CTCF and Enhancer-Promoter Insulation: Mechanisms, Methods, and Clinical Implications in Gene Regulation
This comprehensive review explores the pivotal role of CCCTC-binding factor (CTCF) in the insulation of enhancer-promoter interactions, a fundamental process governing precise gene expression.
CTCF and Enhancer-Promoter Insulation: Mechanisms, Methods, and Clinical Implications in Gene Regulation
Abstract
This comprehensive review explores the pivotal role of CCCTC-binding factor (CTCF) in the insulation of enhancer-promoter interactions, a fundamental process governing precise gene expression. We detail the structural and mechanistic foundations of CTCF-mediated loop formation and boundary establishment. We then examine current methodologies for mapping and perturbing CTCF sites, followed by troubleshooting common experimental challenges. Finally, we validate CTCF's function by comparing its insulation mechanisms to alternative proteins and analyzing disease-associated mutations. This article synthesizes knowledge for researchers and drug developers, highlighting how dysregulation of CTCF insulation contributes to disease and presents novel therapeutic opportunities.
CTCF 101: The Architectural Protein Shaping Genome Topology and Gene Expression
Within the three-dimensional nucleus, enhancer-promoter communication is a fundamental driver of precise spatiotemporal gene expression. Uncontrolled or ectopic interactions can lead to oncogenesis and developmental disorders. This whitepaper, framed within the broader thesis of CTCF's role in genomic architecture, details the mechanisms of enhancer-promoter communication, the critical necessity for its insulation, and the central function of CTCF/cohesin-mediated loop extrusion in establishing these boundaries. We provide current data, experimental protocols, and essential research tools for the study of chromatin insulation.
Core Concepts: Communication and Insulation
Enhancer-promoter communication involves physical proximity facilitated by chromatin looping, often directed by architectural proteins. Insulators are DNA sequences and associated protein complexes that block inappropriate enhancer-promoter interactions. The CCCTC-binding factor (CTCF), in conjunction with cohesin, is the principal architect of insulator function via the formation of topologically associating domains (TADs).
Quantitative Landscape of Insulator Elements
Table 1: Genomic Distribution and Characteristics of Human Insulator Elements (Based on Recent Studies)
| Feature | Quantitative Measure | Method of Determination | Functional Implication |
|---|---|---|---|
| CTCF Binding Sites | ~50,000 - 70,000 sites per diploid genome | ChIP-seq | Primary candidate insulator locations |
| Convergent CTCF Motif Orientation | >90% of TAD boundaries | CUT&RUN, Hi-C | Essential for loop extrusion stall and boundary formation |
| Boundary Strength (Average) | ~2-5 fold reduction in cross-TAD interactions | Micro-C/Hi-C | Quantitative insulation efficacy |
| Cohesin Occupancy at Boundaries | >80% co-localization with CTCF | ChIP-seq for RAD21/SMC1 | Indicates active loop extrusion complex |
| Disruption in Disease | >1,000 somatic mutations in cancer genomes clustered at boundary CTCF sites | WGS of tumor samples | Loss of insulation leads to oncogene activation |
CTCF/Cohesin Mechanism: The Loop Extrusion Model
The prevailing model posits that cohesin complexes extrude chromatin loops until encountering CTCF bound in a convergent orientation, thereby defining TAD boundaries and insulating enhancer-promoter pairs across boundaries.
Diagram 1: Loop Extrusion Insulation Model
Key Experimental Protocols
Protocol 1: Mapping 3D Chromatin Architecture with Hi-C
- Crosslinking: Use 2% formaldehyde on intact nuclei to fix chromatin interactions.
- Digestion: Use a restriction enzyme (e.g., DpnII, HindIII) to digest DNA.
- Proximity Ligation: Under dilute conditions, ligate crosslinked, digested ends to create chimeric junctions.
- Reverse Crosslinking & Purification: Isolate ligated DNA.
- Library Prep & Sequencing: Prepare sequencing library from purified DNA; use paired-end sequencing.
- Data Analysis: Process reads using pipelines (HiC-Pro, Juicer). Generate contact matrices and identify TADs (Arrowhead algorithm) and loops (HiCCUPS).
Protocol 2: Determining CTCF-Mediated Insulation via Degron Systems
- Cell Line Engineering: Create a parental cell line with an auxin-inducible degron (AID) tag on endogenous CTCF using CRISPR-Cas9.
- Control Treatment: Treat with DMSO vehicle.
- Rapid Depletion: Treat with auxin (IAA, 500 μM) for 4-6 hours to degrade CTCF.
- Parallel Assays: Harvest cells for:
- Hi-C: Assess global TAD boundary loss.
- RNA-seq: Identify differentially expressed genes.
- Capture Hi-C (cHi-C): For a specific locus, measure ectopic enhancer-promoter contacts.
- Validation: Use siRNA against cohesin subunit (e.g., RAD21) for comparative phenocopy.
Disrupted Insulation in Disease: An Oncogenic Example
Table 2: Consequences of Insulator Disruption at the MYC Locus in Colorectal Cancer
| Genomic Alteration | Insulation Effect | Quantitative Change in Interaction | Expression Outcome |
|---|---|---|---|
| Deletion of Boundary CTCF Site | Loss of TAD Boundary | ~8-fold increase in contacts between enhancer (upstream) and MYC promoter | MYC overexpression by ~4-fold |
| CTCF Site Somatic Mutation | Weakened CTCF Binding | ~3-fold increase in ectopic contacts | Moderate MYC activation |
| Epigenetic Silencing of Boundary | Loss of CTCF Occupancy | TAD fusion observed in Hi-C | Sustained oncogene dysregulation |
Diagram 2: Oncogene Activation via Insulator Loss
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Insulator Research
| Reagent/Category | Example Product/Assay | Primary Function in Research |
|---|---|---|
| CTCF Antibodies | Anti-CTCF (Cell Signaling, D31H2) | Chromatin immunoprecipitation (ChIP) for mapping binding sites. |
| Cohesin Component Antibodies | Anti-RAD21 (Abcam, EPR16779) | Co-IP or ChIP to assess cohesin complex localization. |
| Epigenetic Editing | dCas9-KRAB / dCas9-p300 | Targeted recruitment to insulator elements to disrupt or reinforce boundary function. |
| Live-Cell Imaging Probes | HaloTag-CTCF / SunTag-CTCF | Real-time visualization of CTCF dynamics upon transcriptional perturbation. |
| Degron Systems | Auxin-Inducible Degron (AID) Tag | Rapid, reversible degradation of CTCF or cohesin to study acute loss-of-function. |
| High-Resolution 3D Mapping | Micro-C Kit (Diagenode) | Nucleosome-resolution chromosome conformation capture. |
| Boundary Reporter Assays | STARR-Seq Enhancer Screens + Insulator Elements | High-throughput functional screening of candidate insulator sequences. |
| CRISPR gRNA Libraries | Custom-designed gRNAs targeting CTCF motifs | High-throughput screening for insulator function at scale. |
CCCTC-binding factor (CTCF) is an architecturally essential, ubiquitously expressed zinc finger protein in vertebrates. In the broader thesis of enhancer-promoter insulation research, CTCF is the principal mediator of this function. It establishes directional chromatin loops, primarily through cohesion recruitment, to generate topologically associating domains (TADs). These structures functionally insulate enhancers from inappropriate promoters, thereby ensuring precise spatiotemporal gene expression—a process critical for development, cellular identity, and disease prevention.
Structural Architecture and Functional Domains of CTCF
CTCF is a large, multi-domain protein (~82 kDa in humans) with a modular structure that dictates its diverse functions.
Table 1: Core Domains of Human CTCF (UniProt ID P49711)
| Domain / Region | Position (aa approx.) | Key Functional Role |
|---|---|---|
| N-terminal Domain (NTD) | 1-275 | Contains regions essential for transactivation and interaction with chromatin modifiers (e.g., CHD8, SIN3A). |
| Central 11x Zinc Finger Array (ZF) | 276-543 | The DNA recognition module. Eleven C2H2-type zinc fingers (ZF1-ZF11) bind a ~50-60 bp cognate sequence. Specific fingers mediate DNA base recognition. |
| Linker Regions | Between ZFs | Variable length and flexibility; critical for adapting to diverse DNA sequences. |
| C-terminal Domain (CTD) | 544-727 | Essential for insulation. Contains sub-regions for homodimerization and, critically, the interaction with cohesin complex subunits (RAD21, SA2). |
| Post-Translational Modifications (PTMs) | Throughout | Phosphorylation, PARylation, and SUMOylation sites regulate DNA binding, protein stability, and partner interactions. |
The Zinc Finger Array: Mechanism of DNA Sequence Recognition
The central 11 zinc fingers are not used equivalently; they form a contiguous binding interface that reads an extended DNA sequence. Recognition is degenerate and combinatorial, allowing CTCF to bind thousands of genomic variants of its core motif. Recent structural studies (Cryo-EM, X-ray) have elucidated the precise interactions.
Table 2: DNA Base Contact Specificity of CTCF Zinc Fingers (Consensus Model)
| Zinc Finger | Primary DNA Subsite Recognition (5'→3') | Key Role in Binding |
|---|---|---|
| ZF1, ZF2 | Variable, often weak/auxiliary | Can contribute to stability on certain motifs. |
| ZF3 | 5' flanking region | Important for anchoring and orientation. |
| ZF4 - ZF7 | Core consensus motif (e.g., CCGCGN) | High-affinity, sequence-specific recognition of the invariant core. |
| ZF8, ZF9 | 3' flanking region | Contributes to affinity and specificity. |
| ZF10, ZF11 | Essential for insulation | Recognize a specific sequence critical for directional insulation. Mutation here ablates insulator function without abolishing binding. |
Key Experimental Protocol: Electrophoretic Mobility Shift Assay (EMSA) for CTCF-DNA Binding
- Protein Purification: Express and purify recombinant full-length CTCF or its ZF domain from E. coli or insect cells.
- Probe Preparation: PCR amplify or anneal oligonucleotides containing the putative CTCF binding site (CBS). Label the 5' end with [γ-³²P]ATP using T4 Polynucleotide Kinase.
- Binding Reaction: Incubate purified CTCF (0-100 nM) with labeled probe (~0.1 nM) in binding buffer (10 mM HEPES pH 7.9, 50 mM KCl, 5 mM MgCl₂, 1 mM DTT, 10% glycerol, 0.1 mg/mL BSA, 0.1% NP-40) for 20-30 minutes at room temperature.
- Electrophoresis: Load reactions onto a pre-run 4-6% non-denaturing polyacrylamide gel in 0.5X TBE buffer at 4°C. Run at constant voltage (~150-200V) until adequate separation is achieved.
- Detection: Dry the gel and visualize shifted protein-DNA complexes using autoradiography or a phosphorimager. For supershift assays, include an anti-CTCF antibody prior to loading.
Diagram 1: CTCF Zinc Finger DNA Recognition Logic
From DNA Binding to Insulation: The Cohesin Collaboration
CTCF binding alone is not sufficient for insulation. The functional output—loop formation and insulation—requires collaborative interaction with the cohesin complex. The dominant model is the "cohesin ring extrusion" model, where CTCF acts as a boundary element.
Diagram 2: CTCF-Cohesin Mediated Loop Extrusion & Insulation
Key Experimental Protocol: Chromatin Conformation Capture (3C)
- Crosslinking: Treat cells with 2% formaldehyde for 10 min at room temperature to fix protein-DNA and protein-protein interactions.
- Lysis and Digestion: Lyse cells and digest chromatin with a frequent-cutter restriction enzyme (e.g., DpnII, HindIII) overnight.
- Proximity Ligation: Under dilute conditions, perform intramolecular ligation with T4 DNA Ligase to join crosslinked DNA fragments.
- Reversal of Crosslinks: Purify DNA and reverse crosslinks with Proteinase K at 65°C.
- Quantitative PCR: Design locus-specific primers around the CTCF site of interest ("viewpoint") and potential interacting regions ("targets"). Use qPCR with SYBR Green to quantify interaction frequency relative to control regions.
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Research Reagents for CTCF/Insulation Studies
| Reagent / Material | Function & Application |
|---|---|
| Anti-CTCF Antibodies (e.g., Millipore 07-729, Active Motif 61311) | For ChIP-seq, CUT&RUN, Western Blot, and immunofluorescence to map genomic binding or assess protein expression/localization. |
| Anti-Cohesin Antibodies (e.g., anti-RAD21, anti-SMC1) | To map cohesin occupancy (ChIP-seq) or validate its interaction with CTCF (Co-IP). |
| Recombinant CTCF Protein (full-length or ZF domain) | For in vitro binding assays (EMSA, SELEX, ITC) and structural studies. |
| dCas9-CTCF Fusion Systems | For targeted recruitment of CTCF to specific loci in vivo to test sufficiency for loop/insulation formation. |
| CTCF Motif Position Weight Matrices (PWMs) (from JASPAR, CIS-BP) | For bioinformatic prediction of CBS in genomic sequences. |
| Cohesin Inhibitors (e.g., TSA, Sororin inhibitors) | To acutely deplete or inhibit cohesin function and study the dynamic loss of TADs/loops. |
| Next-Generation Sequencing Kits (for ChIP-seq, Hi-C) | To generate genome-wide maps of CTCF binding (ChIP-seq) or 3D chromatin architecture (Hi-C, micro-C). |
| Cell Lines with Endogenous CTCF Tag (e.g., GFP-CTCF) | For live-cell imaging and purification of endogenous complexes under native conditions. |
| Mutant CTCF Constructs (e.g., ZF10/11 mutations, dimerization mutants) | To dissect the structural determinants of DNA binding vs. insulation function in rescue experiments. |
Within the framework of enhancer-promoter insulation research, the partnership between CCCTC-binding factor (CTCF) and the Cohesin complex is fundamental for establishing topologically associating domains (TADs) and specific chromatin loops. This guide details the molecular mechanics, experimental evidence, and quantitative data underpinning this collaboration, which is critical for precise gene regulation and a focal point for therapeutic intervention in diseases driven by chromatin architecture dysregulation.
CTCF is an 11-zinc finger protein that binds to specific, non-palindromic DNA sequences. A primary function, within the context of insulation research, is to block inappropriate enhancer-promoter communication. This insulating capability is not intrinsic to CTCF alone but is executed in concert with the Cohesin complex. The prevailing "loop extrusion" model posits that Cohesin is the motor that forms loops, while CTCF functions as a boundary element that halts extrusion at specific sites.
Molecular Mechanism of Collaboration
Key Players
- CTCF: The sequence-specific anchor. Its binding orientation and the motif's strength influence loop formation.
- Cohesin Complex: A ring-shaped ATPase complex (comprising SMC1, SMC3, RAD21, and STAG1/2) that extrudes chromatin.
- NIPBL-MAU2 (Loader): Facilitates the initial loading of Cohesin onto chromatin.
- WAPL (Unloader): Promotes the release of Cohesin from chromatin.
The Loop Extrusion Model
- Loading: The NIPBL-MAU2 complex loads Cohesin onto chromatin.
- Extrusion: Cohesin, utilizing ATP hydrolysis, progressively extrudes chromatin fiber, forming an expanding loop.
- Boundary Arrest: When the extruding Cohesin encounters a pair of CTCF molecules bound in a convergent orientation (head-to-head), extrusion is stalled.
- Stabilization: The stalled Cohesin stably maintains the loop. Antiparallel CTCF binding or sites in the same orientation do not efficiently arrest extrusion.
Diagram 1: The Loop Extrusion and Boundary Arrest Model
Quantitative Evidence and Data
Table 1: Key Quantitative Findings in CTCF/Cohesin Loop Formation
| Observation / Metric | Experimental Value / Finding | Implication |
|---|---|---|
| CTCF Motif Orientation Bias | >90% of loops anchor at convergent CTCF sites. | Convergent orientation is essential for loop boundary function. |
| Cohesin Depletion Effect | ~70-80% reduction in chromatin loop strength (by Hi-C). | Cohesin is the primary driver of loop formation. |
| CTCF Motif Strength Correlation | Stronger motif matches correlate with increased loop anchor insulation score (e.g., ~2-5 fold increase). | Binding affinity determines boundary efficiency. |
| Loop Size Distribution | Median loop size ~200kb, but ranges from 10kb to >1Mb. | Extrusion can traverse significant genomic distances. |
| Cohesin ChIA-PET Peak Overlap | ~85% of RAD21 peaks colocalize with CTCF ChIP-seq peaks. | Demonstrates intimate genomic co-occupancy. |
Core Experimental Protocols
Chromatin Conformation Capture (Hi-C)
Purpose: To genome-wide identify all chromatin interactions and define TADs/loops. Detailed Protocol:
- Crosslinking: Treat cells with 2% formaldehyde to fix protein-DNA and protein-protein interactions.
- Digestion: Lyse cells and digest chromatin with a restriction enzyme (e.g., HindIII or MboI).
- Marking Ends: Fill restriction fragment ends with biotinylated nucleotides.
- Ligation: Perform dilute, in-situ ligation to join crosslinked DNA fragments. Biotin marks ligation junctions.
- Reverse Crosslinking & Purification: Digest proteins, purify DNA, and shear it.
- Pull-down: Capture biotinylated ligation products using streptavidin beads.
- Library Prep & Sequencing: Prepare a sequencing library from captured DNA and perform paired-end sequencing.
- Analysis: Map reads, filter, and use tools (e.g., Juicer, HiC-Pro) to generate interaction matrices and call loops.
CTCF/Cohesin Depletion (CRISPRi or Auxin-Inducible Degron)
Purpose: To causally test the requirement of CTCF or Cohesin for specific loops. Detailed Protocol (Auxin-Inducible Degron - AID):
- Cell Line Engineering: Generate cell line expressing TIR1 E3 ligase and tag endogenous protein of interest (e.g., RAD21) with an AID tag.
- Degradation Induction: Treat cells with 500 µM auxin (IAA) for a time course (e.g., 0, 30, 60, 120 min).
- Validation: Confirm protein depletion via western blot.
- Downstream Assay: Perform Hi-C or 3C-qPCR on induced vs. uninduced cells to assess loop loss kinetics.
ChIP-seq for CTCF and Cohesin
Purpose: To map binding sites of CTCF and Cohesin subunits. Detailed Protocol:
- Crosslinking & Sonication: Fix cells with formaldehyde, lyse, and shear chromatin to ~200-500 bp fragments via sonication.
- Immunoprecipitation: Incubate chromatin with antibody against target (e.g., anti-CTCF, anti-RAD21) coupled to Protein A/G beads.
- Washing & Elution: Stringently wash beads, then elute bound chromatin.
- Reverse Crosslinking & Purification: Digest proteins and purify DNA.
- Library Prep & Sequencing: Prepare sequencing library and sequence.
Diagram 2: Hi-C Experimental Workflow Core Steps
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CTCF/Cohesin Loop Research
| Reagent / Tool | Function & Application |
|---|---|
| Anti-CTCF Antibody (ChIP-grade) | Immunoprecipitation of CTCF-bound DNA for ChIP-seq/CUT&RUN to define anchor sites. |
| Anti-RAD21/SMC1 Antibody | Immunoprecipitation of Cohesin-bound DNA to confirm co-occupancy with CTCF. |
| dCas9-KRAB/CRISPRi System | Epigenetic repression of specific CTCF binding sites to test their necessity for loop formation. |
| Auxin-Inducible Degron (AID) System | Rapid, conditional degradation of Cohesin subunits (e.g., RAD21-AID) to study immediate loop dissolution. |
| HindIII/MboI Restriction Enzymes | Primary enzymes for chromatin digestion in Hi-C protocols. |
| Biotin-14-dATP | Labeling of digested chromatin ends during Hi-C library preparation for selective pull-down. |
| CUT&RUN/CUT&Tag Kits | For low-input, high-resolution mapping of CTCF/Cohesin binding without crosslinking. |
| Hi-C Analysis Software (Juicer, fithic2) | Processing raw sequencing data, generating contact matrices, and calling significant loops. |
Within the broader thesis on CTCF's role in enhancer-promoter insulation, understanding its DNA binding motif is fundamental. CTCF (CCCTC-binding factor) is a master architectural protein that shapes the 3D genome by forming loop domains and insulating enhancers from inappropriate promoters. This function is critically dependent on its sequence-specific binding to thousands of genomic loci. This whitepaper provides a technical deconstruction of the CTCF motif, detailing its core consensus, orientation-dependent function, quantitative measures of binding strength, and experimental methodologies for its study.
The Core CTCF Motif: Sequence Specificity
The canonical CTCF binding motif is an ~15-20 bp sequence with a high degree of conservation. Recent genome-wide analyses (ChIP-seq, SELEX) have refined the consensus.
Table 1: Core CTCF Motif Consensus and Key Positions
| Position (from 5') | Consensus Nucleotide | Information Content (Bits) | Functional Role |
|---|---|---|---|
| 1-4 | CCGC | High (≥2.0) | Critical for initial docking |
| 5-8 | Variable | Low (≤0.5) | Spacer region; some flexibility |
| 9-12 | GNGG | High (≥1.8) | Central core recognition |
| 13-15 | CAC | Moderate (≥1.2) | Stabilizing contacts |
| 16-20 | Variable / TGG | Low to Moderate | Contributes to binding affinity variance |
The motif is non-palindromic and thus possesses a defined orientation, which is crucial for its function in directing asymmetric loop extrusion by cohesin.
Motif Orientation and Genomic Function
The orientation of the CTCF motif is a primary determinant of chromatin loop boundaries. Convergently oriented motifs (forward→←reverse) are the strongest drivers of loop formation and enhancer insulation.
