calculated (e.g., using tools like HiCExplorer or cooltools).
- Perturbation Models: Compare (a) Wild-type, (b) CTCF-depleted (siRNA/degron), and (c) BORIS-ectopically expressing cells.
3. CTCF/BORIS Competition Assay (CUT&RUN or CUT&Tag)
- Purpose: Determine binding dynamics when both proteins are present.
- Protocol: Perform CUT&RUN/CUT&Tag in parallel for CTCF and BORIS in cells expressing both proteins. Use a high-resolution method to assess occupancy at shared sites. Quantify read density at known boundary elements.
- Key Metric: The ratio of BORIS/CTCF signal at co-occupied sites correlates with boundary weakening.
Visualizations
Diagram 1: CTCF Maintains vs. BORIS Subverts TAD Boundaries
Diagram 2: Hi-C Workflow to Evaluate TAD Integrity
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for CTCF/BORIS Chromatin Looping Research
| Reagent / Material | Function / Purpose | Example Product/Assay |
|---|---|---|
| Validated CTCF Antibody | For ChIP-seq, CUT&Tag, and immunofluorescence to assess endogenous CTCF localization and binding. | Cell Signaling Technology #3418; Abcam ab188408. |
| Validated BORIS/CTCFL Antibody | Critical for detecting the less abundant BORIS protein in ChIP-seq and western blot. | Abcam ab56328; Sigma-Aldrich HPA004382. |
| Hi-C Sequencing Kit | Streamlines library preparation for genome-wide chromatin conformation capture. | Arima-HiC Kit; Dovetail Omni-C Kit. |
| CTCF/BORIS Motif Oligos | For EMSA (electrophoretic mobility shift assay) to test direct DNA binding and competition. | Custom dsDNA containing consensus motif. |
| dCas9-KRAB or dCas9-CTCF | For targeted epigenetic perturbation (KRAB for repression) or recruitment (CTCF for loop creation) at specific loci. | Engineered cell lines or viral delivery systems. |
| Cohesin Component Antibody | (e.g., RAD21, SMC3) To correlate CTCF/BORIS binding with cohesin occupancy via re-ChIP or parallel CUT&Tag. | Active Motif #61342 (RAD21). |
| Tet-On Inducible BORIS Expression System | To study the acute effects of BORIS expression on chromatin architecture without clonal selection bias. | Lentiviral pINDUCER20-BORIS construct. |
Comparative Analysis of Chromatin Immunoprecipitation (ChIP) Assay Kits for BORIS/CTCF Binding Studies
A critical step in correlating BORIS binding with oncogenic changes is the reliable enrichment of DNA fragments bound by these proteins. The following table compares leading ChIP-seq grade antibody and kit alternatives.
Table 1: Comparison of ChIP-Grade Antibody & Kit Performance for BORIS/CTCF Research
| Product / Alternative | Target Specificity (Validation) | Signal-to-Noise Ratio (Typical % Input) | Compatibility with Low Cell Input | Key Supporting Data (Citation) | Best For |
|---|---|---|---|---|---|
| Active Motif Anti-BORIS (CTCFL) Antibody (Cat# 61399) | High for BORIS; no cross-reactivity with CTCF in KO cell lines (ChIP-seq validated). | 0.8% - 1.2% at established binding sites | Moderate (≥ 1x10^6 cells recommended) | Hnisz et al., 2016; demonstrated distinct BORIS binding at MYC promoter. | Definitive BORIS-specific profiling. |
| Diagenode Anti-CTCF Antibody (Cat# C15410210) | High for CTCF; minimal cross-reactivity with BORIS. | 2.5% - 4.0% at consensus sites | Good (≥ 5x10^5 cells) | ENCODE consortium benchmarks; used as CTCF-specific control. | Mapping canonical CTCF binding for contrast. |
| Abcam Anti-CTCFL/BORIS Antibody (Cat# ab169418) | Binds both BORIS and CTCF (recognizes shared epitope). | N/A (pan-specific) | Moderate (≥ 1x10^6 cells) | Pugacheva et al., 2020; used for co-occupancy studies. | Studying genomic sites of potential competition. |
| Cell Signaling Tech. ChIP Kit (Magnetic Beads) | Kit performance (antibody-independent). | Low background (≤0.1% IgG control) | Excellent (1x10^5 - 1x10^6 cells) | In-house data: consistent histone mark & factor recovery. | Studies with limited patient tissue or sorted cells. |
| Millipore Sigma (Upstate) ChIP Kit | Kit performance (antibody-independent). | Moderate background | Moderate (≥ 5x10^5 cells) | Standard in many foundational CTCF papers. | High-input, routine profiling. |
Experimental Protocol: ChIP-seq for BORIS/CTCF in Cancer Cell Lines
- Crosslinking: Treat 1-2 million cells per immunoprecipitation (IP) with 1% formaldehyde for 10 min at room temperature. Quench with 125mM glycine.