Table 2: Impact of CTCF Motif Orientation on Genomic Architecture
| Orientation Pairing | Frequency at Loop Anchors | Relative Loop Strength | Predicted Insulation Effect |
|---|---|---|---|
| Convergent (→ ←) | ~70-80% | High | Strong |
| Forward (→ →) | ~10-15% | Low | Weak/None |
| Reverse (← ←) | ~10-15% | Low | Weak/None |
| Divergent (← →) | Rare | Very Low | Minimal |
Quantifying Motif Strength
Motif "strength" is a composite measure of binding affinity and in vivo occupancy, predicted by sequence deviation from consensus and contextual genomic features.
Table 3: Metrics for CTCF Motif Strength Prediction
| Metric | Description | Typical Range/Value | Correlation with ChIP-seq Signal (R) |
|---|---|---|---|
| Position Weight Matrix (PWM) Score | Sum of log-odds scores for each base position versus background model. | 0 (poor) to 20+ (exact consensus) | 0.5 - 0.7 |
| Motif Conservation (PhyloP) | Evolutionary conservation score across species. | -20 (unconserved) to +10 (high) | 0.4 - 0.6 |
| CpG Methylation Status | Methylation at motif CpGs (often within motif) disrupts binding. | 0 (unmethylated) to 1 (fully methylated) | Strong negative correlation |
| Chromatin Accessibility (ATAC-seq) | Open chromatin signal at motif locus. | 0 (closed) to 10+ (highly open) | 0.6 - 0.8 |
Experimental Protocols for CTCF Motif Analysis
Protocol: Determining CTCF Binding Specificity (SELEX-seq)
Objective: Identify high-affinity DNA sequences bound by CTCF. Reagents: Purified recombinant CTCF zinc finger domain, double-stranded random oligonucleotide library (N15-20), Ni-NTA magnetic beads (if tagged), sequencing adapters. Procedure:
- Incubation: Mix CTCF protein with random oligonucleotide library in binding buffer (20 mM HEPES pH 7.9, 100 mM KCl, 1 mM DTT, 0.1% NP-40, 10% glycerol) for 30 mins on ice.
- Pull-down: Add beads to capture protein-DNA complexes. Wash 5x with binding buffer + 0.5 M KCl to remove low-affinity binders.
- Elution: Elute bound DNA with 2% SDS at 65°C.
- Amplification: PCR amplify eluted DNA.
- Iteration: Repeat steps 1-4 for 4-8 rounds of selection.
- Sequencing & Analysis: High-throughput sequence final pool. Use MEME or HOMER for de novo motif discovery.
Protocol: Validating Motif Orientation Function (CRISPR Inversion)
Objective: Test the functional consequence of motif orientation on insulation. Reagents: sgRNAs designed to flank motif, Cas9 protein, donor template containing inverted motif, H3K27ac antibodies (for enhancer mark), RNA FISH probes for target gene. Procedure:
- Design: Design two sgRNAs ~200-500bp apart flanking the endogenous CTCF motif.
- Template: Synthesize a single-stranded DNA donor template containing the inverted CTCF motif sequence with ~60bp homology arms.
- Transfection: Co-transfect sgRNAs, Cas9, and donor template into relevant cell line.
- Screening: Isolate clones and genotype by PCR and Sanger sequencing to confirm inversion.
- Phenotyping:
- 3C/qPCR: Measure changes in chromatin looping at the edited locus.
- ChIP-qPCR: Assess loss of CTCF occupancy.
- H3K27ac ChIP: Measure encroachment of enhancer mark into insulated domain.
- RNA FISH: Quantify mis-expression of the previously insulated gene.
Visualizing CTCF Motif Function in Insulation
Diagram Title: CTCF Motif Orientation Directs Loop Extrusion and Insulation
The Scientist's Toolkit: Key Research Reagents
Table 4: Essential Reagents for CTCF Motif and Insulation Research
| Reagent / Material | Function | Example Supplier / Catalog |
|---|---|---|
| Recombinant Human CTCF Protein (Full-length or ZF Domain) | In vitro binding assays (EMSA, SELEX), structural studies. | Active Motif, Abcam |
| Anti-CTCF Antibody (ChIP-seq Grade) | Chromatin immunoprecipitation to map genomic occupancy. | Cell Signaling Tech., Millipore |
| dCas9-KRAB / dCas9-CTCF Fusion Constructs | Epigenetic perturbation: KRAB for targeted repression, CTCF for targeted recruitment to test sufficiency. | Addgene |
| CTCF Motif Reporter Plasmid Library | High-throughput measurement of binding affinity for motif variants. | Custom synthesis |
| Biotinylated Oligonucleotides (Wild-type & Mutant Motif) | EMSA and pull-down competition assays to measure binding specificity and affinity. | IDT, Sigma |
| 4C-seq or Hi-C Kit | Genome-wide and locus-specific analysis of chromatin architecture and loops. | Arima Genomics, NuGEN |
| Methyltransferase (e.g., M.SssI) / Demethylating Agents (e.g., 5-aza-dC) In vitro methylation of motifs or cellular treatment to study DNA methylation impact on CTCF binding. | NEB, Sigma | |
| Cell Line with Endogenous Tagged CTCF (e.g., CTCF-AID) | Rapid, specific degradation of CTCF to study acute loss-of-function effects on insulation. | Generated via CRISPR |
The CTCF motif is a sophisticated molecular code governing genome topology. Its precise sequence, inherent orientation, and quantitative strength directly determine the efficiency of cohesin loop extrusion and the establishment of insulating boundaries. Decoding this motif—through integrated computational, biochemical, and genetic engineering approaches—is essential for advancing the thesis that targeted disruption or reinforcement of CTCF-mediated insulation represents a novel therapeutic axis in diseases of gene misregulation, including cancer and developmental disorders.
This whitepaper examines the dual mechanisms of CCCTC-binding factor (CTCF) function, framing them within the broader thesis of its paramount role in enhancer-promoter insulation. CTCF is a master architect of 3D genome organization, primarily known for its canonical role in establishing chromatin loops and topologically associating domain (TAD) boundaries via sequence-specific DNA binding to its consensus motif, thereby insulating enhancers from inappropriate promoters. However, emerging evidence underscores non-canonical pathways—including sequence-independent binding, RNA-mediated recruitment, and post-translational modification-driven functions—that expand and modulate its insulating capability. Disentangling these mechanisms is critical for researchers and drug development professionals aiming to decipher gene regulation in development and disease, and for designing therapeutic strategies that target chromatin topology.
Canonical CTCF Binding and Function
The canonical mechanism is defined by CTCF binding to a well-conserved, ~50-60 bp motif through its 11 zinc finger (ZF) domain. This binding is cooperative with cohesin and is essential for loop extrusion and boundary formation.
Key Quantitative Data: Canonical Binding
| Parameter | Typical Value / Observation | Experimental Method |
|---|---|---|
| Consensus Motif Length | ~50-60 bp | SELEX, ChIP-seq |
| Primary ZFs for DNA Contact | ZF3-7 (core motif) | Crystallography, EMSA mutants |
| Binding Sites per Human Genome | ~50,000 - 100,000 | ChIP-seq peak calling |
| Co-binding with Cohesin (Rad21) | ~70-80% of sites | ChIP-seq co-localization |
| Boundary Strength Correlation (CTCF vs. TAD) | R ≈ 0.7-0.9 | Hi-C data correlation analysis |
| Motif Methylation (CpG) Effect | >90% reduction in binding | ChIP-qPCR with methylated oligos |
Detailed Experimental Protocol: ChIP-seq for Canonical CTCF Binding
- Crosslinking: Treat ~10 million cells with 1% formaldehyde for 10 minutes at room temperature. Quench with 125 mM glycine.
- Cell Lysis & Sonication: Lyse cells in SDS lysis buffer. Sonicate chromatin to an average fragment size of 200-500 bp. Confirm fragment size by agarose gel electrophoresis.
- Immunoprecipitation: Incubate chromatin with 2-5 µg of validated anti-CTCF antibody (e.g., Millipore 07-729) overnight at 4°C. Use Protein A/G magnetic beads for capture.
- Washing & Elution: Wash beads sequentially with Low Salt, High Salt, LiCl, and TE buffers. Elute complexes with freshly prepared elution buffer (1% SDS, 0.1M NaHCO3).
- Reverse Crosslinks & Purification: Incubate eluates with 200 mM NaCl at 65°C overnight. Treat with RNase A and Proteinase K. Purify DNA using silica membrane columns.
- Library Prep & Sequencing: Prepare sequencing library using standard kits (e.g., NEBNext Ultra II). Sequence on an Illumina platform to a depth of ~20-40 million reads.
- Data Analysis: Align reads to reference genome (e.g., hg38). Call peaks using MACS2. Motif analysis can be performed with HOMER or MEME.
Non-Canonical CTCF Binding and Function
Non-canonical mechanisms bypass the strict requirement for the consensus motif, enabling CTCF to localize to alternative genomic locations and engage in distinct functional interactions.
Key Quantitative Data: Non-Canonical Pathways
| Parameter | Observation in Non-Canonical Context | Experimental Method |
|---|---|---|
| RNA-Mediated Recruitment | ||
| Fraction of CTCF Bound to RNA (iCLIP) | ~20-30% of cellular CTCF | iCLIP-seq, RIP-seq |
| Jpx RNA-CTCF Interaction Kd | Reported ~100-200 nM | EMSA / RNA Pull-down |
| Protein Partner-Mediated Tethering | ||
| CTCF-YB1 Co-localization Sites | Thousands of motif-weak sites | Co-immunoprecipitation, CUT&Tag |
| Modification-Driven Binding | ||
| Poly(ADP-ribosyl)ation (PAR) at Damage Sites | Rapid, transient recruitment (<5 min) | Live-cell imaging, PAR-ChIP |
| Sequence-Independent (Low-Affinity) Sites | ||
| Occupancy at Low-Complexity DNA | Weaker signal, more cell-type specific | Sensitive ChIP-exo/ChIP-nexus |
Detailed Experimental Protocol: RNA Immunoprecipitation (RIP) for CTCF-RNA Interaction
- Cell Lysis: Lyse cells in polysome lysis buffer (e.g., containing 0.5% NP-40, RNase inhibitors) to preserve RNA-protein complexes.
- Clarification: Centrifuge lysate at 12,000g for 10 min at 4°C. Pre-clear supernatant with protein A/G beads for 30 min.
- Immunoprecipitation: Incubate supernatant with anti-CTCF antibody or species-matched IgG control for 2 hours at 4°C. Add pre-washed magnetic beads and incubate for another hour.
- Stringent Washes: Wash beads 5-6 times with high-salt wash buffer (e.g., containing 500 mM NaCl and 0.1% SDS) to reduce non-specific RNA binding.
- RNA Elution & Isolation: Resuspend beads in Proteinase K buffer and digest at 55°C for 30 min. Extract RNA using acid phenol:chloroform and precipitate with ethanol.
- Analysis: Analyze RNA by qRT-PCR for specific transcripts (e.g., Jpx, Ctcf) or prepare libraries for high-throughput sequencing (RIP-seq).
Visualizing CTCF Mechanisms
Diagram Title: CTCF Canonical and Non-Canonical Functional Pathways
Diagram Title: Experimental Workflow for CTCF ChIP-seq
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Supplier Examples | Function & Application |
|---|---|---|
| Anti-CTCF Antibody (ChIP-grade) | Millipore (07-729), Active Motif (61311), Cell Signaling (3418S) | Specific immunoprecipitation of CTCF-DNA/RNA complexes for ChIP, RIP, CUT&Tag. |
| Cohesin Subunit (Rad21/SMC1) Antibody | Abcam, Bethyl Laboratories | Co-IP or co-localization studies to investigate canonical loop extrusion complexes. |
| Recombinant CTCF Protein (ZF domain) | Active Motif, Abnova | For in vitro binding assays (EMSA, SELEX) to study motif specificity and mutations. |
| Methylated & Unmethylated Motif Oligos | Integrated DNA Technologies (IDT) | Probes to quantitatively assess the impact of CpG methylation on CTCF binding affinity. |
| Jpx / CTCF-targeting siRNAs or ASOs | Dharmacon, Ionis Pharmaceuticals | Functional knockdown of non-coding RNA or CTCF itself to study loss-of-function effects on insulation. |
| PARP Inhibitor (e.g., Olaparib) | Selleckchem, Tocris | To probe the role of PARylation in non-canonical, damage-induced CTCF recruitment. |
| CUT&Tag Assay Kit (for Low-Abundance Targets) | EpiCypher, Cell Signaling (CellCUT&Tag) | Sensitive mapping of CTCF at low-affinity or non-canonical sites with low background. |
| Proximity Ligation Assay (PLA) Kit | Sigma-Aldrich (Duolink) | Visualize in situ protein-protein interactions (e.g., CTCF-YB1) at single-molecule resolution. |
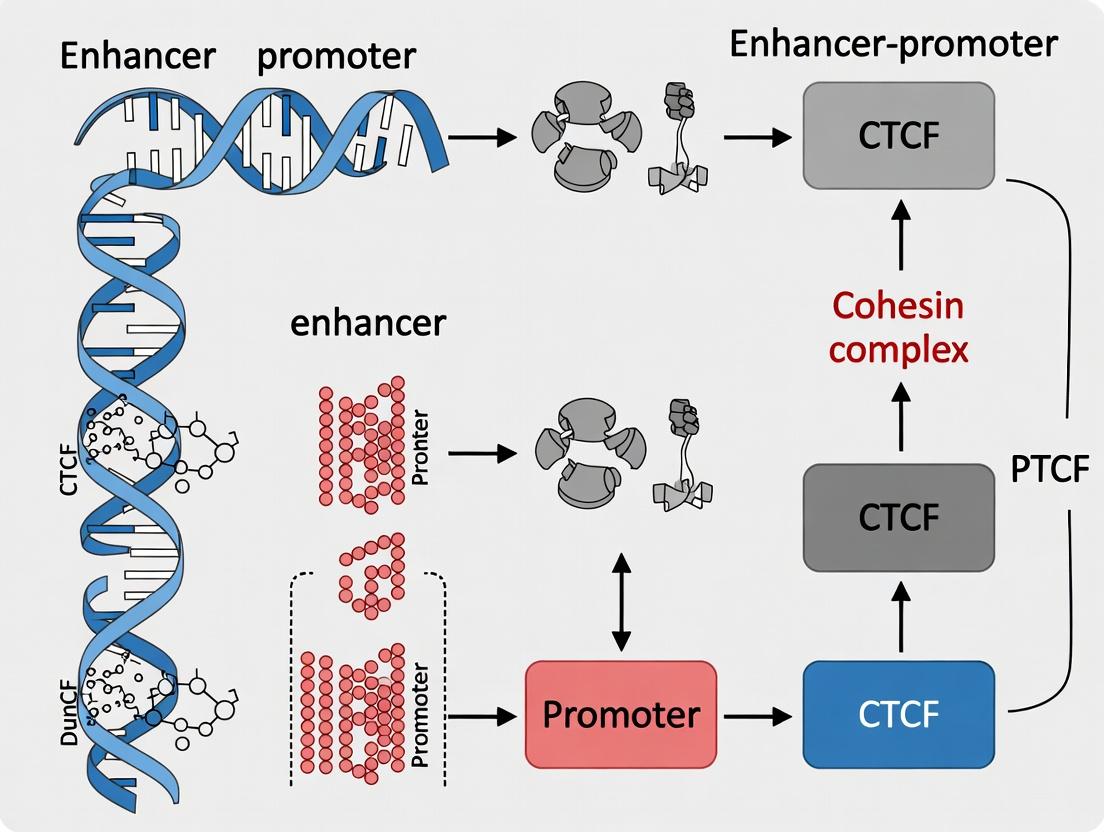
Within the context of enhancer-promoter insulation research, the protein CCCTC-binding factor (CTCF) is established as a central architectural component of the genome. Its primary function, in conjunction with the cohesin complex, is to organize chromatin into discrete three-dimensional structures known as Topologically Associating Domains (TADs). TADs are fundamental units of chromosome folding, characterized by high internal contact frequency and insulation from neighboring regions. This guide elucidates the mechanistic basis of CTCF-mediated loop extrusion and insulation, detailing the experimental paradigms that visualize these processes and their quantitative outcomes.
Core Mechanism: Cohesin Extrusion and CTCF Boundary Definition
The prevailing model for TAD formation is the loop extrusion model. In this model, a cohesin complex is loaded onto chromatin and begins to extrude a growing DNA loop. This process continues unimpeded until the cohesin ring encounters a pair of convergently oriented CTCF binding sites. CTCF binding, especially when bound by its cofactor cohesin, acts as a directional barrier, halting further extrusion. The stabilized loop forms the basis of a TAD boundary, insulating regulatory elements within the loop from those outside.
Diagram: CTCF-Cohesin Loop Extrusion Model
Quantitative Data on CTCF/Cohesin and TAD Properties
Table 1: Key Quantitative Features of TADs and CTCF Binding in Mammalian Genomes
| Feature | Typical Range/Value (Human/Mouse) | Measurement Method | Functional Implication |
|---|---|---|---|
| TAD Size | ~200 kb to 1 Mb | Hi-C | Defines scale of insulated neighborhood. |
| CTCF Motifs per Genome | ~50,000 - 100,000 | ChIP-seq, Motif Search | Potential loop anchor sites. |
| Fraction of CTCF Sites at TAD Boundaries | ~30-40% | Hi-C + CTCF ChIP-seq | Highlights specificity of boundary function. |
| Convergent Orientation Prevalence at Boundaries | >90% | Hi-C + Motif Analysis | Critical for directional insulation. |
| Loop Strength (Contact Frequency) | Varies by locus; can be >10-fold over background | Hi-C (observed/expected) | Correlates with insulation score. |
| Insulation Score Delta at Boundary | Significant dip (negative peak) | Insulation Score Analysis | Quantitative measure of boundary strength. |
Experimental Protocols for Visualization and Validation
Hi-C to Map Chromatin Architecture
Purpose: To genome-wide capture chromatin interaction frequencies and identify TADs. Detailed Protocol:
- Crosslinking: Treat cells with 1-3% formaldehyde to fix protein-DNA and protein-protein interactions.
- Digestion: Lyse cells and digest chromatin with a restriction enzyme (e.g., HindIII, DpnII, MboI).
- End Repair and Biotinylation: Fill in restriction fragment ends and mark with biotin-14-dATP.
- Ligation: Perform proximity ligation under dilute conditions to favor intra-molecular ligation of crosslinked fragments.
- Reverse Crosslinking & Purification: Purify ligated DNA and shear it to ~300-500 bp.
- Pull-down: Capture biotinylated ligation junctions with streptavidin beads.
- Library Prep & Sequencing: Prepare sequencing library from pulled-down DNA and perform paired-end sequencing.
- Data Analysis: Map reads to reference genome, filter for valid interaction pairs, and generate contact matrices. Use algorithms (e.g., Knight-Ruiz normalization, InsulationScore, DirectionalityIndex) to call TAD boundaries.
CTCF/Cohesin Chromatin Immunoprecipitation Sequencing (ChIP-seq)
Purpose: To map the genomic binding sites of CTCF and cohesin subunits (e.g., RAD21, SMC3). Detailed Protocol:
- Crosslinking & Sonication: Fix cells with formaldehyde, lyse, and shear chromatin to 200-500 bp fragments via sonication.
- Immunoprecipitation: Incubate chromatin with validated antibody against target protein (e.g., anti-CTCF, anti-RAD21) and Protein A/G beads. Use species-matched IgG as control.
- Washing & Elution: Wash beads stringently and elute bound chromatin.
- Reverse Crosslinking & DNA Cleanup: Treat eluate with proteinase K and heat to reverse crosslinks, then purify DNA.
- Library Prep & Sequencing: Prepare sequencing library and perform high-throughput sequencing.
- Data Analysis: Map reads, call peaks (using tools like MACS2), and analyze overlap with TAD boundaries and motif orientation.
Acute Depletion/Inversion Experiments (e.g., auxin-inducible degron, CRISPR inversion)
Purpose: To test the causal role and directionality requirement of CTCF sites. Detailed Protocol for CRISPR-mediated CTCF Site Inversion:
- Design: Design two sgRNAs flanking the endogenous CTCF motif. Design a donor template containing the inverted motif sequence.
- Transfection: Co-transfect cells with Cas9 expression vector, sgRNA expression plasmids, and ssODN/donor template.
- Screening: Isolate clones and genotype by PCR and Sanger sequencing to identify homozygous inversions.
- Phenotyping: Perform Hi-C and gene expression analysis (e.g., RNA-seq, RT-qPCR of putative target genes) on validated clones versus wild-type controls.
Diagram: Key Experimental Workflow for TAD Analysis
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents and Tools for CTCF/TAD Research
| Reagent/Tool | Function | Example/Provider |
|---|---|---|
| Anti-CTCF Antibody | Immunoprecipitation for ChIP-seq; validation by WB/IF. | MilliporeSigma (07-729), Active Motif (61311). |
| Anti-RAD21/SMC1 Antibody | Immunoprecipitation of cohesin complex for ChIP-seq. | Abcam (ab992), Bethyl Laboratories. |
| Hi-C Kit | Streamlined, optimized reagents for Hi-C library prep. | Arima-HiC Kit, Dovetail Omni-C Kit. |
| Validated sgRNAs & Donor Templates | For CRISPR-mediated editing of CTCF sites. | Designed via CRISPR design tools, synthesized as ssODNs. |
| Auxin-Inducible Degron (AID) System | For rapid, acute depletion of CTCF or cohesin subunits. | Cell lines expressing osTIR1 and target protein fused to AID tag. |
| 4C-seq Primers & Probes | For targeted investigation of specific locus chromatin interactions. | Custom-designed viewpoint-specific primers. |
| Motif Analysis Software | To identify and determine orientation of CTCF binding motifs. | HOMER, FIMO (MEME Suite), CTCFBSDB. |
| Hi-C Analysis Pipeline | For processing raw sequencing data into normalized contact maps. | HiC-Pro, Juicer, Cooler. |
| TAD Calling Algorithm | To identify TAD boundaries from Hi-C data. | Insulation Score (Crane et al.), Directionality Index (Dixon et al.), Arrowhead (Juicebox). |
Signaling/Regulatory Pathway of CTCF-Mediated Insulation
Diagram: Molecular Pathway of Loop Extrusion and Insulation
Mapping and Manipulating Boundaries: Techniques to Study CTCF Insulation in Research and Therapy
This guide details the principal genome-wide mapping technologies used to investigate the role of CTCF and cohesin in enhancer-promoter insulation and 3D genome architecture. The thesis posits that CTCF-mediated loops, facilitated by cohesin, are fundamental to insulating enhancers from inappropriate promoters, thereby ensuring precise gene regulation. Dysregulation of this architecture is implicated in disease, offering novel targets for therapeutic intervention.
Core Technologies: Principles and Applications
Chromatin Immunoprecipitation Sequencing (ChIP-seq)
Purpose: Maps protein-DNA interactions genome-wide, identifying binding sites for CTCF and cohesin subunits (e.g., SMC1A, RAD21). Principle: Cross-linked chromatin is immunoprecipitated with an antibody against the target protein. The co-precipitated DNA is then sequenced and aligned to the reference genome to identify enriched regions (peaks).
Hi-C
Purpose: Captures genome-wide chromatin interactions, identifying topologically associating domains (TADs) and chromatin loops, many anchored by convergent CTCF motifs. Principle: Chromatin is cross-linked, digested, and ligated under conditions that favor joining of spatially proximal DNA fragments. The resulting chimeric fragments are sequenced to reveal contact frequencies.
Micro-C
Purpose: An enhanced version of Hi-C using micrococcal nuclease (MNase) for digestion, providing higher-resolution maps of chromatin contacts, including those within nucleosome-depleted regions. Principle: Similar to Hi-C but utilizes MNase to cleave linker DNA between nucleosomes, generating a more uniform fragmentation and enabling nucleosome-resolution contact maps.
Quantitative Comparison of Technologies
Table 1: Comparative Analysis of Genome-Wide Mapping Technologies
| Feature | ChIP-seq | Hi-C | Micro-C |
|---|---|---|---|
| Primary Output | Protein binding sites (peaks) | Genome-wide contact matrix | High-resolution contact matrix |
| Typical Resolution | 100-500 bp | 1 kb - 100 kb | < 1 kb (nucleosome-scale) |
| Key Insight for Thesis | Identifies CTCF/cohesin occupancy | Identifies TADs/loop structures anchored by CTCF | Reveals fine-scale loop extrusion and nucleosome positioning |
| Cross-linking Agent | Formaldehyde | Formaldehyde | Formaldehyde + DSG (optional) |
| Digestion Enzyme | Sonication (usually) | Restriction enzyme (e.g., DpnII, HindIII) | Micrococcal Nuclease (MNase) |
| Ligation | No | Proximity ligation | Proximity ligation |
| Primary Application | Candidate cis-regulatory element identification | Macro/meso-scale 3D architecture | Fine-scale 3D architecture and extruder dynamics |
| Cost (Relative) | Low | High | Very High |
Detailed Experimental Protocols
ChIP-seq for CTCF
- Cross-linking: Treat cells with 1% formaldehyde for 10 min at room temperature. Quench with 125 mM glycine.
- Cell Lysis & Sonication: Lyse cells and sonicate chromatin to shear DNA to an average size of 200-500 bp.
- Immunoprecipitation: Incubate lysate with anti-CTCF antibody (e.g., Millipore 07-729) overnight at 4°C. Capture antibody complexes with Protein A/G magnetic beads.
- Washing & Elution: Wash beads stringently. Elute chromatin with elution buffer (1% SDS, 0.1M NaHCO3). Reverse cross-links at 65°C overnight.
- Library Prep & Sequencing: Purify DNA. Prepare sequencing library using standard kits (e.g., Illumina). Sequence on an Illumina platform (≥20 million reads for human).
In-situ Hi-C
- Cross-linking & Digestion: Cross-link cells with 3% formaldehyde. Lyse nuclei. Digest chromatin with a 4-cutter restriction enzyme (e.g., DpnII or MboI).
- Fill-in & Marking: Fill 5´ overhangs with biotinylated nucleotides.
- Proximity Ligation: Dilute and ligate under conditions favoring intramolecular ligation of cross-linked fragments.
- Reverse Cross-linking & Purification: Purify DNA and shear to 300-500 bp. Pull down biotinylated ligation products with streptavidin beads.
- Library Prep & Sequencing: Prepare a standard Illumina sequencing library from the bead-bound DNA. Perform paired-end sequencing.
Micro-C
- Cross-linking (Optional): Cross-link with disuccinimidyl glutarate (DSG) followed by formaldehyde to stabilize protein-protein interactions.
- MNase Digestion: Isolate nuclei. Digest chromatin with MNase to mononucleosome resolution.
- End Repair & Ligation: Repair DNA ends. Perform proximity ligation as in Hi-C.
- Reverse Cross-linking & Library Prep: Reverse cross-links, purify DNA, and prepare a sequencing library. Size-select for ligated di-nucleosomal fragments (~250-350 bp).
- Sequencing: Perform deep paired-end sequencing (≥ 200 million read pairs for human at high resolution).
Visualizations
Title: ChIP-seq Workflow for CTCF
Title: CTCF/Cohesin Mediate Insulating Loop Formation
Title: Technology Evolution Informs Thesis
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents for CTCF/3D Genome Studies
| Reagent/Material | Supplier Examples | Function in Experiment |
|---|---|---|
| Anti-CTCF Antibody | Millipore (07-729), Active Motif (61311) | Immunoprecipitation of CTCF-bound chromatin fragments for ChIP-seq. |
| Anti-SMC1 Antibody | Abcam (ab9262), Bethyl Laboratories | IP of cohesin complex components to map cohesin occupancy. |
| Formaldehyde (37%) | Sigma-Aldrich, Thermo Fisher | Cross-links proteins to DNA and proteins to proteins, stabilizing in vivo interactions. |
| DpnII Restriction Enzyme | NEB | High-fidelity restriction enzyme for in-situ Hi-C protocol to digest chromatin. |
| Micrococcal Nuclease (MNase) | NEB, Worthington | Digests linker DNA for nucleosome-resolution mapping in Micro-C. |
| Biotin-14-dATP | Thermo Fisher (19524016) | Labels digested DNA ends during Hi-C/Micro-C library prep for selective pull-down of ligated junctions. |
| Streptavidin Magnetic Beads | Thermo Fisher (65601), NEB | Isolates biotinylated ligation products to enrich for valid proximity ligation events. |
| Protein A/G Magnetic Beads | Thermo Fisher, Millipore | Captures antibody-protein complexes during chromatin immunoprecipitation. |
| PCR-Free Library Prep Kit | Illumina | Prepares sequencing libraries with minimal amplification bias, critical for Hi-C/Micro-C. |
| High-Fidelity DNA Polymerase | NEB (Q5), KAPA Biosystems | Amplifies low-input ChIP DNA or library fragments with high accuracy. |
| Cell Permeant Cross-linker (DSG) | Thermo Fisher (20593) | Stabilizes protein-protein interactions prior to formaldehyde fixation, improving Micro-C signal for cohesin. |
1. Introduction within Thesis Context
This whitepaper details methodologies for the acute functional disruption of CTCF, a critical architectural protein for 3D genome organization. Within the broader thesis of enhancer-promoter insulation research, precise manipulation of CTCF binding sites (CBS) and protein levels is essential to dissect causality in chromatin looping, insulation, and gene regulation. While population-level CRISPR edits reveal long-term consequences, acute degradation bridges the gap to observe direct, immediate effects, separating primary from secondary adaptations.
2. Core Methodologies
2.1. CRISPR-Cas9 Mediated CBS Deletion/Inversion
This approach permanently alters genomic architecture to test the necessity of specific CBS for insulation.
- Experimental Protocol:
- Target Identification: Using ChIP-seq data, identify CBS at boundaries of topologically associating domains (TADs) or putative insulator elements between an enhancer and promoter of interest.
- gRNA Design: Design two single-guide RNAs (sgRNAs) flanking the ~20-50 bp core consensus sequence of the CBS. Target sequences should have high on-target and low off-target scores (e.g., using CRISPOR, ChopChop).
- Construct Delivery: Clone sgRNA sequences into a plasmid vector (e.g., pX459) expressing SpCas9 and the sgRNA. Transfect target cells (e.g., HCT-116, mESCs) via nucleofection or lipofection.
- Clonal Isolation: 48-72 hours post-transfection, begin selection with puromycin (1-2 µg/mL, 48h). Subsequently, single cells are sorted or diluted into 96-well plates to derive clonal populations.
- Genotyping: Screen clones by PCR across the target locus. Amplicon size changes indicate deletion. Sanger sequencing of PCR products confirms inversion or precise deletion. Validate loss of CTCF binding via ChIP-qPCR.
2.2. Degron Systems for Acute CTCF Depletion
This system enables rapid, reversible protein depletion to study the immediate consequences of CTCF loss.
- Experimental Protocol (AIDv2 System):
- Knock-in Cell Line Generation: Using CRISPR-Cas9 homology-directed repair (HDR), fuse an auxin-inducible degron (AIDv2) and a selectable marker (e.g., FLAG, mClover3) to the N- or C-terminus of the endogenous CTCF gene. AIDv2 is a modified, minimal tag for efficient degradation.
- Stable Expression of OsTIR1: Generate a cell line stably expressing the plant F-box protein OsTIR1 (mutant F74G is commonly used for human cells) under a constitutive or inducible promoter. Alternatively, introduce OsTIR1 via lentiviral transduction.
- Acute Depletion: To degrade CTCF-AID, add 500 µM auxin (Indole-3-acetic acid, IAA) or 500 nM auxin analog 5-Ph-IAA to the culture medium. Depletion typically occurs within 30-60 minutes.
- Validation: Monitor depletion kinetics via western blot (anti-CTCF, anti-FLAG) and immunofluorescence. Reversibility is tested by washing out auxin and replacing with fresh medium.
3. Quantitative Data Summary
Table 1: Comparison of CTCF Disruption Methods
| Parameter | CRISPR Deletion/Inversion | Acute Degron (AID) |
|---|---|---|
| Temporal Resolution | Permanent, static change | Acute (minutes to hours), reversible |
| Effect on CTCF | Eliminates specific binding site(s) | Depletes total cellular protein |
| Primary Readouts | Altered gene expression (RNA-seq), TAD boundary erosion (Hi-C), loss of insulation (STARR-seq) | Rapid transcription changes (PRO-seq, scRNA-seq), cohesin redistribution (ChIP-seq), loop dissolution (acute Hi-C) |
| Time to Effect Analysis | Weeks (clonal expansion required) | Minutes to hours post-auxin addition |
| Key Advantage | Studies locus-specific necessity | Studies acute, global necessity; separates primary/secondary effects |
| Main Limitation | Potential for compensatory genomic adaptations; clonal variability | Requires genomic tagging; basal degradation without auxin possible. |
Table 2: Typical Degradation Kinetics for CTCF-AID Systems
| Cell Line | CTCF Tag | OsTIR1 Expression | Degron Ligand | Time to >90% Depletion | Recovery Time (Washout) | Source |
|---|---|---|---|---|---|---|
| HCT-116 | CTCF-miniAID* | Constitutive (CMV) | 500 µM IAA | 30 min | ~6-8 hours | (Natsume et al., 2016) |
| mESC | CTCF-AIDv2-FLAG | Doxycycline-inducible | 500 nM 5-Ph-IAA | 60 min | ~12 hours | (Wutz et al., 2020) |
| RPE1 | CTCF-mAID-mClover3 | Constitutive (EF1α) | 500 µM IAA | 45 min | N/A | (Gassler et al., 2022) |
4. The Scientist's Toolkit: Essential Reagents
Table 3: Key Research Reagent Solutions
| Reagent / Material | Function / Purpose | Example Catalog # |
|---|---|---|
| SpCas9-sgRNA Vector | Expresses Cas9 nuclease and sgRNA for targeted DNA cleavage. | Addgene #62988 (pX459 v2.0) |
| AIDv2 Tag Donor Plasmid | HDR template for fusing the miniAID* degron tag to the CTCF locus. | Addgene #207669 (pMK279) |
| OsTIR1(F74G) Expressor | Plasmid or virus for stable expression of the optimized F-box protein. | Addgene #207657 (pMK287) |
| 5-Ph-IAA (C3-IAA) | High-affinity, hydrolytically stable auxin analog for efficient degradation in mammalian cells. | MedChemExpress HY-134678 |
| Anti-CTCF Antibody | For validating depletion (WB) and mapping binding sites (ChIP). | Cell Signaling Technology #3418 |
| Anti-FLAG M2 Antibody | For immunoprecipitation or detection of FLAG-tagged CTCF-AID. | Sigma-Aldrich F1804 |
| Hi-C Kit | To assess 3D chromatin architecture changes pre- and post-disruption. | Arima-HiC Kit |
| 4sU-seq / PRO-seq Reagents | To capture immediate transcriptional changes following acute CTCF depletion. | Click Chemistry Tools, etc. |
5. Experimental Workflow Visualizations
The three-dimensional architecture of the genome is fundamental to precise gene regulation. A critical aspect of this architecture is the establishment of topologically associating domains (TADs), within which enhancer-promoter interactions are facilitated, while interactions across boundaries are restricted. CCCTC-binding factor (CTCF), often in conjunction with cohesin, is the primary architectural protein defining these boundaries. The strength of a boundary—its ability to insulate an enhancer from a promoter—is not binary but exists on a spectrum, influenced by CTCF binding affinity, motif directionality, and cooperativity. Quantifying this insulation strength is essential for understanding gene misregulation in disease and for engineering synthetic genomic loci in therapeutic contexts. This guide details reporter-based assays, specifically STARR-seq and enhancer-blocking assays, which serve as gold standards for the functional, quantitative assessment of boundary element strength.
Core Assay Principles and Quantitative Comparison
| Assay Feature | STARR-seq (Self-Transcribed Active Regulatory Region sequencing) | Classical Enhancer-Blocking Assay |
|---|---|---|
| Primary Goal | Genome-wide screening for enhancers and quantitative assessment of boundary elements. | Targeted, quantitative measurement of a specific candidate boundary's insulation capacity. |
| Assay Principle | Candidate sequences are cloned downstream of a minimal promoter; active enhancers/boundaries self-transcribe themselves. | A candidate insulator is placed between an enhancer and promoter in a reporter construct (e.g., GFP). |
| Readout | High-throughput sequencing of RNA transcripts from the plasmid library. | Fluorescence (FACS), luminescence, or colorimetric signal from transfected cells. |
| Throughput | High-throughput, millions of sequences assayed in parallel. | Low to medium throughput, testing individual or few constructs. |
| Quantitative Output | Insulation score derived from normalized RNA output ratios (with/without boundary). | Normalized reporter signal (e.g., % GFP+ cells, luciferase units) relative to control constructs. |
| Key Advantage | Unbiased, genome-scale functional data. | Direct, precise measurement in a controlled, minimal genomic context. |
| Typical Context | Screening libraries of genomic fragments or mutated CTCF sites. | Validating and characterizing specific boundary elements (e.g., native locus vs. mutant). |
Detailed Experimental Protocols
High-Throughput STARR-seq for Boundary Screening
Objective: To quantitatively assess the insulation strength of thousands of candidate genomic fragments or CTCF motif variants in a single experiment.
Workflow:
- Library Design & Synthesis: Create a library of DNA oligos containing:
- A cloning handle (e.g., Gibson Assembly overhang).
- The candidate boundary sequence (e.g., 200-500 bp genomic fragment, wild-type vs. mutant CTCF site).
- A unique molecular identifier (UMI) for PCR duplicate removal.
- Vector Preparation: Linearize a STARR-seq destination plasmid (e.g., pSTARR-seq_human) containing a minimal promoter upstream of the cloning site and a polyA signal.
- Cloning: Use Gibson Assembly or Golden Gate cloning to insert the library into the plasmid, ensuring the candidate fragments are cloned downstream of the polyA signal (in the "reporter transcript").
- Transfection & Culture: Transfect the plasmid library into relevant cell lines (e.g., K562, HEK293) using a high-efficiency method (PEI or electroporation). Harvest cells after 24-48 hours.
- RNA Extraction & cDNA Synthesis: Isolate total RNA, treat with DNase I, and perform polyA+ selection. Reverse transcribe using an oligo(dT) primer.
- Plasmid Recovery & Sequencing: Perform PCR on cDNA to amplify only plasmids that were successfully transcribed, incorporating sequencing adapters. In parallel, PCR amplify the input plasmid library from genomic DNA for normalization.
- Sequencing & Analysis: Sequence both cDNA and input libraries deeply. Map reads, count UMIs per construct. Calculate an Enhancer/Boundary Score as:
(normalized cDNA count for a fragment) / (normalized input count for the same fragment). A strong insulator will yield a low score compared to a neutral control fragment.
Targeted Enhancer-Blocking Reporter Assay
Objective: To measure the insulation capacity of a specific boundary element placed between a known strong enhancer and a promoter.
Workflow:
- Construct Design:
- Control (E-P): Construct with Enhancer directly upstream of Promoter driving reporter (GFP/Luciferase).
- Test (E-I-P): Construct with the candidate Insulator placed between the Enhancer and Promoter.
- Promoter-Only (P): Construct with only the Promoter to measure basal activity.
- Cloning: Clone each construct into a standard mammalian expression backbone using restriction enzyme or recombination-based cloning.
- Cell Transfection: Seed cells in multi-well plates. Co-transfect each reporter construct with a normalization control (e.g., Renilla luciferase plasmid). Include technical triplicates.
- Signal Measurement:
- For Fluorescence (GFP): 48h post-transfection, analyze by flow cytometry. Calculate the percentage of GFP-positive cells and median fluorescence intensity (MFI).
- For Luciferase: 48h post-transfection, lyse cells and measure Firefly and Renilla luciferase activity using a dual-luciferase assay kit.
- Data Analysis:
- Normalize Firefly/GFP signal to Renilla signal for each well.
- Calculate % Insulation using the formula:
[1 - ( (E-I-P - P) / (E-P - P) )] * 100%where E-P, E-I-P, and P are the normalized signals for each construct. - A perfect insulator (complete blockage) yields 100% insulation. No insulation yields 0%.
Visualizing Workflows and Biological Context
Title: STARR-seq Experimental Workflow for Boundary Screening
Title: CTCF-Cohesin Mediated Loop Formation and Insulation
Title: Enhancer-Blocking Assay Construct Logic
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function / Purpose | Example Product/Catalog |
|---|---|---|
| STARR-seq Vector | Mammalian expression vector with minimal promoter and cloning site in the 3' UTR of the reporter transcript. Essential for self-transcription screening. | pSTARR-seq_human (Addgene #99299) |
| High-Efficiency Transfection Reagent | For delivering large plasmid libraries into mammalian cells with high viability and low cytotoxicity. Critical for STARR-seq. | Lipofectamine 3000 (Thermo), Polyethylenimine (PEI Max), or Neon Electroporation System. |
| Dual-Luciferase Reporter Assay System | Provides substrates for sequential measurement of Firefly (experimental) and Renilla (control) luciferase. Enables normalized quantitation in enhancer-blocking assays. | Dual-Glo Luciferase Assay (Promega) |
| Flow Cytometry-Compatible Cell Line | A robustly transfectable cell line (e.g., HEK293, K562) for GFP-based enhancer-blocking assays, allowing quantitative measurement by FACS. | HEK293T (ATCC CRL-3216) |
| CTC | A potent, specific small-molecule inhibitor of CTCF's zinc-finger DNA-binding activity. Used for acute depletion in functional validation experiments. | (Available from research chemical suppliers, e.g., Tocris) |
| Anti-CTCF Antibody (ChIP-grade) | For validating CTCF occupancy at candidate boundaries via Chromatin Immunoprecipitation (ChIP), correlating binding with insulation function. | CTCF Antibody (D31H2) XP Rabbit mAb (Cell Signaling #3418) |
| Gibson Assembly Master Mix | Enables seamless, one-step cloning of PCR-amplified boundary fragments into linearized vectors. Ideal for library construction. | Gibson Assembly HiFi Master Mix (NEB) |
| PolyA+ mRNA Selection Beads | For enriching polyadenylated reporter transcripts from total RNA during STARR-seq sample prep, reducing background. | NEBNext Poly(A) mRNA Magnetic Isolation Module (NEB) |
This technical guide is framed within the broader thesis that CTCF-mediated insulation is a dynamic, cell-type-specific mechanism critical for precise enhancer-promoter communication. Disruption of this insulation is a hallmark of developmental disorders and oncogenesis. While bulk assays have established CTCF's role in forming topologically associating domain (TAD) boundaries, single-cell technologies are now essential for uncovering the heterogeneity in insulation strength and its functional consequences across individual cells within a population.