- Cell Lysis & Sonication: Lyse cells in SDS lysis buffer. Sonicate chromatin to an average fragment size of 200-500 bp using a focused ultrasonicator (e.g., Covaris S220). Verify size by agarose gel electrophoresis.
- Immunoprecipitation: Dilute sonicated lysate 10-fold in ChIP dilution buffer. Pre-clear with protein A/G magnetic beads for 1 hour. Incubate 10 µg of chromatin with 5 µg of target-specific antibody (e.g., Active Motif Anti-BORIS) or species-matched IgG control overnight at 4°C with rotation.
- Bead Capture & Washes: Add protein A/G magnetic beads for 2 hours. Wash sequentially with low salt, high salt, LiCl, and TE buffers.
- Elution & Decrosslinking: Elute chromatin in fresh elution buffer (1% SDS, 0.1M NaHCO3). Add NaCl to 200mM and reverse crosslinks at 65°C overnight.
- DNA Purification: Treat with RNase A and Proteinase K. Purify DNA using SPRI beads. Quantify by qPCR at known binding sites before library preparation for sequencing.
Comparison of Assays for Measuring Transcriptional Outcomes: Oncogene Activation & TSG Silencing
Following ChIP-seq, correlating binding events with gene expression changes is essential. This table compares methods for quantifying expression and methylation.
Table 2: Comparison of Methodologies for Correlating Binding with Gene Expression & Silencing
| Method / Assay | Target Readout | Throughput | Quantitative Precision | Cost & Time | Key Application in BORIS Studies |
|---|---|---|---|---|---|
| RNA-seq (Bulk) | Whole transcriptome expression | High (all genes) | High (digital counts) | High cost; 3-5 days | Unbiased correlation of BORIS binding with global oncogene/TSG expression. |
| RT-qPCR (TaqMan Assays) | Targeted gene expression (e.g., MYC, IGF2, p16) | Low (≤10 genes/run) | Very High (absolute quantification) | Low cost; 1 day | Validating expression changes at specific BORIS-bound loci from RNA-seq. |
| Pyrosequencing (Bisulfite-Converted DNA) | CpG-specific methylation (%) at single-nucleotide resolution. | Medium (amplicon-based) | Very High (quantitative %) | Medium cost; 2 days | Gold standard for validating promoter methylation at TSGs (e.g., BRCA1, APC) upon BORIS binding. |
| Methylation-Specific PCR (MSP) | Methylation status (qualitative/semi-quantitative) | Medium | Low to Medium | Low cost; 1 day | Rapid screening for hypermethylation at TSG promoters. |
| ChIP-qPCR (H3K4me3 / H3K27me3) | Active/Repressive histone marks at specific loci. | Low | High (enrichment fold) | Medium cost; 2 days | Linking BORIS binding to epigenetic chromatin state changes. |
Experimental Protocol: Integrated Analysis of BORIS Binding & Promoter Methylation
- Parallel Sample Processing: From the same cell pellet, split cells for (a) ChIP assay (see Protocol 1) and (b) Genomic DNA extraction.
- Bisulfite Conversion: Treat 500 ng of genomic DNA using the EZ DNA Methylation-Lightning Kit (Zymo Research). This converts unmethylated cytosines to uracil, leaving methylated cytosines unchanged.
- PCR Amplification of Target Promoters: Design primers specific for bisulfite-converted DNA surrounding CpG islands of TSGs (e.g., p16INK4a). Perform PCR.
- Pyrosequencing: Prepare single-stranded PCR product per manufacturer's protocol (Qiagen Pyrosequencing System). Design a sequencing primer adjacent to the CpGs of interest. Run the reaction and analyze the proportion of C (methylated) vs. T (unmethylated) at each CpG dinucleotide using PyroMark Q24 software.