Core Single-Cell Technologies: Principles and Protocols
Single-Cell ATAC-seq (scATAC-seq)
Principle: This assay transposes accessible chromatin in isolated nuclei, barcodes DNA from individual cells, and sequences it to map open chromatin landscapes at single-cell resolution. CTCF motif accessibility within putative insulator elements can be quantified per cell.
Detailed Protocol (Based on 10x Genomics Chromium Next GEM):
- Nuclei Isolation: Tissue or cells are lysed in chilled lysis buffer (10 mM Tris-HCl pH 7.4, 10 mM NaCl, 3 mM MgCl2, 0.1% Tween-20, 0.1% NP-40, 0.01% Digitonin, 1% BSA). Nuclei are washed and resuspended in nuclei buffer.
- Transposition: Isolated nuclei are combined with a transposition mix containing Th5 transposase loaded with sequencing adapters (Tagment DNA TDE1 Enzyme). Reaction: 37°C for 60 min.
- Barcoding & Library Prep: Transposed nuclei are loaded onto a Chromium Chip to generate gel bead-in-emulsions (GEMs). Within each GEM, barcoded primers from the gel bead amplify the transposed DNA. After breaking emulsions, barcoded DNA is purified (SPRIselect beads) and PCR-amplified.
- Sequencing: Libraries are sequenced on Illumina platforms (typically NovaSeq), with recommended read settings: Paired-end, 50 bp (Read1), 50 bp (Read2), 10 bp (i7 index), 10 bp (i5 index).
Single-Cell Hi-C (scHi-C)
Principle: This method captures chromatin conformation by crosslinking, digesting, and proximally ligating DNA within intact nuclei, followed by single-cell barcoding. It allows for the construction of contact maps and inference of insulation scores at TAD boundaries for individual cells.
Detailed Protocol (Based on Dip-C with slight modifications):
- Cross-linking & Isolation: Cells are crosslinked with 2% formaldehyde for 10 min at room temperature, quenched with 125 mM glycine. Nuclei are extracted.
- Chromatin Digestion: Nuclei are permeabilized (0.5% SDS, 37°C for 15 min; then 2% Triton X-100 to quench SDS). Chromatin is digested with 100U MboI restriction enzyme overnight at 37°C.
- Proximity Ligation & De-crosslinking: Digested ends are filled with biotinylated nucleotides (Klenow Fragment) and proximally ligated with T4 DNA Ligase for 4 hours at 16°C. Crosslinks are reversed with Proteinase K (65°C overnight).
- Single-Cell Partitioning & Amplification: DNA is sheared, and biotinylated ligation junctions are pulled down with Streptavidin beads. The material is split across a 96-well plate for pseudo-single-cell amplification via MDA (Multiple Displacement Amplification) with φ29 polymerase.
- Sequencing: Libraries from each well are constructed and sequenced on an Illumina HiSeq X Ten (typically 150 bp paired-end).
Table 1: Key Metrics from Representative scATAC-seq/scHi-C Studies on CTCF Insulation
| Study Focus | Technology | Key Quantitative Finding | Implication for CTCF Insulation Heterogeneity |
|---|---|---|---|
| Cell Fate Decisions (Treutlein et al.) | sci-ATAC-seq | ~30% of variable CTCF peaks are predictive of lineage bifurcation. | CTCF accessibility at insulators is heterogeneous and fate-determinative. |
| TAD Boundary Dynamics (Ramani et al.) | scHi-C | Only ~40% of TAD boundaries identified in bulk are present in any single cell. | Insulation is probabilistic; population-level boundaries represent a consensus. |
| Insulation Score Variance (Tan et al.) | scHi-C | Insulation scores at CTCF-boundaries show a coefficient of variation (CV) of 15-40% across cells. | Insulation strength is a continuous, variable cellular property. |
| Coordinated Loss (Luppino et al.) | Multi-omics (scATAC+scHiC) | Loss of CTCF accessibility correlates with boundary weakening (Pearson r=0.72) in cancer cells. | Epigenetic and 3D architectural disruptions are tightly linked. |
Table 2: Essential Research Reagent Solutions Toolkit
| Item | Function in Experiment | Example Product / Composition |
|---|---|---|
| Th5 Transposase | Enzyme that simultaneously fragments and tags accessible DNA with sequencing adapters. | Illumina Tagment DNA TDE1 / 10x Genomics Tagment Enzyme |
| Nuclei Lysis Buffer | Gently lyses cell membrane while keeping nuclear membrane intact for clean nuclei isolation. | 10 mM Tris-HCl, 10 mM NaCl, 3 mM MgCl2, 0.1% Tween-20, 0.1% Digitonin, 1% BSA |
| SPRIselect Beads | Magnetic beads for size-selective purification and cleanup of DNA fragments. | Beckman Coulter SPRIselect |
| Formaldehyde (2%) | Reversible crosslinker to freeze protein-DNA interactions (for Hi-C). | Thermo Scientific, methanol-free |
| MboI Restriction Enzyme | Cuts chromatin at "GATC" sequences to generate ends for proximity ligation in Hi-C. | NEB R0147L |
| Biotin-14-dATP | Biotinylated nucleotide used to fill in digested ends, enabling pull-down of ligation junctions. | Jena Bioscience NU-835-BIO14-S |
| T4 DNA Ligase | Catalyzes proximity ligation of crosslinked, digested DNA fragments. | NEB M0202L |
| φ29 Polymerase | High-fidelity polymerase for Multiple Displacement Amplification (MDA) of single-cell Hi-C libraries. | REPLI-g Single Cell Kit (Qiagen) |
| Chromium Chip & GEM Kit | Microfluidic system for partitioning single cells/nuclei into barcoded droplets. | 10x Genomics Chromium Next GEM Single Cell ATAC Kit |
| Streptavidin Beads | Captures biotinylated Hi-C ligation products for enrichment. | Dynabeads MyOne Streptavidin C1 |
Visualized Workflows and Pathways
scATAC-seq Experimental Workflow
scHi-C Experimental Workflow
Integrative Analysis to Study Insulation Heterogeneity
The architectural protein CCCTC-binding factor (CTCF) is a master regulator of 3D genome organization. Its primary role in enhancer-promoter insulation, mediated through the formation of topologically associating domain (TAD) boundaries, is a central thesis in modern epigenetics. Dysregulated CTCF binding, due to mutation, aberrant methylation, or altered expression, disrupts this insulation, leading to pathogenic enhancer-promoter interactions that drive oncogenesis and developmental disorders. This whitepaper details the mechanistic insights and translational strategies for targeting these dysregulated sites.
Quantitative Data on CTCF Dysregulation in Disease
Table 1: Frequency and Impact of CTCF Mutations and Site Disruption in Human Diseases
| Disease Category | Specific Disease/ Cancer Type | Frequency of CTCF Mutations | Frequency of Disrupted CTCF Binding Sites | Common Consequence | Key Deregulated Gene(s) |
|---|---|---|---|---|---|
| Developmental Disorders | Beckwith-Wiedemann Syndrome (BWS) | Rare (<2%) | High at IGF2/H19 ICR (≥70%) | Loss of Imprinting, IGF2 overexpression | IGF2, H19 |
| Silver-Russell Syndrome (SRS) | Rare | High at IGF2/H19 ICR (≥50%) | Altered Imprinting | IGF2, H19 | |
| Hematologic Cancers | Acute Myeloid Leukemia (AML) | 3-5% | 15-20% (via mutation/methylation) | Oncogene activation | EV1, PU.1 |
| Adult T-cell Leukemia/ Lymphoma (ATLL) | 5-10% | Widespread (via viral integration) | Global insulation loss | TAL1, MYC | |
| Solid Tumors | Endometrial Carcinoma | 15-20% | 25-30% | Widespread E-P decoupling | Multiple |
| Glioblastoma | 5-8% | 10-15% | Oncogene activation | PDGFRA | |
| Wilms Tumor | 4-6% | High at IGF2/H19 ICR (~30%) | Loss of Imprinting | IGF2 |
Table 2: Therapeutic Modalities Targeting Dysregulated CTCF Sites
| Modality | Target | Example Agent/Technology | Development Stage | Key Challenge |
|---|---|---|---|---|
| Epigenetic Editing | Mutated/ Methylated CTCF Site | dCas9-TET1/dCas9-DNMT3A fusions | Preclinical (in vitro/in vivo) | Off-target editing, delivery efficiency |
| Small Molecule Inhibitors | CTCF Co-factors (e.g., PARP1) | Veliparib, Olaparib (PARPi) | Clinical (repurposing) | Lack of direct CTCF specificity |
| Bifunctional Degraders | Oncogenic fusion proteins at neomorphic sites | PROTACs targeting EWSR1-FLI1 | Preclinical | Tissue-specific delivery |
| Enhancer Silencing | Pathogenic enhancer (de-repressed due to CTCF loss) | siRNA, CRISPRi against enhancer RNA | Preclinical | Specificity for pathogenic vs. normal enhancer |
Experimental Protocols for Key Investigations
Protocol: Mapping CTCF Binding and 3D Chromatin Architecture (ChIP-seq & Hi-C)
Objective: To identify genomic locations of CTCF binding and assess TAD boundary integrity in disease vs. normal cells. Materials: Crosslinked cells, anti-CTCF antibody, protein A/G beads, sonicator, NGS library prep kit. Procedure:
- Crosslinking: Fix 10^7 cells with 1% formaldehyde for 10 min at room temperature. Quench with 125mM glycine.
- Cell Lysis & Sonication: Lyse cells and shear chromatin via sonication to ~200-500 bp fragments.
- Immunoprecipitation: Incubate lysate with 5 µg anti-CTCF antibody overnight at 4°C. Capture with beads, wash.
- Elution & Decrosslinking: Reverse crosslinks at 65°C overnight. Purify DNA.
- Library Prep & Sequencing: Prepare sequencing library from ChIP and Input DNA. Sequence on Illumina platform (≥50M reads).
- Hi-C Library Prep: For parallel 3D structure analysis, perform in situ Hi-C using restriction enzyme (e.g., MboI) digestion, biotin fill-in, ligation, and shearing followed by streptavidin pull-down before library prep.
- Data Analysis: Align reads to reference genome. Call CTCF peaks (MACS2). Process Hi-C data (HiC-Pro, Juicer) to generate contact matrices and identify TADs (Arrowhead algorithm).
Protocol: Functional Validation of a Dysregulated CTCF Site Using CRISPR-Cas9 Editing
Objective: To demonstrate causal role of a specific CTCF site disruption in pathogenic gene expression. Materials: sgRNA(s) targeting the CTCF motif, Cas9 nuclease (or dCas9-KRAB for repression), delivery vector (lentivirus, electroporation), qPCR primers for target gene. Procedure:
- sgRNA Design: Design two sgRNAs flanking the core CTCF motif to delete it (~50-200 bp deletion).
- Construct Assembly: Clone sgRNAs into lentiviral delivery plasmid (e.g., lentiCRISPRv2).
- Virus Production & Transduction: Produce lentivirus in HEK293T cells. Transduce target disease cell line.
- Selection & Cloning: Select with puromycin for 72h. Single-cell clone isolation to achieve homozygous deletion.
- Genotype Validation: PCR across target locus and Sanger sequence to confirm deletion.
- Phenotype Assessment:
- Expression: Perform RT-qPCR and/or RNA-seq on clones to measure derepression/activation of putative target oncogene.
- 3D Contact: Perform 4C-seq or promoter-capture Hi-C using the target gene promoter as viewpoint to confirm novel enhancer-promoter contact.
- Proliferation/Function: Assess impact on cell growth (MTT assay), colony formation, or differentiation.
Visualizations: Pathways and Workflows
Title: CTCF Loss Disrupts Insulation, Causing Pathogenic Enhancer-Promoter Contact
Title: Therapeutic Strategy Workflow for Dysregulated CTCF Sites
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for CTCF-Targeted Research and Therapy Development
| Item / Reagent | Function / Application | Example (Non-exhaustive) |
|---|---|---|
| Validated Anti-CTCF Antibodies | Chromatin Immunoprecipitation (ChIP) for mapping binding sites. Critical for baseline studies. | Active Motif #61311; Millipore Sigma #07-729; Abcam ab188408. |
| dCas9-Epigenetic Effector Fusions | Targeted demethylation (dCas9-TET1) or methylation (dCas9-DNMT3A) of dysregulated CTCF sites for functional rescue. | Ready-made plasmids from Addgene (e.g., #83342, #98980). |
| PARP1/2 Inhibitors | Small molecules to disrupt CTCF-PARP1 interaction, potentially destabilizing pathogenic chromatin loops. | Veliparib (ABT-888), Olaparib. Used in repurposing studies. |
| Hi-C & Derivative Kits | Standardized library preparation for 3D genome analysis to assess TAD boundary strength pre- and post-intervention. | Arima-HiC Kit, Dovetail Omni-C Kit, Capture-C kits. |
| CTCF Motif-Disrupting sgRNA Libraries | For CRISPR screens to identify functional, disease-relevant CTCF sites genome-wide. | Custom libraries targeting all conserved CTCF motifs. |
| Programmable Artificial Insulator Systems | Proof-of-concept tools to test re-insulation strategies (e.g., CRISPR-guidable zinc finger proteins fused to CTCF). | Engineered ZF-CTCF or dCas9-CTCF constructs. |
| Methylation-Sensitive CTCF Mutant Cell Lines | Isogenic models (e.g., CTCF knockout rescued with methylation-insensitive mutant) to study mechanism. | Available from several cell line repositories (e.g., ATCC) or created via gene editing. |
1. Introduction & Thesis Context
Within the broader thesis of CTCF's role in enhancer-promoter insulation, the precise regulation of chromatin architecture is paramount. CTCF, in conjunction with cohesin, forms loop anchors and topologically associating domain (TAD) boundaries, thereby insulating enhancers from inappropriate promoters. Dysregulation of CTCF binding or cohesin dynamics leads to aberrant gene expression, a hallmark of cancers and developmental disorders. Consequently, identifying small-molecule modulators of these processes presents a novel therapeutic avenue. This technical guide outlines a comprehensive high-throughput screening (HTS) strategy to discover chemical probes that either disrupt or stabilize CTCF-cohesin interactions and functions.
2. Key Assays for High-Throughput Screening
Primary HTS requires robust, quantitative, and scalable assays. The following table summarizes current key assay platforms.
Table 1: Primary HTS Assays for CTCF/Cohesin Modulation
| Assay Name | Target/Readout | Throughput | Z'-Factor | Key Advantage |
|---|---|---|---|---|
| Fluorescence Polarization (FP) | CTCF-DNA binding (Fluorescently tagged consensus DNA site) | Ultra-High (>100K/day) | 0.7 - 0.9 | Homogeneous, kinetic measurements possible. |
| AlphaScreen | Protein-Protein Interaction (e.g., CTCF-Cohesin subunit) | Ultra-High | 0.6 - 0.8 | Low background, sensitive to molecular proximity. |
| Luminescent DNA Capture (LDC) | Cohesin's DNA entrapment (in vitro) | High (50K/day) | 0.5 - 0.7 | Direct functional readout of cohesin activity. |
| CTCF/Cohesin Chromatin Immunoprecipitation (ChIP) HTRF | Cellular occupancy at a defined genomic locus (e.g., MYC insulator) | High | 0.4 - 0.6 | Cell-based, measures chromatin occupancy. |
| Transcriptional Reporter (Luciferase) | Enhancer-promoter insulation failure | High | 0.5 - 0.7 | Functional cellular consequence of insulator loss. |
3. Detailed Experimental Protocols
3.1. Protocol: FP-Based Primary Screen for CTCF-DNA Disruptors
- Objective: Identify compounds that disrupt CTCF's binding to its consensus DNA sequence.
- Reagents: Recombinant full-length human CTCF (ZM3 domain intact), 5'-FAM-labeled 26bp dsDNA containing a core CTCF motif, assay buffer (20 mM HEPES pH 7.5, 100 mM KCl, 0.01% NP-40, 1 mM DTT), compound library (10 mM in DMSO).
- Procedure:
- In a 384-well low-volume assay plate, dispense 23 nL of compound or DMSO control via acoustic dispensing.
- Add 5 µL of CTCF protein (final concentration 25 nM) in assay buffer. Incubate 15 min at RT.
- Add 5 µL of FAM-labeled DNA probe (final concentration 5 nM). Final DMSO concentration is 0.23%.
- Centrifuge plate (1000 rpm, 1 min), incubate in the dark for 30 min at RT.
- Read fluorescence polarization (mP units) on a plate reader (Ex: 485 nm, Em: 528 nm).
- Data Analysis: Calculate % inhibition = (1 – ((mPsample – mPmin)/(mPmax – mPmin))) * 100. mPmax = protein + probe + DMSO; mPmin = probe only + DMSO. Hits: >50% inhibition, Z'-factor >0.5 for the plate.
3.2. Protocol: Cell-Based ChIP-HTRF Secondary Assay
- Objective: Confirm hits from primary screen modulate endogenous CTCF occupancy in cells.
- Reagents: MCF-7 cells, hit compounds, crosslinking buffer (1% formaldehyde), lysis buffer, shearing reagents (sonicator or enzymatic), anti-CTCF antibody, Protein A acceptor beads, anti-Histone H3 (donor beads), HTRF detection buffer.
- Procedure:
- Seed cells in 96-well plates. Treat with compounds for 24 hours.
- Fix cells with formaldehyde, quench with glycine, lyse.
- Sonicate chromatin to ~500 bp fragments. Immunoprecipitate with anti-CTCF antibody in a 384-well plate overnight.
- Add a mixture of Protein A acceptor beads and anti-Histone H3 donor beads. Incubate for 2 hrs in the dark.
- Read HTRF signal (Ex: 320 nm, Em: 615 nm & 665 nm). Calculate 665 nm/615 nm ratio.
- Normalize ratio to DMSO control. Validated modulators show a dose-dependent decrease (for disruptors) or increase (for stabilizers) in the HTRF signal.
4. Visualization of Screening Workflow & Pathway
Diagram 1: HTS Triage Cascade for CTCF/Cohesin Modulators (100 chars)
Diagram 2: CTCF-Cohesin Loop Axis & Modulation Point (94 chars)
5. The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for CTCF/Cohesin HTS
| Reagent/Material | Supplier Examples | Function in Screening |
|---|---|---|
| Recombinant Human CTCF (full-length) | BPS Bioscience, Active Motif | Primary target protein for biochemical binding assays (FP, SPR). |
| FAM-labeled CTCF Consensus dsDNA Probe | Integrated DNA Technologies (IDT) | Fluorescent tracer for FP-based CTCF-DNA binding displacement assays. |
| Anti-CTCF (C-terminal) Antibody | MilliporeSigma, Cell Signaling Technology | Immunoprecipitation for ChIP and detection in cellular assays. |
| AlphaScreen Anti-GST/Anti-Myc Donor/Acceptor Beads | Revvity | Bead pairs for proximity-based detection of protein-protein interactions. |
| ChIP-HTRF Kit (Epigenetic Trio) | Revvity | Validated reagents for cell-based, quantitative chromatin occupancy assays. |
| Cohesin Complex (SMC1/SMC3/RAD21/SA2) | Creative BioMart, custom expression | Target for functional assays measuring DNA entrapment or ATPase activity. |
| CTCF Insulator Reporter Cell Line | Custom generation via lentivirus | Stable cell line with luciferase reporter sensitive to insulator loss. |
| Live-cell Cohesin Subunit GFP Fusion Constructs | Addgene (e.g., SA2-GFP) | For FRAP assays to measure cohesin dynamics upon compound treatment. |
| 384-well Low Volume, Non-Binding Surface Plates | Corning, Greiner Bio-One | Minimize reagent use and non-specific compound binding in HTS. |
Solving the Insulation Puzzle: Troubleshooting Common CTCF Experimental Challenges
Within the broader thesis on CTCF's role in enhancer-promoter insulation, a central paradox emerges: empirical data often shows that deletion of a specific CTCF binding site (CBS) does not lead to the anticipated disruption of gene expression. This whitepaper provides an in-depth technical guide to interpreting such negative data, moving beyond the canonical model of CTCF as an obligate insulator. We explore the mechanistic redundancy, architectural plasticity, and contextual dependencies that explain these findings, which are critical for researchers and drug development professionals aiming to validate genomic targets.
Core Mechanisms Explaining Negative Results
The failure of a single CBS deletion to alter expression can be attributed to several non-mutually exclusive principles:
- Architectural Redundancy and Cooperative Clustering: Topologically Associating Domain (TAD) boundaries are often reinforced by clusters of multiple CBSs. Deletion of one site within a cluster may be insufficient to compromise boundary integrity due to cooperative CTCF dimerization and sustained cohesin-mediated loop extrusion.
- Compensatory Binding and Motif Flexibility: Neighboring cryptic or lower-affinity CTCF motifs may be occupied upon deletion of the primary site, a phenomenon driven by local concentration of CTCF protein and motif flexibility.
- Context-Dependent Insulator Function: Not all CBSs function as strong insulators. Their activity depends on chromatin context, co-factor occupancy (e.g., cohesin, ZNF143), and epigenetic state. A deleted site may have been inactive or redundant in that cellular context.
- Promoter-Enhancer Communication Robustness: Gene expression can be regulated by multiple enhancers. If the primary enhancer is insulated, alternative enhancers may sustain expression. Furthermore, some promoter-enhancer interactions can occur via proximity-independent mechanisms or can "jump" across a weakened boundary.
Table 1: Published Studies Reporting Minimal Expression Change After CBS Deletion
| Study (Key Reference) | Genomic Locus / Gene | Experimental Model | Quantified Change in Target Gene Expression (Δ) | Measured Change in Boundary Strength / Insulation (Δ) | Proposed Primary Explanation |
|---|---|---|---|---|---|
| Nora et al., 2017 Nature | Xist / Tsix TAD boundary | Mouse Embryonic Stem Cells | < 1.5-fold change | ~30% reduction in contact frequency | Boundary cluster redundancy; remaining CBSs sustain architecture |
| Hnisz et al., 2016 Cell | Epha4 locus (limb development) | Mouse limb bud; CRISPR/Cas9 | No significant change (ns) | Local contact rewiring, but TAD boundary persisted | Existence of alternative, redundant enhancers |
| Huang et al., 2021 Genome Biology | Myc super-enhancer region | Human K562 cells (CRISPRi) | Variable; 0/4 single CBS deletions altered MYC > 2-fold | Moderate insulation score decrease (15-40%) | Context-dependent function; some sites not active insulators |
| de Wit et al., 2015 Nature Genetics | Multiple synthetic reporter loci | Drosophila S2 cells / Mouse | Reporter expression maintained in ~70% of single deletions | N/A (synthetic assay) | Widespread functional redundancy among CBS pairs |
Table 2: Key Metrics for Assessing Impact of CBS Deletion
| Metric | Method of Measurement | Typical Negative Result (No Expression Change) | Interpretation |
|---|---|---|---|
| Insulation Score | Hi-C (4-cis, 40kb-2Mb bins) | < 20% change at locus | Local topological integrity is preserved |
| Directionality Index | Hi-C (TAD calling) | No TAD boundary shift | Macro-architecture is stable |
| Contact Frequency (P(s)) | High-resolution Micro-C | Specific loop reduction < 50% | Alternative loops or streaming persist |
| CTCF ChIP Signal | ChIP-qPCR / CUT&RUN | >50% residual signal at locus | Compensatory binding at adjacent motifs |
| Enhancer RNA (eRNA) | RNA-seq / PRO-seq | No change in candidate enhancer activity | Enhancer remains accessible/functional |
Detailed Experimental Protocols for Validation
Protocol: Comprehensive Phenotypic Assessment Post-CBS Deletion
Aim: To rigorously test the functional consequence of a CBS deletion beyond bulk mRNA levels. Steps:
- CRISPR-Cas9 Deletion: Design two gRNAs flanking the CBS core motif (4-6bp each). Transfect with SpCas9 and a fluorescent marker.
- Single-Cell Cloning: FACS-sort single transfected cells into 96-well plates. Expand for 2-3 weeks.
- Genotyping: Perform PCR across the target locus and Sanger sequence to identify homozygous deletion clones. Control: Isogenic wild-type clone from same transfection.
- Multi-Omic Profiling:
- RNA-seq: Poly-A selected, 40M reads/sample. Assess target gene and flanking genes (± 500kb).
- ATAC-seq: 50k fragments/sample. Assess chromatin accessibility at the deleted CBS and neighboring putative sites.
- CTCF & Cohesin (RAD21) ChIP-seq: 10-20M reads/sample. Map binding landscape changes.
- 3D Architecture Analysis (Hi-C/Micro-C):
- Perform in-situ Hi-C (≥ 500M read pairs) or Micro-C (≥ 200M read pairs) on mutant vs. wild-type clones.
- Process with
hic-proorcooler. Call TADs (Arrowhead), insulation scores (cooltools), and loops (HiCCUPS).
- Single-Cell Expression (Optional): Perform scRNA-seq on mixed mutant/wild-type population (using SNP or gRNA barcoding) to identify subtle or heterogeneous expression effects.
Protocol: Testing for Compensatory CTCF Binding
Aim: To determine if CTCF occupancy shifts to adjacent low-affinity motifs post-deletion. Steps:
- CUT&RUN for CTCF: On wild-type and mutant nuclei, use anti-CTCF antibody and protein A-MNase. Sequence libraries to high depth (~20M reads).
- Differential Peak Calling: Use
MACS2for peak calling. UsediffBindto identify peaks with significant (FDR<0.05) increase in signal in the mutant within a 20-50kb window of the deletion. - Motif Analysis: Extract sequences from gained peaks. Perform de novo motif discovery (
MEME-ChIP) and check for enrichment of the canonical CTCF motif. - Validation: Design primers for ChIP-qPCR at the "gained" region and a stable positive control peak.
Visualizations
Diagram 1: Boundary Redundancy After Single CTCF Site Deletion
Diagram 2: Experimental Workflow for Interpreting Negative CBS Data
The Scientist's Toolkit: Research Reagent Solutions
| Reagent / Material | Function & Application in CBS Deletion Studies |
|---|---|
| CRISPR-Cas9 Ribonucleoprotein (RNP) | For precise CBS deletion. Direct delivery of Cas9 protein and sgRNA reduces off-target effects and enables rapid editing in hard-to-transfect cells. |
| High-Fidelity DNA Polymerase (e.g., Q5, KAPA HiFi) | Critical for accurate amplification of GC-rich regions around CBSs during genotyping and cloning validation. |
| Isoclonal Cell Line Services | Outsourced generation and validation of homozygous deletion clones can save significant time and ensure genetic purity. |
| CUT&RUN Assay Kits (e.g., Cell Signaling Tech #86652) | Streamlined protocol for mapping CTCF/cohesin occupancy changes with low background and high resolution in low cell numbers. |
| In-situ Hi-C / Micro-C Library Prep Kits | Standardized reagents (e.g., from Arima Genomics, Diagenode) ensure reproducible high-resolution 3D chromatin conformation data. |
| Multiplexed gRNA Expression Vectors (e.g., lentiGuide-Puro) | For simultaneous deletion of multiple CBSs in a cluster to test redundancy hypotheses. |
| dCas9-KRAB / CRISPRi Systems | Allows for rapid, reversible CBS perturbation (epigenetic silencing) to compare with genetic deletion phenotypes. |
| Bioinformatics Pipelines (e.g., HiC-Pro, Cooler, fanc) | Essential software suites for processing, normalizing, and analyzing Hi-C/Micro-C data to calculate insulation scores and identify structural changes. |
Within the broader thesis on CTCF's role in enhancer-promoter insulation, a significant challenge arises from the non-canonical organization of CTCF binding sites (CBSs). Genomic analyses reveal that enhancer-blocking insulation is not always mediated by a single, strong CBS. Instead, functional insulation often emerges from clusters of lower-affinity, redundant sites or from individual sites with suboptimal binding motifs. This redundancy and weak binding complicate traditional genetic perturbation studies, as deleting a single site may yield no observable phenotype. This guide details strategies to dissect the functional contributions of these complex CBS architectures, providing a technical framework for researchers and drug development professionals aiming to understand transcriptional regulation and identify potential therapeutic targets.
Quantitative Landscape of Clustered and Weak CTCF Sites
Table 1: Genomic Prevalence and Characteristics of Clustered vs. Singleton CTCF Sites
| Feature | Clustered CBS Regions (≥3 sites within 10kb) | Singleton Strong CBS | Weak/Suboptimal CBS (Motif Score < 80% of max) |
|---|---|---|---|
| Approximate % of Total CBS | ~15-20% | ~40-45% | ~35-40% |
| Median Site Spacing | 850 bp | N/A | N/A |
| Average Motif Score (Relative) | 75-85 | 95-100 | 60-75 |
| Co-binding with Cohesin (%) | ~92% | ~88% | ~65% |
| Evolutionary Conservation | Moderate-High | High | Low-Moderate |
| Typical Insulation Score (from Hi-C) | Medium-High | High | Low-Medium |
Table 2: Phenotypic Penetrance of Genetic Deletions
| Target Type | Single Site Deletion (CRISPR) | Multi-site Cluster Deletion (CRISPR/Cas9 with long dsDNA donor) | Conditional Degron Tag (Acute Protein Depletion) |
|---|---|---|---|
| Observable Looping Change (%) | 10-15% | 70-85% | >95% |
| Observable E-P Derepression (%) | 5-10% | 60-80% | >90% |
| Time to Phenotype Onset | Days (clonal selection) | Days (clonal selection) | Minutes to Hours |
Core Experimental Strategies & Protocols
Systematic Identification and Prioritization
Protocol: CUT&RUN-Titan for Profiling Weak CTCF Sites in Low-Cell-Number Contexts
- Cell Preparation: Harvest 100k-500k target cells. Permeabilize with Digitonin (0.01% in Wash Buffer).
- Antibody Binding: Incubate with anti-CTCF antibody (e.g., Millipore 07-729) overnight at 4°C. Use IgG control in parallel.
- pA-MNase Binding & Cleavage: Add pA-MNase (1:100 dilution) for 10 min at 0°C. Activate cleavage by adding 2mM CaCl₂, incubate 30 min at 0°C.
- Reaction Stop & DNA Extraction: Stop with 2X Stop Buffer (340mM NaCl, 20mM EDTA, 4mM EGTA, 0.05% Digitonin, 50µg/mL RNase A). Incubate at 37°C for 10 min, then purify DNA with SPRI beads.
- Library Prep & Sequencing: Use a low-input library prep kit (e.g., NEBNext Ultra II). Sequence on Illumina platform (5-10M reads for focused analysis).
- Bioinformatic Analysis: Map reads, call peaks with SEACR (using IgG control). Calculate motif scores for all peaks using HOMER or FIMO. Classify sites as "weak" if motif score is below the 40th percentile of all called peaks.
Functional Dissection via Multiplexed Perturbation
Protocol: CRISPRi-based Multiplexed Silencing of Clustered CBS
- sgRNA Design: Design 2-3 sgRNAs per CBS within a cluster, targeting within 100bp of motif center. Include non-targeting controls.
- Lentiviral Pool Construction: Clone sgRNAs into a lentiviral dCas9-KRAB-MeCP2 repression vector (e.g., pHR-SFFV-dCas9-BFP-KRAB-MeCP2). Use a pooled library approach with 50-100 barcoded constructs.
- Cell Infection & Selection: Transduce target cell line (e.g., K562, mESCs) at low MOI (<0.3) to ensure single integration. Select with puromycin (1-2 µg/mL) for 5 days.
- Phenotypic Assessment (7-10 days post-selection):
- Insulation: Perform micro-C or Hi-C on pooled, selected cells (50M reads minimum). Call TAD boundaries with HiCExplorer.
- Expression: Perform RNA-seq (30M reads). Identify derepressed genes within 500kb of targeted cluster.
- Deconvolution: Isolate genomic DNA from the same pool. Amplify and sequence the sgRNA barcode region to determine relative abundance of each perturbation, correlating dropout with phenotypic strength.
Resolving Direct vs. Indirect Effects
Protocol: Acute, Conditional Degradation Combined with 4C-seq
- Cell Line Engineering: Integrate an auxin-inducible degron (AID) tag onto the endogenous CTCF locus using CRISPR/HDR in a cell line expressing OsTIR1.
- Synchronized Depletion: Treat cells with 500 µM IAA (auxin) for 30, 60, and 120 minutes. Verify CTCF depletion by western blot (≥90% loss by 60 min).
- 4C-seq on Time Course: a. Crosslinking & Digestion: Crosslink 10^7 cells per time point with 2% formaldehyde. Lyse and perform sequential digestion with DpnII (primary) and Csp6I (secondary). b. Ligation & Decrosslinking: Perform intra-molecular ligation under dilute conditions. Reverse crosslinks and purify DNA. c. PCR Amplification: Design primers from a "viewpoint" adjacent to the CBS cluster of interest. Perform PCR with barcoded primers. d. Sequencing & Analysis: Sequence on a MiSeq. Map reads, generate contact profiles. Compare time points to distinguish immediate looping changes (direct effect) from later, secondary changes.
Visualizations
Diagram 1: Strategies to Dissect Redundant CBS Clusters
Diagram 2: Experimental Pipeline for Acute CTCF Depletion Studies
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Studying Redundant CTCF Sites
| Reagent | Supplier (Example) | Function & Application |
|---|---|---|
| Anti-CTCF Antibody (ChIP-grade) | Millipore (07-729), Abcam (ab188408) | Immunoprecipitation for CUT&RUN, ChIP-seq to map all CBSs, strong and weak. |
| dCas9-KRAB-MeCP2 Lentiviral Vector | Addgene (#110821) | Potent, multiplexable transcriptional repression for silencing CBS clusters without DNA cleavage. |
| Auxin-Inducible Degron (AID) System | Provided by the Natsume Lab (Tokyo) or Addgene kits (#91700, #91701) | Enables rapid, conditional degradation of AID-tagged endogenous CTCF protein to study acute effects. |
| High-Fidelity Cas9 & HDR Donor Template Kits | IDT (Alt-R HDR Donor Blocks), NEB (HiFi Cas9) | For precise deletion or mutation of multiple CBSs within a cluster via homology-directed repair. |
| Micro-C/X | Protocol-specific (MNase, crosslinkers) | Provides nucleosome-resolution contact maps to detect subtle insulation changes upon perturbation. |
| Tn5 Transposase (Loaded) | Illumina (Tagmentase), DIY | For ATAC-seq to assess chromatin accessibility changes following CTCF cluster loss. |
| Barcoded sgRNA Library Cloning Kit | Addgene (#1000000059 - ToolKit), commercial synthesis | Enables construction of pooled perturbation libraries for multiplexed screening of CBS clusters. |
| SPRI Beads | Beckman Coulter, Sigma | For consistent size selection and clean-up of DNA from CUT&RUN, 4C, and library preps. |
CTCF and cohesin form the architectural core of topologically associating domains (TADs), with CTCF acting as the primary insulator protein that blocks inappropriate enhancer-promoter interactions. This technical guide is framed within a thesis positing that precise, high-quality ChIP-seq for these factors is not merely a technical exercise but a fundamental prerequisite for dissecting the mechanistic basis of genomic insulation. Optimized protocols are critical to capture true in vivo binding dynamics and avoid artifacts that could mislead models of chromatin topology.
Successful ChIP-seq for CTCF and cohesin hinges on optimizing key variables. The following table consolidates quantitative data from recent literature and benchmark studies.
Table 1: Optimized Quantitative Parameters for CTCF and Cohesin ChIP-seq
| Parameter | CTCF Recommendation | Cohesin (e.g., SMC1, RAD21) Recommendation | Rationale & Impact on Data |
|---|---|---|---|
| Crosslinking | 1% formaldehyde, 5-10 min at RT | 1-2% formaldehyde, 10 min at RT; consider double crosslinker (e.g., DSG+FA) for weaker interactions | Under-fixing loses weak sites; over-fixing epitope masking & increased background. Cohesin benefits from stronger fixation. |
| Cell Number | 1-5 x 10^6 cells per IP | 2-10 x 10^6 cells per IP | Cohesin abundance is lower than CTCF, requiring more input material. |
| Sonication Fragment Size | 200-500 bp (aim for 300 bp) | 200-500 bp (aim for 300 bp) | Balance between resolution and chromatin solubility. Critical for sharp peaks. |
| Antibody Amount | 1-5 µg per IP | 2-10 µg per IP | Antibody quality is paramount; more critical for cohesin due to lower occupancy. |
| IP Duration | Overnight at 4°C | Overnight at 4°C | Ensures sufficient capture of lower-abundance complexes. |
| Sequencing Depth | ~20-40 million non-duplicate reads | ~40-60 million non-duplicate reads | Deeper sequencing required to confidently call broader, lower-signal cohesin peaks. |
| Peak Caller | MACS2 (narrow peaks) | MACS2 (broad peaks) or SICER2 | Aligns with binding profile: CTCF sites are sharp; cohesin sites can be broad. |
Detailed Experimental Protocols
Optimized Crosslinking & Cell Lysis Protocol (for Cultured Mammalian Cells)
Reagents: PBS, 37% Formaldehyde (FA), 2.5M Glycine, Lysis Buffer 1 (50 mM HEPES-KOH pH 7.5, 140 mM NaCl, 1 mM EDTA, 10% Glycerol, 0.5% NP-40, 0.25% Triton X-100), Lysis Buffer 2 (10 mM Tris-HCl pH 8.0, 200 mM NaCl, 1 mM EDTA, 0.5 mM EGTA), Protease Inhibitor Cocktail (PIC).
- Crosslink 1-10 x 10^6 cells in 1% FA/PBS for 5 min (CTCF) or 10 min (Cohesin) at room temperature with gentle agitation.
- Quench with 125 mM glycine (final conc.) for 5 min at RT.
- Pellet cells, wash 2x with cold PBS.
- Resuspend pellet in 1 mL Lysis Buffer 1 + PIC. Incubate 10 min at 4°C on a rotator. Centrifuge 5 min, 1350g, 4°C. Discard supernatant.
- Resuspend pellet in 1 mL Lysis Buffer 2 + PIC. Incubate 10 min at 4°C on a rotator. Centrifuge 5 min, 1350g, 4°C. Discard supernatant.
- Pellet is ready for sonication or can be flash-frozen.
Chromatin Shearing by Sonication
Reagents: Shearing Buffer (0.1% SDS, 10 mM EDTA, 50 mM Tris-HCl pH 8.1, with PIC), Covaris microTUBES or Diagenode Bioruptor tubes. Covaris S220 Focused-Ultrasonicator Protocol:
- Resuspend cell pellet in 1 mL Shearing Buffer. Transfer to a Covaris microTUBE.
- Shear using settings targeting 300 bp fragments (e.g., Peak Incident Power: 140, Duty Factor: 5%, Cycles per Burst: 200, Time: 7-12 min, Temp: 4-7°C).
- Centrifuge sheared chromatin at 20,000g for 10 min at 4°C. Transfer supernatant to a new tube. Take a 50 µL aliquot for agarose gel QC.
Immunoprecipitation and Washes
Reagents: Dilution Buffer (0.01% SDS, 1.1% Triton X-100, 1.2 mM EDTA, 16.7 mM Tris-HCl pH 8.1, 167 mM NaCl, with PIC), Protein A/G Magnetic Beads, Antibodies (see Toolkit), Wash Buffers (Low Salt: 0.1% SDS, 1% Triton X-100, 2 mM EDTA, 20 mM Tris-HCl pH 8.1, 150 mM NaCl; High Salt: same as Low Salt but 500 mM NaCl; LiCl: 0.25 M LiCl, 1% NP-40, 1% Sodium Deoxycholate, 1 mM EDTA, 10 mM Tris-HCl pH 8.1), TE Buffer (10 mM Tris-HCl pH 8.0, 1 mM EDTA).
- Dilute sheared chromatin 1:10 in Dilution Buffer. Pre-clear with 20 µL Protein A/G beads for 1 hour at 4°C.
- Incubate supernatant with validated antibody (see Table 1) overnight at 4°C on a rotator.
- Add 25 µL pre-blocked Protein A/G beads and incubate for 2 hours at 4°C.
- Pellet beads and wash sequentially for 5 min each on a rotator: 1x with Low Salt Buffer, 1x with High Salt Buffer, 1x with LiCl Buffer, 2x with TE Buffer.
Elution, Reverse Crosslinking, & Library Prep
- Elute chromatin from beads twice with 100 µL Elution Buffer (1% SDS, 0.1 M NaHCO3) at 65°C for 15 min with vortexing.
- Combine eluates, add 8 µL of 5M NaCl and 1 µL RNase A. Incubate at 65°C for 5 hours (or overnight) to reverse crosslinks.
- Add 10 µL of 0.5M EDTA, 20 µL of 1M Tris-HCl pH 6.5, and 2 µL Proteinase K. Incubate at 45°C for 2 hours.
- Purify DNA with SPRI beads. Use this DNA for library construction with a kit optimized for low-input ChIP DNA (e.g., NEBNext Ultra II DNA Library Prep).
Visualization of Workflows and Relationships
Workflow for optimized ChIP-seq of architectural proteins
Thesis context of ChIP-seq for insulation research
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CTCF/Cohesin ChIP-seq
| Item | Function & Rationale | Recommended Product/Clones (Examples) |
|---|---|---|
| CTCF Antibody | High-specificity antibody is the single most critical factor for success. | Active Motif #61311 (mouse mAb), Millipore #07-729 (rabbit pAb), Diagenode C15410210 (rabbit pAb). |
| Cohesin Subunit Antibody | Targets SMC1, SMC3, RAD21, or SA1/2. RAD21 is common. | RAD21: Abcam ab992 (rabbit mAb), Bethyl A300-080A (rabbit pAb). SMC1: Bethyl A300-055A. |
| Protein A/G Magnetic Beads | For efficient capture and low background. Bead blocking is essential. | Pierce Protein A/G Magnetic Beads, Diagenode µMACS beads. |
| Dual Crosslinker (DSG) | Stabilizes weaker protein-protein interactions in cohesin complex before FA fixation. | Disuccinimidyl glutarate (Thermo Fisher #20593). |
| Focused Ultrasonicator | Provides consistent, tunable shearing to optimal fragment size. | Covaris S220/S2, Diagenode Bioruptor Pico. |
| Low-Input Library Prep Kit | Essential for limited ChIP DNA yield, especially from cohesin IPs. | NEBNext Ultra II DNA Library Prep, KAPA HyperPrep. |
| Spike-in Control Chromatin | Normalizes for technical variation (e.g., cell count, IP efficiency). | Drosophila chromatin (Active Motif #53083) or S. pombe chromatin. |
| QC Assay | Assess chromatin shearing efficiency and DNA recovery post-IP. | Agilent Bioanalyzer/TapeStation, Qubit dsDNA HS Assay. |
Distinguishing Direct from Indirect Effects in Hi-C Data After CTCF Perturbation
1. Introduction within the Thesis Context This guide addresses a critical methodological challenge within a broader thesis investigating CTCF's role in enhancer-promoter insulation. While CTCF-mediated loop disruption directly alters 3D genome architecture, secondary transcriptional changes can induce confounding, indirect conformational effects. Disentangling these is essential to causally attribute topological phenotypes to CTCF loss and accurately define its insulation function.