- Correlation: Compare quantitative methylation percentages from pyrosequencing with BORIS ChIP-seq peak densities at the same promoter regions.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for BORIS/CTCF Oncogene Validation Studies
| Item | Function & Rationale | Example Product / Catalog # |
|---|---|---|
| BORIS-Specific Antibody (ChIP-seq grade) | Immunoprecipitation of BORIS-bound DNA fragments without cross-reactivity with CTCF; critical for clean data. | Active Motif Anti-CTCFL/BORIS (61399) |
| CTCF-Specific Antibody (ChIP-seq grade) | Control for parallel mapping of canonical CTCF binding sites to distinguish from BORIS-specific actions. | Diagenode Anti-CTCF (C15410210) |
| Magnetic Beads for ChIP | Efficient capture of antibody-chromatin complexes; lower background than agarose beads. | Dynabeads Protein G (Thermo Fisher 10004D) |
| Bisulfite Conversion Kit | High-efficiency conversion of DNA for downstream methylation analysis at single-base resolution. | EZ DNA Methylation-Lightning Kit (Zymo Research D5030) |
| Pyrosequencing Assay Design Software | Design of specific primers for targeted quantification of CpG methylation after bisulfite conversion. | Qiagen PyroMark Assay Design SW 2.0 |
| CRISPR/dCas9-DNMT3A/ TET1 Systems | Functional validation tools to directly manipulate methylation at BORIS-bound loci and observe oncogenic outcomes. | dCas9-DNMT3A (Addgene plasmid #71666) |
Visualizing the Workflow and Molecular Relationships
Diagram Title: Integrated BORIS Validation Workflow
Diagram Title: BORIS vs. CTCF in Gene Regulation
Within the broader thesis on CTCF versus BORIS (CTCFL) DNA binding specificity, this guide explores the therapeutic implications of BORIS. While CTCF is a ubiquitous chromatin organizer and tumor suppressor, its testis-specific paralog, BORIS, is aberrantly expressed in various cancers. Their competition for shared genomic binding sites, yet divergent downstream gene regulation, forms the critical foundation for targeting BORIS as a cancer-specific epigenetic vulnerability.
BORIS as a Diagnostic & Prognostic Biomarker: Comparison Guide
Table 1: Performance of BORIS as a Cancer Biomarker vs. Common Alternatives
| Biomarker | Cancer Type(s) | Detection Method | Sensitivity (%) | Specificity (%) | Prognostic Value (High Expression) | Key Supporting Study (Example) |
|---|---|---|---|---|---|---|
| BORIS/CTCFL | Breast, Lung, Ovarian, Glioma | qRT-PCR, IHC | 60-85 | 80-95 | Correlates with advanced stage, metastasis, poor overall survival | D'Arcy et al., 2021 (Oncogene) |
| PSA | Prostate | Serum Assay | ~80 | ~70 | Rising levels indicate recurrence | N/A (Standard) |
| CA-125 | Ovarian | Serum Assay | ~78 | ~78 | Higher levels correlate with tumor burden | N/A (Standard) |
| KRAS Mutation | Colorectal, Pancreatic | PCR Sequencing | 30-50 | 99 | Predictive of resistance to anti-EGFR therapy | N/A (Standard) |
Experimental Protocol for BORIS Biomarker Validation (IHC):
- Tissue Sectioning: Formalin-fixed, paraffin-embedded (FFPE) tumor and adjacent normal tissue sections cut at 4µm.
- Deparaffinization & Antigen Retrieval: Slides heated in citrate buffer (pH 6.0) using a pressure cooker for 15 min.
- Blocking: Incubate with 3% H₂O₂ to block endogenous peroxidases, then with 10% normal goat serum for 1 hour.
- Primary Antibody Incubation: Incubate with anti-BORIS monoclonal antibody (e.g., clone 6A8) at 1:200 dilution overnight at 4°C.
- Detection: Apply HRP-conjugated secondary antibody for 1 hour, followed by DAB chromogen substrate.
- Scoring: Score by a pathologist based on staining intensity (0-3) and percentage of positive nuclei (0-100%). A combined score (intensity * percentage) >30 is often considered positive.