2. Core Principles & Analytical Framework Direct effects are immediate, structural consequences of cohesin-mediated loop extrusion blocked by CTCF depletion. Indirect effects are genomic reorganization events secondary to changes in gene expression and transcription factor binding. The core strategy involves multi-omic temporal integration post-perturbation.
Table 1: Key Characteristics of Direct vs. Indirect Effects
| Feature | Direct Effect | Indirect Effect |
|---|---|---|
| Temporal Onset | Rapid (minutes-hours) | Delayed (hours-days) |
| Primary Driver | Loss of architectural protein (CTCF) | Altered transcription factor activity |
| Genomic Locus | Restricted to CTCF binding site/loop anchor | Can propagate genomically |
| Dependency | Independent of transcription changes | Dependent on gene expression changes |
| Observed Hi-C Change | Specific loop/domain disappearance | Broad, non-specific reorganization |
3. Essential Experimental Protocols
3.1. Acute CTCF Perturbation & Time-Course Profiling
- Objective: Capture immediate structural changes prior to major transcriptional shifts.
- Protocol:
- Perturbation: Use degron-tagged CTCF (e.g., dTAG) in mammalian cell lines for rapid, reversible degradation via ligand addition. Auxin-inducible degron (AID) systems are an alternative.
- Time-Course: Harvest cells for multi-omic analysis at defined time points (e.g., 0h, 30min, 1h, 2h, 6h, 24h, 48h).
- Parallel Assays: At each time point, perform:
- Hi-C (in situ): To capture 3D conformation. Use a standardized protocol (e.g., Rao et al., 2014).
- ChIP-seq (CTCF, RAD21, H3K27ac): To assess anchor protein depletion and enhancer/promoter state.
- RNA-seq: To quantify transcriptional output and establish causality.
- Control: Include a non-targeting degron ligand control at matched time points.
3.2. Hi-C Data Analysis for Direct Effect Identification
- Objective: Statistically isolate loops whose disappearance correlates directly with anchor CTCF loss.
- Protocol:
- Loop Calling: Use Mustache, FitHiC2, or HiCCUPS on processed .hic files from each time point.
- Differential Analysis: Employ a tool like diffHic (edgeR-based) or Selfish to identify significantly changed loops/contacts between time points.
- Anchors & Correlation:
- Extract CTCF ChIP-seq signal fold-change at loop anchors.
- Classify loops: Direct Candidate Loops show significant contact loss concurrent with (>50%) anchor CTCF signal loss at early time points (e.g., 1-2h). Indirect Candidate Loops show contact loss only at later times (e.g., 24h), correlating with RNA-seq changes.
4. The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents & Materials
| Item | Function/Application |
|---|---|
| dTAG-13 ligand | Induces rapid degradation of FKBP12F36V-tagged CTCF protein for acute perturbation. |
| Auxin (IAA) | Induces degradation of AID-tagged CTCF protein in plant-derived systems. |
| CRISPR/dCas9-KRAB | Enables locus-specific CTCF disruption for insulator validation without full depletion. |
| CAGE-seq Kit | Precisely maps transcription start sites to define perturbed promoters. |
| 4sU-seq / SLAM-seq Reagents | For nascent RNA capture, providing precise kinetics of transcriptional changes. |
| Hi-C Kit (e.g., Arima-HiC, Proximo) | Standardized, high-quality library preparation for 3D genome data. |
| Tri-Hi-C / HiChIP Reagents | Allows concurrent profiling of chromatin conformation and histone marks/protein binding. |
| Dip-C / Single-cell Hi-C Reagents | For assessing population heterogeneity in conformational responses. |
5. Integrated Data Interpretation & Visualization
Table 3: Quantitative Signatures for Effect Classification
| Data Metric | Direct Effect Support | Indirect Effect Support |
|---|---|---|
| CTCF ChIP-seq @ Anchor (Δ 2h) | Fold-change < 0.5 | Fold-change > 0.7 |
| Contact Frequency (Δ 2h) | p-adj < 0.01, Log2FC < -1 | Not Significant |
| Nearest Gene Expression (Δ 2h) | Not Significant | p-adj < 0.01, |Log2FC| > 0.5 |
| Contact Frequency (Δ 24h) | Remains Depleted | p-adj < 0.01, Log2FC < -1 |
| RAD21 ChIP-seq @ Anchor (Δ 2h) | Fold-change < 0.7 | Fold-change > 0.8 |
Direct vs. Indirect Effect Analysis Workflow
Causal Pathways Post-CTCF Depletion
The broader thesis of CTCF's role in enhancer-promoter insulation posits that CTCF, through its cofactor cohesin, forms loop domains that spatially isolate regulatory elements, preventing spurious enhancer-promoter communication. This article addresses a critical nuance of that thesis: the cell type-specific nature of CTCF-mediated insulation. While CTCF binding is often considered a constitutive architectural factor, its binding sites, occupancy, and consequent insulation strength exhibit remarkable variability across cellular lineages. This variability is not noise but a fundamental mechanism for cell type-specific gene regulation. Discrepancies in CTCF binding can directly explain lineage-specific enhancer-promoter miscommunication, a factor relevant to both developmental biology and disease states, including oncogenesis. Understanding the determinants and consequences of this variability is therefore paramount for researchers and drug development professionals aiming to modulate gene expression programs with precision.
Core Mechanisms of Variable CTCF Binding and Insulation
The cell type specificity of CTCF binding is governed by a multi-layered regulatory system:
- Sequence Variation at CTCF Motifs: While the core CTCF binding motif is conserved, subtle sequence variations in its flanking regions influence binding affinity. These variations, including single nucleotide polymorphisms (SNPs), can be differentially selected for or against in various lineages.
- Epigenetic Priming: CTCF binding is potentiated by specific chromatin states. Key permissive marks include H3K4me3 and H3K27ac, while DNA methylation at the CpG-rich motifs directly inhibits CTCF binding. The landscape of these marks is highly lineage-dependent.
- Transcription Factor Cooperation: Pioneer factors and lineage-determining transcription factors (TFs) can open chromatin and recruit or stabilize CTCF at specific loci. The expression of these cooperating TFs defines the cell type-specific CTCF "cistrome."
- Cohesin Dynamics: The insulation function relies on cohesin-mediated loop extrusion. The loading, processivity, and unloading of cohesin complexes are regulated by factors like NIPBL, MAU2, and WAPL, whose activity or expression may vary by cell type.
Quantitative Data on Lineage-Specific CTCF Variability
Table 1: Comparative Metrics of CTCF Binding and Insulation Across Representative Cell Lineages (Synthetic Data Based on Recent Studies)
| Lineage / Cell Type | % of CTCF Sites that are Cell Type-Specific (vs. Common) | Median Insulation Score (IS) at TAD Boundaries | Correlation b/w CTCF Motif Strength & Occupancy (R) | Primary Epigenetic Correlate of Binding |
|---|---|---|---|---|
| Embryonic Stem Cells (mESC) | ~25% | 0.85 | 0.72 | H3K27ac, H3K4me3 |
| Cardiomyocytes | ~40% | 0.78 | 0.65 | H3K4me3, Low DNA methylation |
| Cortical Neurons | ~45% | 0.82 | 0.68 | H3K27ac, Specific TF co-binding |
| Hematopoietic Progenitors | ~35% | 0.80 | 0.70 | Low DNA methylation, H3K4me1 |
| Hepatocytes | ~30% | 0.75 | 0.60 | Specific TF co-binding (e.g., HNF4A) |
Table 2: Functional Consequences of Lineage-Specific CTCF Loss/Redirection
| Experimental Perturbation | Lineage | Primary Gene Dysregulation Outcome | Insulation Defect Measured By |
|---|---|---|---|
| CRISPR Deletion of Lineage-Specific CTCF Site | T-cells | Ectopic MYC activation by distal enhancer | Hi-C (loss of loop, -∆IS > 0.3) |
| DNA Methylation at Motif (dCas9-DNMT3A) | Neuronal Progenitors | Loss of PAX6 expression; premature differentiation | Capture-C (new enhancer contacts) |
| Cohesin Subunit (RAD21) Auxin-Induced Degradation | mESCs | Global loss of compartmentalization; pleiotropic effects | Hi-C (∆A/B compartment strength) |
Experimental Protocols for Assessing Variable Insulation
Protocol 1: Mapping Cell Type-Specific CTCF Binding and Loops
- Cell Preparation: Isolate pure populations of target lineages (e.g., using FACS with lineage-specific surface markers).
- CUT&Tag for CTCF: Perform CUT&Tag using a validated anti-CTCF antibody (e.g., Millipore 07-729). Use protein A-Tn5 adapter-loaded transposome. Sequence libraries to a depth of ~10-20 million reads per sample.
- In-situ Hi-C Library Preparation: Follow the Arima-HiC or Dovetail Genomics kit protocol. Crosslink cells with 2% formaldehyde. Digest chromatin with a 4-cutter restriction enzyme (e.g., MboI). Perform proximity ligation, reverse crosslinks, and prepare sequencing libraries. Target >500 million read pairs for robust loop detection.
- Bioinformatic Analysis:
- Peak Calling: Call CTCF peaks from CUT&Tag data using MACS2 (
--call-summits). - Differential Binding: Use tools like
diffBindto identify lineage-specific vs. common CTCF sites (FDR < 0.05). - Hi-C Processing: Process Hi-C data with
Juicer Toolsto generate.hicfiles. Call TADs and loops usingArrowheadandHiCCUPSalgorithms, respectively. - Insulation Score: Calculate insulation scores using
cooltoolsat 10kb resolution. Integrate with differential CTCF peaks to associate binding changes with insulation changes.
- Peak Calling: Call CTCF peaks from CUT&Tag data using MACS2 (
Protocol 2: Functional Validation of an Insulating Element via CRISPR Deletion
- sgRNA Design: Design two sgRNAs flanking the putative lineage-specific insulating CTCF site (300-500 bp region). Include off-target control sgRNAs.
- Delivery: Transfect target cells with a Cas9-expressing plasmid and sgRNA pairs via nucleofection (e.g., Lonza 4D-Nucleofector).
- Clonal Isolation: 48-72 hours post-transfection, single-cell sort into 96-well plates. Expand clones for 2-3 weeks.
- Genotyping: Screen clones by PCR across the target locus and Sanger sequencing to identify homozygous deletions.
- Phenotypic Assay:
- Perform RT-qPCR for genes predicted to be misregulated (e.g., the oncogene now contacting a new enhancer).
- Conduct 4C-seq or promoter-focused Capture-C from the viewpoint of the affected gene in mutant vs. wild-type clones to confirm novel chromatin contacts.
- Assess functional outcomes (e.g., proliferation assay if an oncogene is activated).
Visualization of Pathways and Workflows
Title: Lineage-Specific CTCF Binding Drives Insulation
Title: Experimental Workflow for Variable Insulation Study
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Studying CTCF Binding and Insulation
| Item | Example Product/Catalog # | Function in Experiment |
|---|---|---|
| Validated Anti-CTCF Antibody (for ChIP/CUT&Tag) | Millipore Sigma, 07-729 | Immunoprecipitation of CTCF-bound chromatin for mapping binding sites. |
| In-situ Hi-C Kit | Arima Genomics, Arima-HiC Kit | Standardized reagents for high-quality, reproducible chromatin conformation capture libraries. |
| dCas9-DNMT3A/3L Fusion Plasmid | Addgene, #71685 (dCas9-DNMT3A) | Targeted DNA methylation to epigenetically disrupt CTCF binding at specific loci. |
| dCas9-TET1 Fusion Plasmid | Addgene, #84475 | Targeted DNA demethylation to potentially create new CTCF binding sites. |
| Cohesin Auxin-Inducible Degron Cell Line | Generated via CRISPR; RAD21-mAID | Rapid, reversible degradation of cohesin to acutely dissect its role in insulation. |
| High-Fidelity Cas9 & sgRNA Cloning Vector | Addgene, #48139 (pX330) | For precise CRISPR-Cas9 knockout of specific CTCF binding sites. |
| 4C-seq/Capture-C Kit | Custom oligo pools (IDT) & 3C kit (Diagenode) | Profiling chromatin interactions from specific viewpoints to validate insulation changes. |
| Lineage-Specific Cell Surface Marker Antibodies (for FACS) | e.g., CD34, CD45, NCAM | Isolation of pure, relevant cell populations for comparative studies. |
The architectural protein CTCF (CCCTC-binding factor) is a master regulator of three-dimensional genome organization, with its primary function being the insulation of enhancer-promoter interactions to ensure precise transcriptional regulation. Its role in forming the boundaries of topologically associating domains (TADs) and facilitating chromatin loops is well-established. However, the identification of these loops through high-throughput chromatin conformation capture techniques (e.g., Hi-C, Micro-C) and subsequent computational "loop callation" is fraught with technical artifacts. False positive loops, arising from algorithmic biases, experimental noise, and biological confounding factors, can lead to erroneous conclusions about enhancer-promoter insulation, directly impacting downstream research in gene regulation and drug target validation.
False positives in loop calling algorithms stem from multiple interrelated sources. A precise understanding of these is the first step toward mitigation.
Table 1: Major Sources of False Positives in Loop Callation
| Source Category | Specific Artifact | Impact on Loop Calling |
|---|---|---|
| Experimental Noise | PCR Duplicates & Chimeric Reads | Creates spurious, high-frequency ligation junctions that mimic loops. |
| Incomplete Digestion/Ligation Bias | Non-uniform background contact probability, causing regional false enrichments. | |
| Data Resolution & Coverage | Low Sequencing Depth | Insufficient statistical power to distinguish true signal from noise. |
| Algorithmic Biases | Distance-Based Bias Correction Failure | Over- or under-correction of the inherent distance-dependent contact decay. |
| Parameter Sensitivity (e.g., window size) | Over-merging of nearby peaks or detection of non-convergent peaks. | |
| Biological Confounders | "Sticky" Genomic Regions (e.g., highly transcribed) | Non-specific, high-frequency interactions independent of CTCF/cohesin. |
| Convergent CTCF Motifs Without Loop Formation | Occupancy without productive extrusion, leading to algorithm mis-identification. |
Experimental Protocols for Ground-Truth Validation
To assess and mitigate false positives, orthogonal experimental validation is mandatory. The following protocols are considered gold standards.
CRISPR-Deletion Followed by 4C-seq or Hi-C
Objective: To confirm the functional necessity of a predicted CTCF-mediated loop.
- Design gRNAs: Design two pairs of CRISPR-Cas9 gRNAs targeting the core CTCF binding sites at both putative anchor regions of the called loop.
- Generate Deletion Clones: Transfert cells with ribonucleoprotein complexes (RNPs) and isolate monoclonal cell populations via limiting dilution or FACS. Validate deletions via PCR and Sanger sequencing.
- Perform 4C-seq: For a focused view, use the viewpoint at one anchor to perform 4C-seq on both wild-type and deletion clones. Process cells with a primary restriction enzyme (e.g., DpnII) and a secondary cutter (e.g., Csp6I/NlaIII). Generate sequencing libraries from the circularized DNA.
- Perform Micro-C/Hi-C: For a genome-wide assessment, perform high-resolution (e.g., Micro-C) on wild-type and deletion clones. This assesses both the specific loop loss and broader TAD integrity.
- Analysis: A true positive loop will show a specific loss of the interaction signal between the deleted anchors in 4C-seq or Hi-C contact maps, without a global disruption of surrounding chromatin structure.
CTCF Degron-Depletion Coupled with Live-Cell Imaging
Objective: To dynamically correlate loop loss with CTCF removal and measure insulation decay.
- Generate Cell Line: Engineer a cell line expressing an auxin-inducible degron (AID) tag fused to endogenous CTCF (using homology-directed repair).
- Induce Depletion: Treat cells with auxin (IAA) to trigger rapid CTCF degradation (within ~30-60 minutes). Include a no-IAA control.
- Parallel Processing:
- Harvest for Hi-C: Collect cells at multiple time points (e.g., 0, 30min, 2h, 6h) post-auxin addition for in situ Hi-C.
- Live-Cell Imaging: In parallel, perform live-cell imaging of a fluorescently tagged genomic locus (e.g., via CRISPR imaging) to visualize the real-time loss of chromatin compaction or locus colocalization upon CTCF loss.
- Integration: Correlate the time-dependent loss of called loops in Hi-C data with the biophysical changes observed via imaging, confirming the direct role of CTCF in maintaining those specific interactions.
Algorithmic Mitigations and Best Practices
Table 2: Comparison of Advanced Loop Calling Algorithms & Mitigation Features
| Algorithm | Core Methodology | Key False Positive Mitigation Feature | Best Use Case |
|---|---|---|---|
| HiCCUPS (from Juicer) | Iterative correction + Poisson distribution modeling | Multiple normalization layers and statistical thresholds. | Standard in situ Hi-C data, robust for high-coverage datasets. |
| Mustache | Statistical learning on local contact matrices | Models expected contact probability from local neighborhood to define significance. | Sensitive detection in moderate-coverage data, less parameter tuning. |
| FitHiC2 | Non-parametric spline fitting for distance bias | Stratifies reads by genomic distance and applies a monotonic spline regression to model the background. | Focused on improving significance estimates for mid-to-long-range interactions. |
| Peakachu (Random Forest / Deep Learning) | Machine learning trained on ChIA-PET data | Learns complex patterns of true loops versus noise from orthogonal data. | Low-coverage Hi-C or single-cell Hi-C data. |
| SIP (Structure Inference Package) | Integrates epigenetic signals (CTCF, ChIP-seq) | Uses biological prior knowledge (e.g., convergent CTCF motifs) to weight loop calling. | Prioritizing biologically plausible, protein-mediated loops. |
Best Practice Workflow:
- Pre-processing & Mapping: Use dedicated pipelines (e.g., HiC-Pro, Juicer) that flag and remove PCR duplicates and chimeric reads.
- Normalization: Apply an iterative correction (ICE) or matrix balancing (KR) method to account for technical biases.
- Multi-Algorithm Call: Run at least two complementary algorithms (e.g., HiCCUPS and Mustache) on the same dataset.
- High-Confidence Set: Define a consensus loop set as the intersection of calls from multiple algorithms. This dramatically reduces false positives.
- Biological Filtering: Filter the consensus set for loops anchored at pairs of convergent CTCF motifs with strong ChIP-seq signal. This enriches for cohesin-mediated loops.
Visualizing the Experimental and Analytical Framework
Title: Loop Callation and False Positive Mitigation Workflow
Title: Orthogonal Validation Strategies for CTCF Loops
Table 3: Research Reagent Solutions for Loop Validation Studies
| Reagent / Resource | Function & Application | Key Consideration |
|---|---|---|
| dCas9-Degron Fusions (e.g., dCas9-AID, dCas9-SunTag-sfGFP-AID) | Targeted degradation of endogenous CTCF at specific anchor sites for locus-specific loop disruption studies. | Requires cloning and delivery of large constructs; control for off-target dCas9 binding. |
| Auxin (IAA) & OsTIR1 Stable Cell Lines | Rapid, inducible protein degradation system when used with AID-tagged proteins. Enables kinetic studies of loop decay. | Use non-leaky, high-efficiency OsTIR1-expressing lines. Optimize IAA concentration and timing. |
| High-Fidelity Restriction Enzymes (e.g., DpnII, MboI, Csp6I) | Critical for Hi-C/3C library preparation. Ensure complete digestion to minimize ligation bias artifacts. | Batch test enzyme activity; use high concentrations and extended digestion times. |
| Proximity Ligation Assay (PLA) Probes | In situ validation of specific chromatin loops via fluorescence microscopy. Uses antibodies against anchor-bound proteins (CTCF, RAD21). | Low throughput but provides single-cell, spatial validation. Requires high-quality antibodies. |
| CTCF Monoclonal Antibody (e.g., Millipore 07-729) | For ChIP-seq to map binding sites and filter loops, and for immunofluorescence/PLA validation. | Validate lot performance for ChIP-seq efficiency. Critical for biological filtering step. |
| Bioinformatic Pipelines (Juicer, HiC-Pro, Cooler) | Standardized processing of Hi-C data from raw reads to normalized contact matrices. Essential for reproducible loop calling. | Choose pipeline compatible with your sequencing protocol (e.g., HiC-Pro for standard Hi-C, Juicer for in situ Hi-C). |
Beyond CTCF: Validating Insulation Function and Comparing Architectural Proteins
The functional interplay between enhancers and promoters is precisely regulated to ensure accurate spatiotemporal gene expression. A cornerstone of this regulation is the insulation of genomic neighborhoods, a process central to broader research on enhancer-promoter communication. The zinc finger protein CCCTC-binding factor (CTCF), in conjunction with cohesin, is the principal architectural protein mediating the formation of topologically associating domain (TAD) boundaries and chromatin loops. The prevailing thesis posits that CTCF-driven loops are critical for insulating enhancer-promoter interactions, preventing aberrant crosstalk. This whitepaper establishes the gold standard experimental framework for directly testing this thesis by systematically correlating acute CTCF depletion with quantitative changes in 3D genome architecture (via Hi-C) and consequent transcriptional outcomes.
Core Mechanistic Framework and Signaling Pathways
CTCF's role in insulation is executed through a loop extrusion mechanism. Cohesin complexes are loaded onto chromatin and actively extrude DNA until they encounter convergently oriented CTCF binding sites, forming stable, anchored loops. These loops partition the genome into discrete regulatory units.
Diagram Title: Loop Extrusion Model for CTCF-Mediated Insulation
Key Experimental Protocols for Establishing Correlation
Protocol 1: Acute and Specific CTCF Depletion
- Method: Auxin-Inducible Degron (AID) Tagging of Endogenous CTCF.
- Procedure:
- Introduce an AID tag and an OsTIR1-expressing construct into the target cell line via CRISPR/Cas9-mediated knock-in.
- Treat cells with 500 µM indole-3-acetic acid (IAA, auxin) for a time-course (e.g., 0h, 3h, 6h, 24h). A control group receives solvent only.
- Harvest cells at each time point. Validate CTCF degradation efficiency (typically >90% by 6h) via western blot (anti-CTCF antibody) and quantitative immunofluorescence.
- Advantage: Enables rapid, reversible, and specific degradation, avoiding compensatory mechanisms seen in genetic knockouts.
Protocol 2: High-Resolution Hi-C for Loop Calling
- Method: In-situ Hi-C with deep sequencing.
- Procedure:
- Crosslink chromatin in ~1 million cells per condition (Control vs. CTCF-depleted) with 2% formaldehyde.
- Lyse cells, digest chromatin with a 4-cutter restriction enzyme (e.g., MboI or DpnII).
- Fill ends with biotinylated nucleotides and perform proximity ligation under dilute conditions.
- Reverse crosslinks, purify DNA, and shear to ~350 bp fragments.
- Pull down biotin-labeled ligation junctions with streptavidin beads for library construction.
- Sequence on an Illumina platform to achieve >1 billion valid read pairs per sample for high-resolution analysis (e.g., 5-10 kb binning).
- Analysis: Process reads using standardized pipelines (HiC-Pro, Juicer). Call chromatin loops using algorithms (HiCCUPS, FitHiC2). Differential loop analysis is performed with tools like diffHic or Selfish.
Protocol 3: Transcriptional Profiling and Enhancer Activity Assessment
- Method: RNA-seq and H3K27ac ChIP-seq.
- Procedure:
- Isolate total RNA from matched samples. Prepare poly-A selected libraries and sequence to a depth of ~40 million reads per sample. Map reads and quantify gene expression (TPM, DESeq2 for differential analysis).
- For ChIP-seq, crosslink and sonicate chromatin. Immunoprecipitate with an anti-H3K27ac antibody. Sequence libraries and call peaks (MACS2). Identify differential enhancer regions.
- Integration: Overlap differentially expressed genes (|log2FC| > 1, adj. p < 0.05) with genomic regions losing Hi-C loops/contacts and gaining aberrant H3K27ac signals.
Quantitative Data Synthesis
Table 1: Representative Quantitative Outcomes from CTCF Depletion Studies
| Assay | Control Condition | CTCF-Depleted Condition | Key Metric | Typical Magnitude of Change |
|---|---|---|---|---|
| Western Blot | Full-length CTCF protein | Degraded CTCF protein | CTCF Protein Level | >90% reduction at 6h post-auxin |
| In-situ Hi-C | Defined TAD boundaries | Eroded TAD boundaries | Boundary Insulation Score | 40-60% reduction at specific loci |
| Hi-C Loop Calls | ~10,000 significant loops | Loss of specific loops | Loop Contact Frequency | 50-80% decrease for affected loops |
| RNA-seq | Normalized gene expression | Dysregulated genes | Differentially Expressed Genes | Hundreds to thousands; both up & down |
| H3K27ac ChIP-seq | Focal enhancer peaks | Ectopic/aberrant peaks | Gained Enhancer Signals | At loci near lost loop anchors |
Table 2: Correlation Matrix: Loop Loss vs. Transcriptional Dysregulation
| Gene Locus | Associated Loop Strength (Control) | Loop Strength (ΔCTCF) | % Loop Loss | Gene Expression (Control, TPM) | Gene Expression (ΔCTCF, TPM) | Expression Fold Change | Predicted New Enhancer Contact |
|---|---|---|---|---|---|---|---|
| Gene A (Insulated) | 45.2 | 9.1 | -80% | 10.5 | 85.3 | +8.1x | Yes (via H3K27ac) |
| Gene B (Insulated) | 38.7 | 11.6 | -70% | 15.2 | 5.1 | -3.0x | Yes |
| Gene C (Non-Target) | N/A | N/A | N/A | 120.4 | 118.9 | ~1x | No |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for CTCF/3D Genome Functional Studies
| Item | Function/Application | Example Product/Catalog |
|---|---|---|
| Anti-CTCF Antibody | Validation of CTCF degradation (WB, IF) and ChIP-seq. | Cell Signaling Technology, #3418 (Rabbit mAb) |
| Auxin (IAA) | Inducer for AID-tagged protein degradation. | Sigma-Aldrich, I2886 |
| Hi-C Kit | Streamlined protocol for in-situ Hi-C library prep. | Arima Hi-C Kit, ARIMA50001 |
| Streptavidin Beads | Pulldown of biotinylated ligation junctions in Hi-C. | Dynabeads MyOne Streptavidin C1, Invitrogen |
| Anti-H3K27ac Antibody | Mapping active enhancers via ChIP-seq. | Abcam, ab4729 |
| DpnII Restriction Enzyme | Frequent-cutter for high-resolution Hi-C. | NEB, R0543M |
| OsTIR1 Plasmid | Expresses the E3 ubiquitin ligase for the AID system. | Addgene, #80074 (pCMV-OsTIR1) |
| CRISPR/Cas9 Tools | For endogenous tagging of CTCF with AID. | Synthetic gRNAs, Cas9 protein (IDT, Alt-R) |
Integrated Analysis Workflow
The gold standard requires integration of multi-omics data to establish direct causal relationships.
Diagram Title: Integrated Workflow from CTCF Loss to Phenotype
This guide outlines the definitive approach to mechanistically link CTCF loss to architectural and transcriptional phenotypes. By employing acute depletion, high-resolution Hi-C, and integrated genomics, researchers can move beyond correlation to establish causality, directly testing the core thesis of CTCF's indispensable role in enhancer-promoter insulation. This framework is critical for understanding disease-associated CTCF mutations and for evaluating therapeutic strategies aimed at modulating 3D genome architecture.
1. Introduction and Thesis Context
Within the framework of enhancer-promoter insulation research, CTCF is often regarded as the paradigmatic architectural protein in mammals. However, a comprehensive thesis on its role must account for its functional parallels and distinctions with other insulator-binding proteins (IBPs) across species, such as Drosophila melanogaster's BEAF-32 and Su(Hw). This whitepaper provides an in-depth technical comparison of these key IBPs, focusing on their mechanisms, genomic localization, and functional outcomes. Understanding these nuances is critical for researchers and drug development professionals aiming to manipulate genomic architecture for therapeutic purposes.
2. Core Mechanistic and Functional Comparisons
Insulators function primarily through two non-mutually exclusive mechanisms: enhancer-blocking (preventing inappropriate enhancer-promoter communication) and barrier activity (protecting genes from heterochromatic silencing). Different IBPs execute these functions through distinct mechanistic pathways.
Table 1: Quantitative Comparison of Key Insulator-Binding Proteins
| Feature | CTCF (Mammals) | Su(Hw) (Drosophila) | BEAF-32 (Drosophila) |
|---|---|---|---|
| DNA-Binding Motif | 11-Zinc Finger | 12-Zinc Finger | Novel Zinc Finger/Myb-like |
| Consensus Binding Sequence | ~20-50 bp, variable | ~24 bp (specific) | ~10-12 bp (CGATA motif) |
| Primary Partner Protein | Cohesin (RAD21, SMC1/3) | Mod(mdg4) | CP190, Chromator |
| Key Genomic Localization | TAD Boundaries, Promoters | Specific loci (e.g., gypsy retrotransposon), promoters | Promoters, especially housekeeping genes |
| Enhancer-Blocking Mechanism | Cohesin-Mediated Loop Extrusion Arrest | Direct Tethering to Nuclear Matrix | Formation of Specialized Chromatin Domains |
| Barrier Activity | Moderate, via histone modification recruitment | Strong, via recruitment of H3K4me3/H3K9ac modifiers | Strong, via prevention of heterochromatin spreading |
| Evolutionary Conservation | High (Vertebrates) | Low (Limited to Drosophilids) | Very Low (Limited to Drosophilids) |
3. Detailed Experimental Protocols
3.1. Chromatin Conformation Capture (3C) to Validate Insulator Function
- Purpose: To assess if a putative CTCF/BEAF-32/Su(Hw) site forms a chromatin loop boundary or insulates enhancer-promoter contacts.
- Protocol:
- Cross-linking: Treat cells with 1-3% formaldehyde for 10 min at room temperature to fix protein-DNA interactions.
- Lysis & Digestion: Lyse cells and digest chromatin with a restriction enzyme (e.g., HindIII) overnight.
- Ligation: Dilute digested chromatin to favor intramolecular ligation under T4 DNA ligase for 4 hours.
- Reversal & Purification: Reverse cross-links with Proteinase K, purify DNA.
- Quantitative Analysis: Design primers flanking the putative insulator and potential interacting regions. Perform quantitative PCR (qPCR) using SYBR Green. Interaction frequency is calculated relative to a control region.
3.2. ChIP-seq for IBP and Histone Modification Profiling
- Purpose: To map genomic binding sites of IBPs and associated chromatin marks.
- Protocol:
- Cross-linking & Sonication: Cross-link cells as in 3C. Sonicate chromatin to ~200-500 bp fragments.
- Immunoprecipitation: Incubate chromatin with validated antibody (anti-CTCF, anti-BEAF-32, anti-Su(Hw)) or control IgG overnight at 4°C. Capture antibody-chromatin complexes with Protein A/G beads.
- Wash & Elution: Wash beads stringently (e.g., high-salt wash). Elute complexes and reverse cross-links.
- Library Prep & Sequencing: Purify DNA, prepare sequencing library (end-repair, A-tailing, adapter ligation), amplify, and sequence on an Illumina platform.
- Data Analysis: Align reads to reference genome. Call peaks using tools like MACS2. Compare peaks with histone modification ChIP-seq datasets (e.g., H3K4me3 for active barriers, H3K9me3 for heterochromatin).
4. Visualizations of Key Pathways and Workflows
Title: CTCF-Cohesin Loop Extrusion Arrest Model
Title: Drosophila Insulator Protein Interaction Network
Title: ChIP-seq Experimental Workflow for IBPs
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Insulator Research
| Reagent/Material | Function/Application | Example/Notes |
|---|---|---|
| Validated ChIP-grade Antibodies | Specific immunoprecipitation of IBPs and histone marks for ChIP. | Anti-CTCF (Active Motif 61311), Anti-Su(Hw) (DSHB), Anti-BEAF-32 (DSHB). |
| CRISPR/Cas9 System | For generating knockout cell lines or deleting specific insulator sequences to study functional loss. | sgRNAs targeting CTCF binding motifs or IBP genes. |
| Chromatin Conformation Capture Kits | Standardized protocols for 3C, 4C, Hi-C assays. | Takara Bio Hi-C Kit, Merck 3C Kit. |
| Programmable DNA-Binding Domains (dCas9-Fusions) | Tethering proteins to specific loci to study insulator establishment. | dCas9-CTCF to test de novo boundary formation. |
| Nuclear Matrix Preparation Buffer | Isolate nuclear scaffolds to study tethering of insulator complexes (e.g., Su(Hw)-Mod(mdg4)). | Contains lithium diiodosalicylate (LIS) for extraction. |
| Reporter Assay Vectors | Functional testing of enhancer-blocking activity in vivo or in vitro. | Vectors with minimal promoter, reporter gene (luciferase), and flanking insulator test sites. |
The architectural protein CCCTC-binding factor (CTCF) is a master regulator of 3D chromatin organization, playing a non-redundant role in enhancer-promoter insulation via the formation of topologically associating domain (TAD) boundaries. The broader thesis of modern chromatin research posits that precise CTCF-mediated insulation is critical for lineage-specific gene expression programs. Consequently, the pathological disruption of CTCF-binding sites (CBS) represents a potent, genetically encoded mechanism for oncogenic reprogramming. This whitepaper analyzes how somatic mutations and structural variants (SVs) that specifically target CBS serve as a form of "disease as validation," providing direct in vivo evidence for the necessity of intact chromatin architecture in maintaining cellular homeostasis and suppressing tumorigenesis.
Mechanisms of CBS Disruption in Cancer Genomes
CTCF binding is sequence-specific, primarily to a ~15-20 bp motif. Disruption occurs via:
- Single Nucleotide Variants (SNVs) and Indels: Direct alteration of the core motif, ablating CTCF binding.
- Focal Deletions: Removal of the entire CBS or its flanking sequences.
- Copy-Number Neutral Structural Variants (Inversions, Translocations): Rearrangement that separates a CBS from its target gene or insulator element, or places a new enhancer within a now-permissive TAD.
- Epigenetic Silencing: While not a genetic lesion, hypermethylation of CpG dinucleotides within the CBS can also evict CTCF.
These alterations can lead to oncogene activation (e.g., by allowing a super-enhancer to contact a dormant proto-oncogene) or tumor suppressor gene silencing (e.g., by allowing an encroaching repressive domain).
Quantitative Landscape of CBS Disruptions Across Cancers
The following tables summarize key quantitative findings from pan-cancer analyses (sourced from recent studies including ICGC/PCAWG, TCGA, and newer cohort studies).
Table 1: Prevalence of Somatic Mutations in CBS Across Major Cancer Types
| Cancer Type | % of Tumors with CBS SNV/Indel | Recurrent CBS Hotspots (Example Genes Affected) | Avg. CBS Mutations per Tumor (SNV/Indel) |
|---|---|---|---|
| Colorectal Adenocarcinoma | ~12% | IGF2, WNT6, MYC | 0.8 |
| Lung Adenocarcinoma | ~8% | TERT, PDGFRA | 0.6 |
| Breast Invasive Carcinoma | ~5% | ESR1, MYC | 0.4 |
| Glioblastoma Multiforme | ~15% | PDGFRA, EGFR | 1.2 |
| Melanoma | ~10% | TERT promoter | 0.9 |
Table 2: Structural Variants Impacting CBS and Their Functional Consequences
| SV Type | Example Cancer | Target Locus | Consequence | Frequency in Cohort |
|---|---|---|---|---|
| Micro-deletion | Colorectal | chr11p15.5 (IGF2 ICR) | Loss of imprinting, IGF2 activation | 7% |
| Inversion | Medulloblastoma | chr2p (GFI1B/GFI1 enhancers) | Oncogene activation via enhancer hijacking | 5-10% of Group 3/4 |
| Translocation | T-ALL | chr7 (TCRB) to chr9 (NOTCH1) | NOTCH1 activation by TCR enhancer | Rare but recurrent |
| Tandem Duplication | Glioma | chr7 (EGFR) | Alters CBS spacing, creates new regulatory contacts | Common in EGFRvIII |
Table 3: Key Research Reagent Solutions
| Reagent / Material | Function in CBS Disruption Research |
|---|---|
| Anti-CTCF ChIP-grade Antibody | For chromatin immunoprecipitation to assess CTCF binding loss at mutated sites. |
| CBS Motif Wild-Type & Mutant Oligonucleotides | For EMSA (gel shift) assays to validate impact of mutation on in vitro CTCF binding. |
| Isogenic Cell Line Pairs (WT vs. CBS mutant) | Engineered via CRISPR-Cas9 to directly test the functional impact of a specific CBS mutation. |
| Capture Hi-C or HiChIP Kit | To map changes in 3D chromatin interactions (TAD boundaries, loops) following CBS disruption. |
| Dual-Luciferase Reporter Assay System | To test enhancer-promoter communication in insulator-defective vs. intact configurations. |
| Bisulfite Conversion Kit | To assay CpG methylation status at CBS, which can modulate CTCF binding independent of sequence. |
Experimental Protocols for Validating CBS Disruption
Protocol 4.1: Validating Loss of CTCF BindingIn Vitro
Title: EMSA for CTCF Motif Disruption Analysis
- Probe Preparation: Synthesize and biotin-label double-stranded DNA oligonucleotides spanning the wild-type and mutant CBS sequence.
- Protein Extract: Prepare nuclear extract from a cell line with robust CTCF expression (e.g., HEK293T).
- Binding Reaction: Incubate 5-20 fmol of labeled probe with 5-10 µg of nuclear extract in binding buffer (10 mM HEPES, 50 mM KCl, 1 mM DTT, 2.5 mM MgCl2, 10% glycerol, 0.05% NP-40, 1 µg poly(dI:dC)) for 20 min at room temperature.
- Competition (Specificity Control): Include reactions with 100-fold molar excess of unlabeled wild-type or mutant probe.
- Supershift: Add 1-2 µg of anti-CTCF antibody to confirm the identity of the DNA-protein complex.
- Electrophoresis: Run reactions on a pre-run 6% non-denaturing polyacrylamide gel in 0.5x TBE at 100V for 60-90 min.
- Detection: Transfer to a nylon membrane, crosslink, and detect biotin signal using a chemiluminescent kit.
Protocol 4.2: AssessingIn VivoCTCF Occupancy and Chromatin Architecture
Title: Integrated ChIP-seq & Hi-C to Map CBS Disruption Effects Part A: ChIP-seq for CTCF
- Crosslinking & Lysis: Crosslink cells (WT and mutant/isogenic model) with 1% formaldehyde for 10 min. Quench with glycine, lyse, and sonicate chromatin to 200-500 bp fragments.
- Immunoprecipitation: Incubate chromatin with anti-CTCF antibody conjugated to magnetic beads overnight at 4°C. Include an IgG control.
- Washing & Elution: Wash beads sequentially with low-salt, high-salt, LiCl, and TE buffers. Elute complexes and reverse crosslinks.
- Library Prep & Sequencing: Purify DNA, prepare sequencing library (end-repair, A-tailing, adapter ligation, PCR amplification), and sequence on an Illumina platform. Part B: In-Situ Hi-C for 3D Architecture
- Crosslinking & Digestion: Crosslink cells as above. Lyse nuclei and digest chromatin with a 4-cutter restriction enzyme (e.g., MboI or DpnII).
- Marking & Proximity Ligation: Fill overhangs with biotinylated nucleotides and perform proximity ligation under dilute conditions to favor intra-molecular ligation.
- Purification & Shearing: Reverse crosslinks, purify DNA, and shear to ~350 bp. Pull down biotin-labeled ligation junctions with streptavidin beads.
- Library Prep & Sequencing: Prepare sequencing library directly on beads, then sequence paired-ends.
- Analysis: Map reads, filter for valid interaction pairs, and construct contact matrices. Call TADs (e.g., using Arrowhead algorithm) and compare boundaries between conditions.
Protocol 4.3: Functional Assay for Altered Enhancer-Promoter Communication
Title: STARR-seq Assay for Enhancer Hijacking Detection
- Library Construction: Clone genomic fragments (e.g., from a region spanning a CBS SV) into a plasmid downstream of a minimal promoter and upstream of a reporter gene (e.g., GFP).
- Transfection: Transfect the library into a relevant cancer cell line (carrying the CBS mutation/SV) and a control line.
- FACS Sorting & Sequencing: After 48h, sort cells based on reporter activity (High vs. Low GFP). Isolve plasmid DNA from sorted populations.
- Quantification: Use high-throughput sequencing to quantify the abundance of each genomic fragment in the high-activity pool vs. the input library. Fragments with significantly higher enrichment in the mutant background represent hijacked enhancers.
Visualizations
Diagram Title: CTCF Insulation Loss Leading to Oncogenic Enhancer Hijacking
Diagram Title: Computational Pipeline for Identifying CBS-Disrupting Variants
This whitepaper examines the evolutionary conservation of CTCF binding sites and their functional role in chromatin insulation and three-dimensional genome architecture. Framed within the broader thesis on CTCF's role in enhancer-promoter insulation, we assess how conserved sequence motifs translate to conserved topological and regulatory functions across species, from invertebrates to mammals. Understanding this conservation is critical for interpreting non-coding genetic variation and for developing targeted therapeutic interventions in gene regulation.
Core Principles of CTCF Conservation
CTCF (CCCTC-binding factor) is an 11-zinc finger DNA-binding protein that plays a pivotal role in genome organization by mediating chromatin looping, serving as a barrier insulator, and demarcating topologically associating domain (TAD) boundaries. Its binding site, a ~20-50 bp sequence motif, is highly conserved, but the degree of functional conservation of the associated insulation activity is more variable and context-dependent.
Quantitative Data on CTCF Site Conservation
The following table summarizes key comparative genomics data on CTCF conservation.
Table 1: Conservation Metrics for CTCF Sites and Function Across Species
| Species Comparison | % CTCF Sites with Orthologous Sequence | % Conserved Sites with Functional Insulation Activity (by Assay) | Typical Assay for Insulation Function | Reference Key Findings |
|---|---|---|---|---|
| Human - Mouse | ~60-70% | ~40-50% (Hi-C/TAD boundary assay) | Hi-C, STARR-seq, Enhancer-blocking assay | Core motif essential, but cofactor (cohesin) dynamics differ. Many TAD boundaries conserved. |
| Human - Chicken | ~40% | ~20-30% (Enhancer-blocking) | Reporter assays in hybrid cells | Insulation function less conserved than binding; dependent on genomic context. |
| Mammals - Drosophila (BEAF-32/CTCF) | <10% (sequence) | N/A (Different protein) | Hi-C, FISH | Architectural role convergent; BEAF-32/Su(Hw) fulfill analogous insulator roles. |
| Vertebrate - Invertebrate (CTCF ortholog) | ~15-25% in C. elegans | Demonstrated in specific loci | 4C, CRISPR deletion | CTCF orthologs can establish chromatin boundaries but with different partner proteins. |
Experimental Protocols for Assessing Conservation
Protocol 1: Identifying Conserved CTCF Sites (Bioinformatics Pipeline)
Objective: To identify evolutionarily conserved CTCF binding sites across multiple species.