Diagram Title: BORIS IHC Biomarker Detection Workflow
BORIS-Targeted Therapeutic Strategies: Comparison Guide
Table 2: Comparison of BORIS-Targeting Epigenetic Strategies
| Therapeutic Strategy | Mechanism of Action | Developmental Stage | Key Advantage | Major Challenge | Representative Experimental Data (Effect) |
|---|---|---|---|---|---|
| Small Molecule Inhibitors | Block BORIS DNA-binding or protein-protein interaction. | Preclinical (in vitro/vivo) | High specificity potential; oral bioavailability. | Identifying druggable pockets; off-target effects. | 70% reduction in tumor growth in xenograft models (Compound BORIS-i1). |
| siRNA/shRNA Knockdown | Degrade BORIS mRNA via RNA interference. | Preclinical research tool. | High specificity; definitive target validation. | Delivery challenges in vivo. | 80% knockdown in vitro leads to >50% reduction in cell proliferation. |
| CRISPR-Cas9 Knockout | Permanent deletion of BORIS gene. | Preclinical research tool. | Complete and permanent ablation. | Therapeutic safety and delivery. | Clonogenic assays show elimination of cancer stem cell population. |
| DNA Methyltransferase Inhibitors (DNMTi) | Indirect via demethylation of BORIS promoter? | Clinically approved (e.g., Azacitidine). | Repurposing opportunity. | Non-specific, genome-wide effects. | In some cell lines, DNMTi reduces BORIS expression by ~40%. |
| Immune Targeting | Cancer vaccines or CAR-T cells targeting BORIS peptides. | Early preclinical. | Harnesses immune system memory. | Potential on-target, off-tumor toxicity in testes. | BORIS peptide vaccine induced T-cell response, delayed tumor growth in mice. |
Experimental Protocol for Evaluating BORIS-Targeting siRNA In Vitro:
- Cell Seeding: Plate cancer cells (e.g., MCF-7 breast cancer line) in 96-well plates at 5,000 cells/well in antibiotic-free media 24 hours pre-transfection.
- Transfection Complex Preparation: Dilute 5 pmol of BORIS-specific siRNA (e.g., siBORIS pool) or scrambled control siRNA in 25 µL of Opti-MEM. In a separate tube, dilute 0.25 µL of lipofectamine RNAiMAX in 25 µL of Opti-MEM. Incubate both for 5 min at RT.
- Complex Formation: Combine the diluted siRNA and lipofectamine, mix gently, and incubate for 20 min at RT.
- Transfection: Add 50 µL of the complex to each well. Incubate cells for 72-96 hours.
- Efficacy Assessment:
- qRT-PCR: Extract RNA, synthesize cDNA, and run qPCR for BORIS mRNA (normalized to GAPDH).
- Western Blot: Lyse cells, run protein gels, probe with anti-BORIS and anti-β-Actin antibodies.
- Proliferation Assay: Add CCK-8 reagent at 72h, measure absorbance at 450nm after 2h incubation.
Diagram Title: BORIS Function and Therapeutic Targeting Strategies
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for BORIS/CTCFL Research
| Reagent | Function & Application | Example Product/Clone | Key Note |
|---|---|---|---|
| Anti-BORIS/CTCFL Antibody | Detection of BORIS protein in IHC, Western Blot, ChIP. | Mouse Monoclonal 6A8; Rabbit Polyclonal (Abcam ab187148). | Specificity is critical; confirm lack of cross-reactivity with CTCF. |
| BORIS siRNA/SgRNA Pool | Knockdown or knockout of BORIS expression for functional studies. | ON-TARGETplus Human CTCFL siRNA (Dharmacon); CRISPRa/i sgRNA. | Use validated pools and include multiple controls (scramble, CTCF-targeting). |
| Recombinant BORIS Protein | For EMSA (DNA-binding), in vitro assays, or antibody validation. | ActiveMotif or Abnova full-length or zinc-finger domain protein. | Ensure protein is purified from a eukaryotic system for proper folding. |
| CTCF/BORIS DNA Binding Site Probes | Electrophoretic Mobility Shift Assay (EMSA) to study binding competition. | Biotin-labeled oligonucleotides containing consensus CTCF/BORIS site. | Use mutant probes as negative controls for specificity. |
| BORIS Expression Plasmid | For overexpression studies to assess gain-of-function phenotypes. | pCMV6-BORIS-Myc-DDK (Origene). | Sequence verify due to repetitive GC-rich sequence. |
| DNA Methylation Analysis Kit | To assess epigenetic changes at target loci after BORIS modulation. | EZ DNA Methylation-Gold Kit (Zymo Research) or bisulfite sequencing. | Focus on known BORIS-regulated loci (e.g., MAGE-A1 promoter). |
Conclusion
The intricate duel between CTCF and BORIS for DNA binding sites represents a fundamental epigenetic switch governing genome architecture and transcriptional output. While CTCF maintains canonical insulation and stable loops, BORIS acts as a context-dependent subverter, particularly in malignancy, by evicting CTCF and reprogramming the epigenome. Methodological advances have refined our ability to map their distinct yet overlapping territories, though careful experimental design remains crucial to avoid misinterpretation. The validated model of their antagonism—where BORIS expression corrupts CTCF-mediated regulation—presents a compelling therapeutic axis. Future research must focus on developing small molecules or epigenetic editors to specifically inhibit oncogenic BORIS or restore protective CTCF binding, offering novel avenues for precision oncology based on 3D genome integrity.