- Data Acquisition: Download ChIP-seq peaks for CTCF for species of interest (e.g., human, mouse, dog) from public repositories (ENCODE, NCBI GEO).
- Peak Center Annotation: Extract summit coordinates (±250 bp) for each peak.
- LiftOver: Use cross-species genomic alignment tools (e.g., UCSC LiftOver) to map peak coordinates from the reference genome to a target genome. Require a minimum chain score (e.g., >0.5) for a successful conversion.
- Motif Analysis: Scan conserved genomic intervals for the presence of the canonical CTCF motif (JASPAR MA0139.1) using FIMO (MEME Suite) with a p-value threshold (e.g., p < 1e-5).
- Validation: Compare computationally lifted peaks with experimentally determined CTCF peaks in the target species. Calculate the percentage overlap (e.g., using BEDTools intersect).
Protocol 2: Functional Validation of Insulation Using CRISPR/Cas9 Deletion
Objective: To test if a conserved CTCF site is necessary for enhancer-promoter insulation in vivo.
- Guide RNA Design: Design two sgRNAs flanking the conserved CTCF site (~500-2000 bp deletion).
- Cell Transfection: Transfect target cells (e.g., mouse embryonic stem cells) with a plasmid expressing Cas9 and the two sgRNAs.
- Clonal Selection: Isolate single-cell clones and expand. Genotype by PCR and Sanger sequencing to identify homozygous deletions.
- Phenotypic Assessment:
- Hi-C: Perform in-situ Hi-C on mutant and wild-type clones. Analyze for changes in TAD boundary strength at the deletion site.
- 3C-qPCR: Design primers to test specific enhancer-promoter interactions hypothesized to be insulated by the site.
- Gene Expression: Perform RNA-seq or RT-qPCR on genes near the deletion to assess dysregulation due to loss of insulation.
Protocol 3: Cross-Species Insulation Assay (Reporter Assay)
Objective: To test if a CTCF site from one species retains insulation function in the cellular context of another.
- Cloning: Amplify the genomic region containing the CTCF site from Species A. Clone it into a standard enhancer-blocking reporter vector (e.g., pNI vector) between an enhancer and a promoter driving a luciferase reporter.
- Cell Culture and Transfection: Culture cells from Species B. Co-transfect the reporter construct with a Renilla luciferase control plasmid.
- Measurement: After 48 hours, perform a dual-luciferase assay. Normalize firefly luciferase activity to Renilla.
- Analysis: Compare activity of the construct with the CTCF site insert to a positive control (insulator present) and a negative control (no insulator). Reduced reporter activity indicates insulation function.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CTCF Conservation and Function Studies
| Reagent / Material | Function in Research | Example Product / Assay |
|---|---|---|
| Anti-CTCF Antibodies | Chromatin immunoprecipitation (ChIP) to map endogenous CTCF binding sites across species. | Millipore 07-729 (rabbit monoclonal), Active Motif 61311. |
| Cross-species Genomic Alignment Files | Bioinformatics lifting of coordinates between genomes. | UCSC LiftOver chain files (e.g., hg38ToMm39.over.chain.gz). |
| Insulator Reporter Vectors | Functional testing of sequence's enhancer-blocking capability in vitro. | pNI (Neomycin Insulator) Vector, pGL4.23-based custom vectors. |
| Dual-Luciferase Reporter Assay System | Quantitative measurement of enhancer-blocking in reporter assays. | Promega Dual-Luciferase Reporter (DLR) Assay System (E1910). |
| Hi-C Kit | Assessing genome-wide chromatin architecture and TAD boundary strength. | Arima-HiC+ Kit, Dovetail Omni-C Kit. |
| CRISPR/Cas9 Knockout Kits | Generating precise deletions of CTCF sites in cell lines for functional loss-of-function studies. | Synthego synthetic sgRNA + Cas9 3NLS, IDT Alt-R CRISPR-Cas9 System. |
| Phylogenetically Diverse Cell Lines | For cross-species functional assays. | ATCC collections: Human (HEK293T), Mouse (mESC), Dog (MDCK), Chicken (DT40). |
Visualizing Key Concepts and Workflows
Diagram 1: Workflow for Assessing CTCF Conservation
Diagram 2: CTCF/Cohesin Mediated Insulation Across Species
The assessment of CTCF site and insulation function across species reveals a core conserved mechanism—CTCF-cohesin mediated loop extrusion—that is adaptable and context-dependent. While the architectural role is ancient, the specific genomic implementation varies. For researchers and drug development professionals, this implies that:
- Disease Modeling: Genomic insulators in animal models may not perfectly recapitulate human function, requiring careful validation.
- Target Identification: Non-coding variants in conserved CTCF sites are high-priority candidates for causative roles in developmental disorders and cancer.
- Therapeutic Intervention: Disrupting specific, disease-relevant chromatin loops by targeting CTCF binding or cohesion function presents a novel, though challenging, epigenetic therapy avenue. Understanding conservation is key to evaluating potential on-target toxicities across biological systems.
Complementary or Redundant? Evaluating the Roles of CTCF and Chromatin Modifiers (e.g., PRC2) in Gene Silencing
Understanding the interplay between architectural proteins and chromatin modifiers is central to modern epigenetics. Within the broader context of CTCF's canonical role in enhancer-promoter insulation, its functional relationship with silencing machinery like the Polycomb Repressive Complex 2 (PRC2) is complex. This guide examines whether these systems act in complementary, synergistic, or redundant pathways to establish and maintain gene silencing, a critical consideration for manipulating gene expression in disease and therapy.
Core Mechanisms: CTCF and PRC2
CTCF (CCCTC-Binding Factor): A zinc-finger protein primarily known for its role in forming topologically associating domain (TAD) boundaries and insulating enhancer-promoter interactions. Its silencing function is often indirect, stemming from this insulation activity.
PRC2 (Polycomb Repressive Complex 2): A histone methyltransferase complex that catalyzes the trimethylation of histone H3 at lysine 27 (H3K27me3), a canonical repressive chromatin mark associated with facultative heterochromatin and stable gene silencing.
Table 1: Comparative Roles in Gene Silencing
| Feature | CTCF | PRC2 |
|---|---|---|
| Primary Biochemical Function | DNA binding, architectural protein, insulator | Histone methyltransferase (H3K27me3) |
| Direct Silencing Mechanism | Limited; via blocking enhancer access | Direct; deposition of repressive histone mark |
| Genomic Localization | TAD boundaries, insulator elements, promoters | CpG islands, promoters of developmentally regulated genes |
| Effect on Chromatin State | Can partition active and repressive domains | Establishes and maintains facultative heterochromatin |
| Temporal Dynamics | Relatively stable, constitutive binding | Dynamic during development; stable maintenance |
| Co-localization Frequency | ~20-30% of PRC2-bound sites in mammalian ESCs (source: recent ChIP-seq meta-analyses) | Subset overlaps with CTCF sites, often at insulated borders of Polycomb domains |
Table 2: Experimental Outcomes from Perturbation Studies
| Experiment | CTCF Depletion Impact | PRC2 (EZH2) Depletion Impact | Dual Depletion Impact |
|---|---|---|---|
| Hox Gene Clusters | Ectopic enhancer contacts, partial derepression | Strong derepression, loss of H3K27me3 | Synergistic derepression, complete loss of topological boundaries |
| Imprinted Control Regions | Loss of allele-specific insulation and silencing | Variable; some regions unaffected | Often additive, suggesting independent pathways |
| Genome-wide H3K27me3 Levels | Minimal direct change | Drastic global reduction | Similar to PRC2 depletion alone |
| TAD Boundary Integrity | Severe disruption, boundary erosion | Minor local changes at co-bound sites | Exacerbated boundary loss compared to CTCF depletion alone |
Key Experimental Protocols
ChIP-seq for Mapping Co-occupancy
Objective: To identify genomic sites where CTCF and PRC2 core subunits (e.g., SUZ12, EZH2) co-localize.
- Crosslinking: Treat cells (e.g., mESCs) with 1% formaldehyde for 10 min at room temperature.
- Cell Lysis & Chromatin Shearing: Lyse cells and sonicate chromatin to ~200-500 bp fragments.
- Immunoprecipitation: Incubate chromatin with antibodies against CTCF and SUZ12/EZH2 in separate reactions. Use Protein A/G magnetic beads.
- Washing & Elution: Wash beads stringently, elute complexes, and reverse crosslinks.
- Library Prep & Sequencing: Purify DNA, prepare sequencing libraries, and perform high-throughput sequencing (Illumina).
- Analysis: Map reads, call peaks (using MACS2), and identify overlapping peaks.
Auxin-Inducible Degron (AID) System for Acute Depletion
Objective: To rapidly deplete CTCF and/or EZH2 and assess immediate effects on silencing and topology.
- Cell Line Engineering: Generate mESC lines expressing osTIR1 and AID-tagged fusions of CTCF and/or EZH2.
- Acute Degradation: Treat cells with 500 µM auxin (IAA) for 1-6 hours.
- Validation: Confirm depletion via western blot (CTCF, EZH2, H3K27me3).
- Downstream Assays: Perform RNA-seq (gene expression), ChIP-seq (H3K27me3, CTCF occupancy), and Hi-C (chromatin architecture) on depleted vs. control cells.
4C-seq to Assess Enhancer-Promoter Interactions
Objective: To measure changes in specific chromatin interactions (e.g., at a Hox cluster) upon perturbation.
- Crosslinking & Digestion: Crosslink cells, lyse, and digest chromatin with a primary restriction enzyme (e.g., DpnII).
- Proximity Ligation: Dilute and ligate under conditions favoring intramolecular ligation.
- Reverse Crosslinking & Secondary Digestion: Purify DNA, digest with a second enzyme.
- Circularization & PCR: Ligate to form circles, perform inverse PCR with bait-specific primers.
- Sequencing & Analysis: Sequence PCR products and map interaction frequencies from the bait viewpoint.
Visualizations
Diagram 1: CTCF & PRC2 in Silencing
Diagram 2: Acute Depletion Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Materials
| Reagent/Material | Provider Examples | Primary Function in Analysis |
|---|---|---|
| Anti-CTCF Antibody (ChIP-grade) | Active Motif, Cell Signaling Technology, Abcam | Immunoprecipitation of CTCF for ChIP-seq to map binding sites. |
| Anti-H3K27me3 Antibody (ChIP-grade) | MilliporeSigma, Diagenode, Abcam | Detection of PRC2-mediated repressive histone mark. |
| Anti-SUZ12/EZH2 Antibody (ChIP-grade) | Cell Signaling Technology, Active Motif | Immunoprecipitation of PRC2 complex components. |
| Auxin-Inducible Degron (AID) System | Takahashi lab (original), Addgene plasmids | For rapid, conditional degradation of target proteins (CTCF, EZH2). |
| Trimethylated H3K27 Peptide Standards | Active Motif, EpiCypher | Controls and competitors for specificity validation in ChIP. |
| DpnII, HindIII, other Restriction Enzymes | NEB, Thermo Fisher | Chromatin digestion for 3C-based methods (4C-seq, Hi-C). |
| Proximity Ligation Reagents | Thermo Fisher, NEB, homemade ligation buffer | Facilitates ligation of crosslinked, digested chromatin fragments. |
| CTCF Motif Mutagenesis Kits (CRISPR-Cas9) | Synthego, IDT, ToolGen | To disrupt specific CTCF sites and study loss-of-function effects. |
| EZH2 Inhibitors (e.g., GSK126, EPZ-6438) | Selleckchem, Cayman Chemical | Pharmacological inhibition of PRC2 catalytic activity. |
The investigation of CCCTC-binding factor (CTCF) is central to understanding the three-dimensional architecture of the mammalian genome and its regulation of gene expression. Within the broader thesis on CTCF's role in enhancer-promoter insulation, a critical challenge is the accurate identification and functional assessment of non-canonical or putative CTCF binding sites. Not all genomic sequences containing a CTCF motif are functionally equivalent; their binding strength and consequent ability to act as insulator elements and form chromatin loop boundaries exhibit significant quantitative variation. This whitepaper details the development and application of quantitative predictive models that move beyond simple motif presence/absence to algorithmically score putative sites for their binding affinity and predicted insulatory potential. These models are essential for interpreting non-coding genetic variation, understanding disease-associated genomic rearrangements, and predicting the outcomes of genetic engineering in therapeutic contexts.
Core Algorithmic Approaches & Quantitative Data
Modern predictive models integrate multiple genomic and epigenomic features. The table below summarizes key features used in state-of-the-art algorithms and their quantitative contribution to predicting site strength.
Table 1: Genomic Features for Predictive Modeling of CTCF Site Strength
| Feature Category | Specific Feature | Data Type / Source | Quantitative Contribution (Typical Weight Range) | Rationale |
|---|---|---|---|---|
| Primary Sequence | Core Motif Score | Position Weight Matrix (PWM) Match (e.g., JASPAR MA0139.1) | High (0.3-0.5) | Fidelity to the 20bp consensus determines base-level binding energy. |
| Motif Flanking Sequence | k-mer composition / DNA shape features | Medium (0.1-0.2) | Adjacent sequences influence DNA flexibility and protein docking. | |
| Chromatin Context | DNase I Hypersensitivity (DHS) | Signal intensity from ATAC-seq or DNase-seq | High (0.2-0.4) | Open chromatin is prerequisite for factor accessibility. |
| Histone Marks | ChIP-seq signals for H3K4me3, H3K27ac, H3K9me3 | Medium (0.1-0.3) | Active promoter/enhancer marks (H3K4me3, H3K27ac) correlate with functional binding; heterochromatin marks (H3K9me3) are anti-correlated. | |
| Cohort Binding Data | Conservation across Cell Types | Aggregated CTCF ChIP-seq from ENCODE/Roadmap | High (0.3-0.4) | Sites bound across diverse cell types (constitutive) are more likely to be strong, canonical insulators. |
| Binding Profile Shape | ChIP-seq peak shape metrics (e.g., summit sharpness) | Medium (0.1-0.2) | Sharp, high-intensity peaks indicate high-affinity binding. | |
| 3D Architecture | Loop Anchor Overlap | Overlap with Hi-C/ChIA-PET defined TAD boundaries | Medium-Low (0.05-0.15) | Functional insulators often coincide with topological domain borders. |
Advanced models, such as convolutional neural networks (CNNs) or gradient boosting machines (e.g., XGBoost), are trained on large-scale CTCF ChIP-seq datasets. Performance metrics for these models are summarized below.
Table 2: Performance of Predictive Model Architectures
| Model Type | Training Dataset (Example) | Primary Output | Typical AUC-PR | Key Advantage |
|---|---|---|---|---|
| Logistic Regression | CTCF ChIP-seq peaks vs. shuffled motifs | Binary classification (bound/unbound) | 0.70 - 0.85 | Interpretable feature weights. |
| Gradient Boosting (XGBoost) | ENCODE consensus peak set with matched features | Probabilistic score (0-1) for binding strength | 0.88 - 0.94 | Handles non-linear feature interactions effectively. |
| Convolutional Neural Network (CNN) | Genomic sequence windows (±250bp) with associated ChIP signal | Binding intensity prediction | 0.90 - 0.96 | Can learn complex sequence motifs and patterns de novo. |
| Multi-modal Network | Integrated sequence, chromatin, and conservation data | Unified "Insulation Score" | 0.92 - 0.97 | Holistic prediction of functional outcome. |
Experimental Protocols for Validation
Predicted sites require empirical validation. Below are detailed protocols for key validation experiments cited in related research.
Protocol 3.1:In VitroValidation by Electrophoretic Mobility Shift Assay (EMSA)
Purpose: To quantitatively assess the protein-DNA binding affinity of a putative CTCF site. Methodology:
- Probe Preparation: Synthesize complementary biotinylated oligonucleotides spanning the putative CTCF site (≥40bp, site centered). Anneal to form double-stranded probes.
- Protein Extract: Prepare nuclear extract from a relevant cell line (e.g., HEK293T, K562) or use purified recombinant CTCF zinc finger domain.
- Binding Reaction: Incubate 20 fmol of labeled probe with 5-20 µg of nuclear extract in binding buffer (10 mM HEPES, 50 mM KCl, 1 mM DTT, 2.5% glycerol, 50 ng/µL poly(dI·dC)) for 30 minutes at room temperature.
- Competition Assay: Include unlabeled wild-type or mutant oligonucleotides in molar excess (10x-100x) to demonstrate binding specificity.
- Electrophoresis: Resolve protein-DNA complexes on a pre-run, non-denaturing 6% polyacrylamide gel in 0.5x TBE at 100V for 60-90 minutes at 4°C.
- Detection: Transfer to a nylon membrane, cross-link, and detect biotin-labeled DNA using a chemiluminescent substrate. Quantify band intensity to derive apparent dissociation constants (Kd).
Protocol 3.2:In VivoValidation by CRISPR/Cas9 Deletion and 4C-seq
Purpose: To functionally test the insulatory potential of a predicted CTCF site by assessing changes in chromatin architecture upon its deletion. Methodology:
- sgRNA Design & Cloning: Design two sgRNAs flanking the target CTCF site. Clone into a Cas9-expressing plasmid (e.g., pSpCas9(BB)-2A-Puro).
- Cell Line Engineering: Transfect target cell line. Select with puromycin (1-2 µg/mL for 48h). Isolate single-cell clones and screen by PCR and Sanger sequencing to identify homozygous deletions.
- 4C-seq Library Preparation:
- Crosslinking & Digestion: Fix 10 million wild-type and knockout cells with 2% formaldehyde. Lyse and perform primary restriction digest with a 6-cutter (e.g., DpnII).
- Proximity Ligation: Dilute and ligate under conditions favoring intramolecular ligation.
- Reverse Crosslinking & Secondary Digestion: Purify DNA, reverse crosslinks, and perform a secondary digest with a 4-cutter (e.g., NlaIII).
- Second Ligation & Amplification: Ligate to NlaIII-compatible adapters and perform inverse PCR using primers designed for a "viewpoint" gene of interest near the deleted site. Sequence the resulting library.
- Data Analysis: Map reads, generate contact frequency profiles from the viewpoint. A loss of insulation manifests as the appearance of new, ectopic contacts between the enhancer and promoter across the deleted boundary, quantified by changes in contact frequency.
Visualizations
Title: Predictive Model Feature Integration Pipeline
Title: Experimental Validation of Insulation Loss via Deletion
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for CTCF Site Analysis
| Item | Function / Application | Example Product / Catalog Number |
|---|---|---|
| Anti-CTCF Antibody (ChIP-grade) | Immunoprecipitation of CTCF-bound chromatin for ChIP-seq validation of predicted sites. | Cell Signaling Technology #2899; Active Motif #61311. |
| Recombinant CTCF Protein (ZF domain) | In vitro binding assays (EMSA, SELEX) to measure binding affinity without nuclear extract complexity. | Abcam ab165091; homemade purification from E. coli. |
| Biotinylated Oligonucleotide Probes | Sensitive detection of protein-DNA complexes in EMSA without radioactivity. | Custom synthesis from IDT or Sigma. |
| CRISPR/Cas9 Knockout Kit | Generation of clonal cell lines with deletions of putative CTCF sites for functional validation. | Synthego (sgRNA design/ synthesis); Addgene plasmid #62988 (pX330). |
| 4C-seq Kit / Components | Mapping chromatin contacts from a specific viewpoint to assess insulation changes. | CoolMPS 4C-seq kit (Precision Genomics); custom protocols with DpnII, NlaIII, T4 DNA Ligase. |
| CTCF Motif Position Weight Matrix | Core sequence model for initial site scanning and feature generation. | JASPAR MA0139.1; HOCOMOCO v11. |
| Pre-trained CTCF Binding Prediction Model | Starting point for scoring novel genomic sequences. | Basenji2, DeepBind CTCF models (available on GitHub). |
| Cell Line-Specific CTCF ChIP-seq Data | Positive control datasets for model training and benchmarking. | ENCODE Portal (e.g., K562, GM12878, HepG2). |
| DNase I or ATAC-seq Kit | Profiling open chromatin as a critical input feature for predictive models. | Illumina DNase-seq kit; 10x Genomics ATAC-seq kit. |
Conclusion
CTCF-mediated enhancer-promoter insulation is a cornerstone of three-dimensional genome organization, ensuring precise spatiotemporal gene control. From foundational mechanisms involving cohesin-driven loop extrusion to advanced methodologies for boundary manipulation, our understanding has profound implications. Troubleshooting experiments requires careful consideration of redundancy and assay specificity, while validation through comparative analysis and disease genomics underscores its critical non-redundant functions. Looking forward, integrating single-cell multi-omics and high-resolution structural data will refine our models. For biomedical research, the druggability of the CTCF-cohesin axis presents a promising, albeit challenging, frontier for treating diseases driven by epigenetic dysregulation, such as cancer and neurodevelopmental disorders, by rewriting pathogenic gene expression programs.